Lying in bed exhausted but unable to fall asleep — or waking at 3am with a racing mind despite being depleted — is a commonly reported experience of perimenopause. This has a measurable physiological basis in the HPA (hypothalamic-pituitary-adrenal) axis and in how reproductive hormones regulate cortisol output.
This article covers the HPA axis mechanism behind the wired-but-tired state, the cortisol curve flattening that produces it, and the autonomic component that sustains it. It does not cover the full spectrum of hormonal sleep disruption — for that broader context, see Hormonal Sleep Disruption in Women. HPA dysregulation is one pathway among several that affect sleep during menopause, and understanding it can clarify why this particular pattern feels so contradictory.
Does Menopause Disrupt the Hypothalamic-Pituitary-Adrenal Axis?
The HPA axis is the cortisol-regulating pathway. It works as a cascade: the hypothalamus releases CRH (corticotropin-releasing hormone), which prompts the pituitary to release ACTH (adrenocorticotropic hormone), which tells the adrenal glands to produce cortisol. Under normal conditions, cortisol feeds back to the hypothalamus and pituitary to limit further CRH release — a negative feedback loop that keeps cortisol within range.
Progesterone plays a regulatory role in this loop through its metabolite allopregnanolone. Allopregnanolone is a potent modulator of GABA-A receptors — the same receptor class that benzodiazepines target. When allopregnanolone binds to GABA-A receptors in the hypothalamus, it provides tonic (ongoing) inhibition of CRH neurons, dampening HPA axis output. This is one reason progesterone has a calming effect.
Gordon et al. (2015) proposed the model for how this changes in perimenopause: as progesterone declines, allopregnanolone levels drop, GABA-A-mediated inhibition of the HPA axis weakens, and the axis becomes disinhibited — meaning it produces more cortisol with less provocation. This is a structural change in hormonal regulation, not a response to life stress.
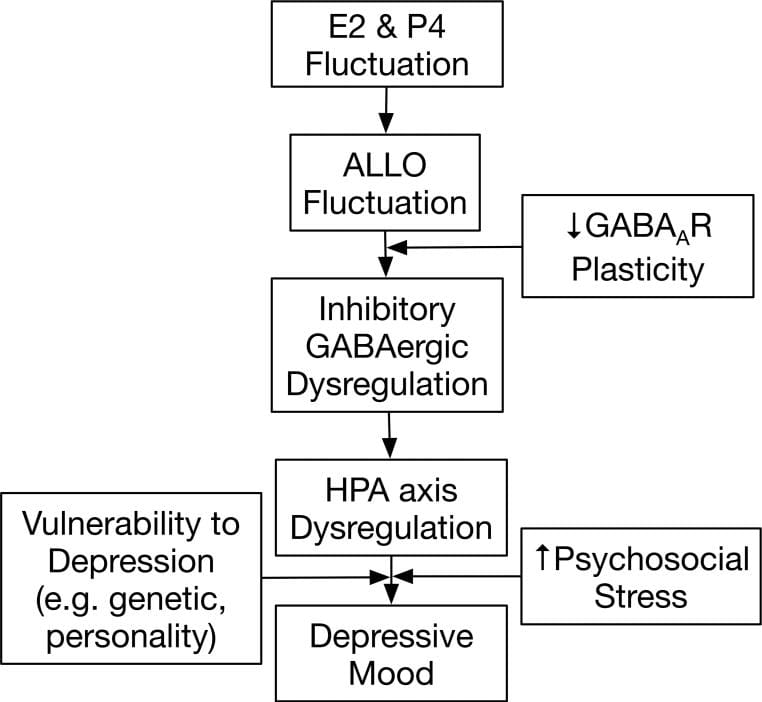
Woods et al. (2009) reinforced this distinction: overnight cortisol elevation in perimenopausal women was driven by reproductive hormonal changes, not by psychosocial stress levels. Women with higher perceived stress did not show proportionally higher overnight cortisol — the elevation tracked with hormonal status instead.
