Chronic low-grade inflammation increases with age and is consistently associated with testosterone decline in men. The inflammatory cytokines that accumulate over decades — TNF-alpha, IL-6, IL-1-beta — interfere with the molecular machinery of testosterone production, and this has consequences for sleep architecture, energy, and long-term brain health.
This article covers the molecular mechanisms by which inflammatory cytokines suppress testosterone — both at the hypothalamic level (GnRH) and at the testicular level (Leydig cells) — and how sleep deprivation feeds this inflammatory cycle. For the broader landscape of how hormones disrupt sleep in men, see the full hormonal sleep disruption guide.
Inflammation-driven testosterone suppression is one of several factors that can disrupt sleep in men. Other contributors — visceral fat and aromatase activity, cortisol dysregulation, GABA deficiency, and age-related hormonal decline — may be present at the same time.
How Do Inflammatory Cytokines Suppress Testosterone Production?
The suppression happens through two molecular pathways that converge on the same outcome: less testosterone.
At the hypothalamic level, inflammatory cytokines activate NF-kB — a transcription factor — inside GnRH neurons. NF-kB activation impairs GnRH gene transcription and reduces the pulsatile GnRH release that the pituitary depends on to release luteinizing hormone (LH). Less GnRH means less LH, and less LH means less stimulation of Leydig cells in the testes. A 2024 review in Frontiers in Endocrinology mapped this cascade in detail, showing how TNF-alpha, IL-1-beta, and IL-6 all converge on NF-kB activation in the hypothalamus as part of what the authors term “inflammaging” — chronic age-related inflammation that progressively impairs the hypothalamic-pituitary-gonadal (HPG) axis (Xing et al., 2024a).
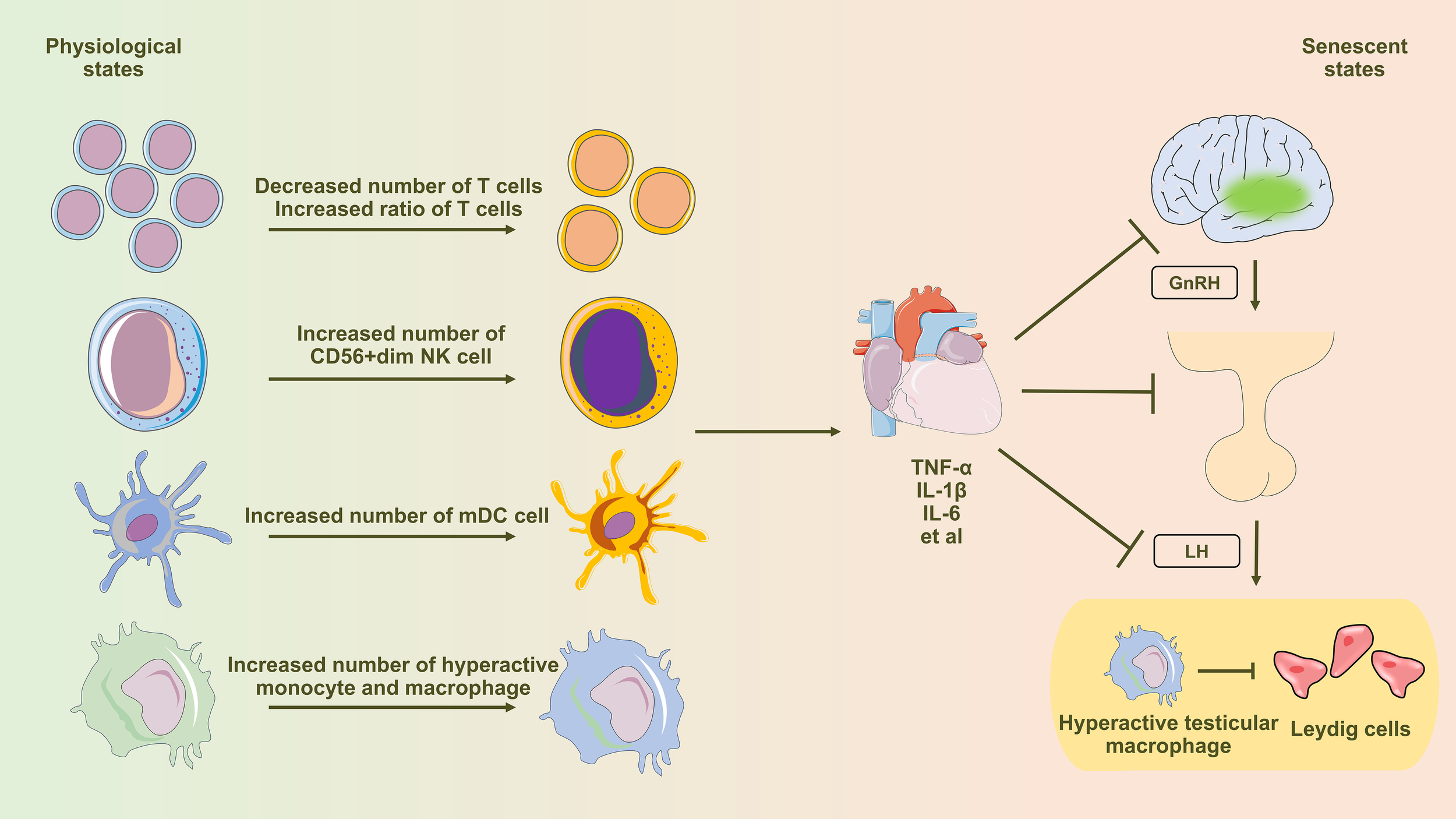
At the testicular level, TNF-alpha acts on Leydig cells through the P55 receptor. The molecular target is steroidogenic acute regulatory protein (StAR) — the protein responsible for transporting cholesterol into the mitochondria, where the steroidogenic pathway begins. StAR is the rate-limiting step of testosterone synthesis. In cultured porcine Leydig cells, TNF-alpha produced dose-dependent suppression of testosterone output, with half-maximal suppression at a concentration of 1.6 ng/ml. The inhibition was localized to cholesterol transport: when researchers bypassed StAR by providing a pre-converted cholesterol intermediate, TNF-alpha had no effect on downstream enzyme activity. The block is at StAR, not at the enzymes that convert cholesterol into testosterone (Mauduit et al., 1998).