Troia et al. (2025) reported that 80-90% of perimenopausal women experience sleep disturbance, with overlapping mechanisms including HPA axis changes, vasomotor episodes, and mood disruption. The HPA pathway is one contributor within a broader hormonal picture — but it is the driver of the wired-but-tired pattern in particular, because it determines cortisol timing.
Why Are You Exhausted During the Day but Wired at Night?
Cortisol follows a 24-hour rhythm called the diurnal cortisol curve. In a well-regulated pattern, cortisol peaks within 30-45 minutes of waking — this is the cortisol awakening response (CAR), which provides the alertness and energy needed to start the day. Cortisol then declines throughout the afternoon and reaches its lowest point in the late evening, which allows melatonin to rise and sleep onset to proceed.
When the HPA axis is disinhibited — as it becomes in perimenopause — this curve flattens. Evening cortisol stays elevated, and the morning CAR is blunted. The consequences map onto the wired-but-tired experience: elevated evening cortisol produces arousal at the wrong time (wired at night), while a suppressed morning CAR produces insufficient energy at wake time (exhausted during the day).
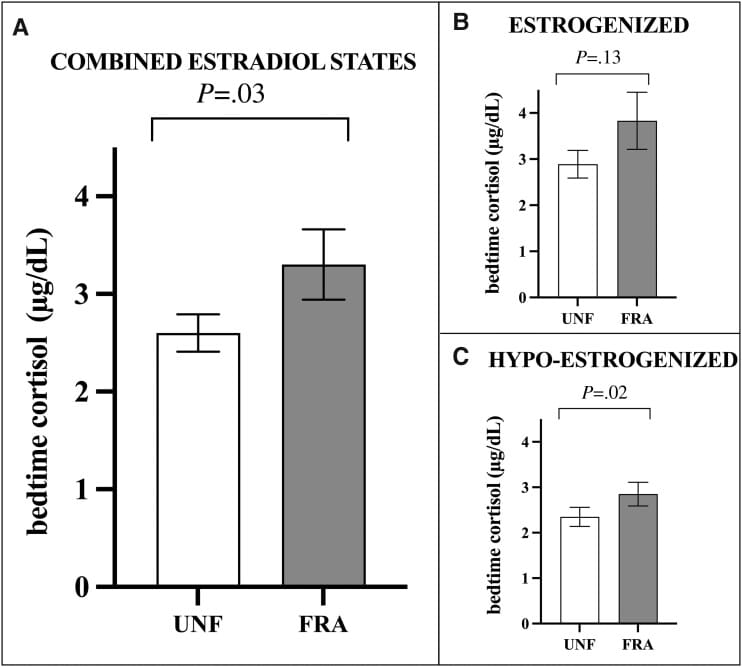
Cohn et al. (2023) quantified this in an experimental human model. Using a GnRH agonist to suppress estradiol (mimicking the hormonal state of menopause), they measured cortisol under conditions of normal sleep and fragmented sleep. Sleep fragmentation raised bedtime cortisol by 27% (P = .03) and suppressed the morning cortisol awakening response by 57%. The effect was amplified in the hypo-estrogenized state — low estradiol made the cortisol disruption worse (P = .02 for bedtime cortisol in the hypo-estrogenized condition).
The “crashing fatigue” that many perimenopausal women describe in the afternoon follows from this same pattern. A blunted CAR means less cortisol-driven alertness from the moment of waking, and by mid-afternoon, the deficit accumulates into exhaustion. Yet evening cortisol remains elevated — so the fatigue coexists with an inability to fall asleep or stay asleep.
This is the core of the paradox: the exhaustion is from low morning cortisol. The inability to sleep is from high evening cortisol. Both are present simultaneously because the cortisol curve has flattened — it is a dysregulated rhythm, not a contradiction.
Dressle et al. (2022) confirmed this at scale in a meta-analysis of 31 case-control studies: individuals with chronic insomnia showed moderately elevated cortisol compared to controls (standardized mean difference = 0.50, with a stronger effect of 0.67 in blood-based cortisol measurements). Chronic insomnia and HPA hyperactivation are associated — and in perimenopausal women, the hormonal changes provide a mechanism for that association.
Does Perimenopause Cause Anxiety at Night?
The nighttime anxiety that accompanies perimenopause involves two converging hormonal pathways. Estrogen supports serotonin synthesis and receptor availability — when estrogen declines, serotonergic tone drops, which affects mood regulation, sleep-wake cycling, and the ability to transition between arousal states. Separately, progesterone loss reduces GABA-A-mediated inhibition, lowering the threshold at which autonomic arousal activates. Together, these pathways produce a lower bar for activation and a reduced capacity to return to a calm baseline.
Garg and Munshi (2025) reviewed both pathways and reported a 56% increase in insomnia prevalence in menopausal women compared to premenopausal women. The review connected estrogen decline to reduced serotonin production (affecting sleep-wake regulation) and noted that both estrogen and progesterone modulate GABA, with their decline reducing the calming capacity that supports sleep onset and maintenance.
de Zambotti et al. (2016) provided experimental evidence of how this manifests in autonomic regulation. In their study, perimenopausal women with insomnia were exposed to a cognitive stressor before sleep and then monitored with polysomnography. Control participants recovered parasympathetic (vagal) tone during sleep within 4-6 hours — their autonomic activity returned to a rest-favoring state. Perimenopausal women with insomnia showed no parasympathetic recovery throughout the entire night. Their autonomic activity remained in sympathetic dominance — elevated heart rate, heightened alertness, increased cortisol output — from bedtime through waking.
This sustained sympathetic activation is the physiological basis of what many women describe as a “racing mind” or nighttime dread. The experience is often attributed to psychological anxiety, but the de Zambotti data suggests it is arousal-based rather than worry-based — autonomic arousal remains in an activated state, and the racing thoughts are a downstream consequence of that activation rather than the cause of it.
Nocturnal panic attacks — sudden waking with racing heart, chest tightness, and a sense of dread — are a recognized presentation during perimenopause. Declining estrogen can increase amygdala reactivity (the amygdala is the brain’s fear-processing region), while reduced GABA-A-mediated inhibition from progesterone loss lowers the arousal threshold. The combination can produce episodes that resemble panic attacks but have a hormonal rather than psychological origin. Women experiencing this for the first time in their 40s or early 50s may attribute it to an anxiety disorder when the hormonal contribution is the more relevant driver.
Whether nighttime anxiety persists after the menopause transition is variable. Some women report improvement as hormonal levels stabilize in postmenopause. Others find the pattern persists — if the HPA dysregulation and autonomic activation have run for years, the disrupted sleep-cortisol cycle can become self-sustaining even after the original hormonal trigger has stabilized.
Does Exercise or Mindfulness Lower Cortisol in Menopausal Women?
Exercise timing and intensity affect cortisol output differently. Moderate-intensity exercise earlier in the day — walking, cycling, resistance training at manageable loads — tends to reduce evening cortisol and improve sleep onset. Intense exercise in the late afternoon or evening can elevate cortisol at a time when it should be declining, which may worsen the wired-but-tired pattern in women whose HPA axis is already disinhibited.
Sahola et al. (2024) found that objective sleep architecture (measured by polysomnography, not self-report) correlated with cortisol levels in menopausal women. Women with more disrupted sleep architecture showed higher cortisol, while those with better-preserved sleep structure showed lower cortisol. This relationship may work in both directions — approaches that improve sleep structure can reduce cortisol, and approaches that reduce cortisol can improve sleep structure.
Mindfulness-based practices — meditation, breathing exercises, yoga — have shown modest cortisol-reducing effects in some populations, but the data in menopausal women is limited. The effects tend to be smaller than those of exercise and may require consistent daily practice over weeks to appear.
Adaptogens, particularly ashwagandha (Withania somnifera) and phosphatidylserine, have some evidence for cortisol modulation. Fidecicchi et al. (2024) reviewed neuroendocrine mechanisms in menopause and noted that several compounds can influence HPA axis output. However, the data in menopausal women is thin, and the magnitude of effect tends to be modest compared to the upstream hormonal driver. Ashwagandha has shown cortisol-reducing effects in trials involving stressed adults, but whether those results translate to the hormonally-driven HPA dysregulation of perimenopause is not well established.