This is a different molecular mechanism from aromatase-driven testosterone conversion, which is covered in the belly fat and testosterone article. Aromatase converts existing testosterone into estradiol. Inflammatory cytokines prevent testosterone from being produced in the first place.
Leydig cells do mount a defense against this inflammatory suppression. A 2024 study using bioinformatics analysis of aging rodent testes and TNF-alpha-exposed Leydig cell models found that a protein called TNFAIP3 (also known as A20) is upregulated in aging testes as a compensatory response to rising TNF-alpha. TNFAIP3 inhibits P38 MAPK — a downstream effector of TNF-alpha — and supports the transcription factor CEBPB, which maintains steroidogenic gene expression. When researchers knocked down TNFAIP3, TNF-alpha-induced testosterone suppression worsened and Leydig cell death increased. When they overexpressed it, both effects reversed. In aging testes, however, this defense appears to be outpaced by the rising inflammatory burden — testosterone still declines (Xing et al., 2024b).
Can Chronic Inflammation Cause Low Testosterone in Men?
The molecular mechanisms described above produce measurable testosterone deficiency in men — and the human data is consistent across age groups.
In younger men: A study of 60 men (mean age 37) divided into groups based on testosterone levels found that TNF-alpha was 61% higher in men with subnormal total testosterone compared to men with normal levels (p = 0.006). Macrophage inflammatory protein-1-alpha (MIP1-alpha) was 84% higher in the low-testosterone group (p = 0.030). In regression analysis, TNF-alpha showed an independent inverse association with both total and free testosterone after adjusting for age and BMI. These men were free of cardiovascular disease or diabetes — the inflammation-testosterone link existed before any overt metabolic condition had developed (Bobjer et al., 2013).
In older men: A 2025 study of 313 men aged 50-75 compared 146 men meeting testosterone deficiency criteria with 167 testosterone-sufficient men. Total testosterone in the deficient group was 2.66 ng/ml versus 4.91 ng/ml in the sufficient group (p < 0.001). High-sensitivity CRP was 6.04 mg/l in the testosterone-deficient group versus 4.24 mg/l in testosterone-sufficient men (p = 0.004). In the testosterone-deficient group, hsCRP correlated with waist circumference (R = 0.305), BMI (R = 0.300), and triglycerides — indicating that inflammation, adiposity, and testosterone deficiency cluster together. Even in men without testosterone deficiency, a modest negative correlation between hsCRP and testosterone persisted (R = -0.182, p = 0.018), suggesting a graded inflammatory burden on testosterone production at all androgen levels (Rotter et al., 2025).