These are modulating approaches — they can influence cortisol output and improve the daily experience of the wired-but-tired pattern. But the upstream driver is hormonal. The HPA axis disinhibition that produces the flattened cortisol curve is a consequence of declining progesterone and estrogen. For information on how hormone therapy relates to sleep during menopause, see Does HRT Help with Sleep in Menopause? and Does Progesterone Help You Sleep?.
The wired-but-tired pattern can involve overlapping causes beyond HPA dysregulation alone. Hormonal changes might compound with metabolic, inflammatory, circadian, or autonomic factors to produce the full experience. Identifying which causes might be contributing is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Does Ashwagandha Help with Menopause Sleep?
Ashwagandha’s cortisol-lowering effects have been demonstrated in trials involving chronically stressed adults — reductions in salivary cortisol ranging from 15-30% over 8-12 weeks in some studies. However, these trials were conducted in general stressed populations, not in perimenopausal or menopausal women. Whether the cortisol-reducing effect is sufficient to counteract HPA axis disinhibition driven by declining allopregnanolone is an open question.
Anecdotal reports from perimenopausal women describe a common pattern: ashwagandha provides noticeable benefit for 2-4 weeks, then the effect diminishes. This is consistent with adaptogenic compounds that modulate HPA output without addressing the underlying hormonal change — the body may adjust to the modulatory input while the upstream driver persists. Ashwagandha may be one component of a broader approach, but it does not resolve hormonally-driven cortisol dysregulation on its own (Fidecicchi et al., 2024).
Can Perimenopause Cause Panic Attacks at Night?
The distinction matters because the approach differs. Panic attacks originating from an anxiety disorder involve cognitive and behavioral patterns that respond to cognitive-behavioral strategies. Panic attacks with a hormonal substrate — driven by increased amygdala reactivity from estrogen decline and reduced GABA-A inhibition from progesterone decline — may not respond to the same strategies because the driver is neurochemical rather than cognitive.
Women who develop nocturnal panic attacks for the first time during perimenopause, with no prior history of anxiety disorders, may benefit from evaluating hormonal status rather than focusing exclusively on anxiety-based approaches. The timing — new onset in the 40s or early 50s coinciding with menstrual cycle changes — is an indicator that the hormonal pathway may be the relevant contributor (Garg & Munshi, 2025). For more on how progesterone decline affects sleep maintenance, see Does Low Progesterone Cause Insomnia in Perimenopause?.
Does the Wired-but-Tired Feeling Go Away After Menopause?
The self-sustaining cycle works like this: elevated evening cortisol disrupts sleep, disrupted sleep further elevates evening cortisol and suppresses morning cortisol, and the flattened curve persists regardless of the original trigger. Dressle et al. (2022) showed that chronic insomnia is associated with persistent HPA hyperactivation — the longer the insomnia has been present, the more entrenched the cortisol pattern becomes.
Women who address the hormonal and sleep-related contributors earlier in perimenopause may have a better chance of the pattern resolving with the menopause transition. Women who have lived with the wired-but-tired pattern for years may find that the cortisol dysregulation has become independent of the original hormonal trigger — meaning the pattern can persist into postmenopause even after hormonal levels stabilize. For more on the cortisol amplification cycle and 3am waking, see Why Do You Wake Up at 3am During Menopause?.
Related Reading
- Hormonal Sleep Disruption in Women — Full overview of hormonal pathways affecting sleep in perimenopause and menopause
- Why Do You Wake Up at 3am During Menopause? — The cortisol amplification pattern behind early-morning waking in perimenopause and menopause
- Why Doesn’t Melatonin Work for Menopause Insomnia? — The estrogen-serotonin-melatonin chain and why supplemental melatonin misses the upstream hormonal cause
- Why Can’t You Sleep Before Your Period? — How the progesterone crash in the late luteal phase fragments sleep and raises core body temperature
- Does Hormone Replacement Therapy Help with Sleep in Menopause? — What the clinical evidence shows about HRT’s effect on sleep architecture and hot-flash-driven waking
- Does Progesterone Help You Sleep? — The GABA-A receptor mechanism behind progesterone’s sleep-promoting effects and why dose timing matters
- Does Low Progesterone Cause Insomnia in Perimenopause? — Sleep maintenance and progesterone decline (publishing soon)
- Can Night Sweats Disrupt Your Deep Sleep? — Thermoregulatory disruption (publishing soon)
- Does Menopause Reduce Your Deep Sleep? — Estrogen-dependent sleep architecture changes (publishing soon)
- Does PMDD Cause Insomnia? — Severe cyclical sleep disruption (publishing soon)
- Does Menopause Cause Sleep Apnea in Women? — Underdiagnosed hormone-airway connection (publishing soon)
- Does FSH Affect Your Sleep During Menopause? — FSH as active sleep disruptor (publishing soon)
References
1. Gordon, J. L., Girdler, S. S., Meltzer-Brody, S. E., Stika, C. S., Thurston, R. C., Clark, C. T., Prairie, B. A., Moses-Kolko, E., Joffe, H., & Wisner, K. L. (2015). Ovarian hormone fluctuation, neurosteroids and HPA axis dysregulation in perimenopausal depression: a novel heuristic model. American Journal of Psychiatry, 172(3), 227-236. https://pmc.ncbi.nlm.nih.gov/articles/PMC4513660/
2. Troia, L., Garassino, M., Volpicelli, A. I., Fornara, A., Libretti, A., Surico, D., & Remorgida, V. (2025). Sleep disturbance and perimenopause: a narrative review. Journal of Clinical Medicine, 14(5), 1479. https://pmc.ncbi.nlm.nih.gov/articles/PMC11901009/
3. Cohn, A. Y., Grant, L. K., Nathan, M. D., Wiley, A., Abramson, M., Harder, J. A., Crawford, S., Klerman, E. B., Scheer, F. A. J. L., Kaiser, U. B., Rahman, S. A., & Joffe, H. (2023). Effects of sleep fragmentation and estradiol decline on cortisol in a human experimental model of menopause. Journal of Clinical Endocrinology & Metabolism, 108(11), e1347-e1357. https://pmc.ncbi.nlm.nih.gov/articles/PMC10584010/
4. Dressle, R. J., Feige, B., Spiegelhalder, K., Schmucker, C., Benz, F., Mey, N. C., & Riemann, D. (2022). HPA axis activity in patients with chronic insomnia: a systematic review and meta-analysis of case-control studies. Sleep Medicine Reviews, 62, 101588. https://pubmed.ncbi.nlm.nih.gov/35091194/
5. Garg, R., & Munshi, A. (2025). Sleep and brain function at menopause. Journal of Mid-Life Health, 15(4), 221-224. https://pmc.ncbi.nlm.nih.gov/articles/PMC11824937/
6. de Zambotti, M., Sugarbaker, D., Trinder, J., Colrain, I. M., & Baker, F. C. (2016). Acute stress alters autonomic modulation during sleep in women approaching menopause. Psychoneuroendocrinology, 66, 1-10. https://pmc.ncbi.nlm.nih.gov/articles/PMC4788552/
7. Sahola, N., Toffol, E., Kalleinen, N., & Polo-Kantola, P. (2024). Worse sleep architecture but not self-reported insomnia and sleepiness is associated with higher cortisol levels in menopausal women. Maturitas, 187, 108053. https://pubmed.ncbi.nlm.nih.gov/38909441/
8. Fidecicchi, T., Giannini, A., Chedraui, P., Luisi, S., Battipaglia, C., Genazzani, A. R., Genazzani, A. D., & Simoncini, T. (2024). Neuroendocrine mechanisms of mood disorders during menopause transition: a narrative review and future perspectives. Maturitas, 188, 108087. https://pubmed.ncbi.nlm.nih.gov/39111089/
9. Woods, N. F., Mitchell, E. S., & Smith-DiJulio, K. (2009). Cortisol levels during the menopausal transition and early postmenopause: observations from the Seattle Midlife Women’s Health Study. Menopause, 16(4), 708-718. https://pubmed.ncbi.nlm.nih.gov/19322116/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 9 references cited