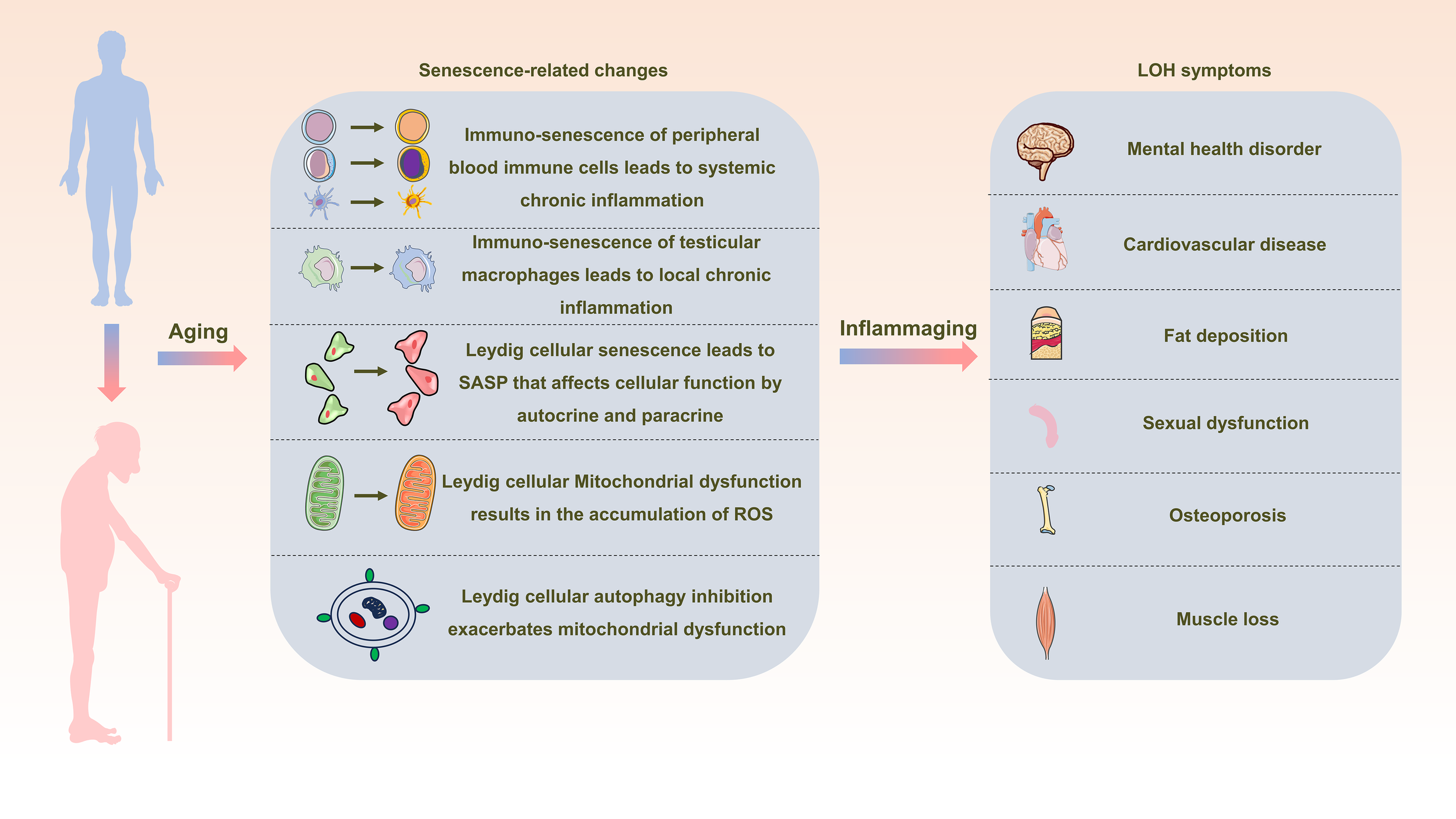
The concept underlying both findings is what researchers call “inflammaging” — the progressive accumulation of chronic low-grade inflammation with age. Senescent cells accumulate, autophagy becomes less effective, and immune regulation deteriorates. This creates a progressively more inflammatory environment for testosterone-producing cells. The Leydig cell defense (TNFAIP3) ramps up in response, but cannot keep pace with the rising cytokine load (Xing et al., 2024a).
Does Sleep Deprivation Cause Inflammation That Lowers Testosterone?
Sleep deprivation raises the same inflammatory cytokines — IL-6, TNF-alpha, CRP — that suppress testosterone through the GnRH and Leydig cell pathways described above. This creates a loop where poor sleep drives inflammation, inflammation suppresses testosterone, and low testosterone degrades sleep quality.
The landmark human study on sleep and testosterone is the 2011 Leproult and Van Cauter trial published in JAMA. Ten healthy young men (mean age 24.3) underwent one week of sleep restricted to a 5-hour window (measured sleep averaged 4 hours 48 minutes). Daytime testosterone measured across waking hours dropped from 18.4 nmol/L to 16.5 nmol/L — a 10-15% reduction. The suppression was strongest in the afternoon and evening window (2-10 PM), where testosterone fell from 17.9 to 15.5 nmol/L (p = 0.02). Self-reported vigor declined progressively across the week, tracking the testosterone drop (Leproult & Van Cauter, 2011).
One finding from this study is relevant to the inflammation hypothesis: cortisol was not elevated during the sleep restriction period. The testosterone suppression was not driven by cortisol excess — other pathways were involved. Sleep deprivation is associated with elevated IL-6, TNF-alpha, and CRP — the same cytokines that suppress GnRH pulsatility in the hypothalamus and StAR protein expression in Leydig cells. The cortisol-independent testosterone drop in the Leproult trial is consistent with an inflammation-mediated mechanism, though the study did not measure inflammatory markers directly.
At the population level, a 2024 analysis of 8,748 adults from NHANES (2011-2016) examined age- and sex-stratified relationships between sleep duration and testosterone. The relationship varied by age: in young men (20-40), short sleep was associated with elevated testosterone (possibly reflecting acute stress responses), while in middle-aged men (41-64), increased sleep duration was paradoxically associated with lower testosterone — potentially reflecting reverse causation from underlying conditions like sleep apnea. Sleep quality alone — measured by self-reported trouble sleeping — did not predict testosterone after adjusting for confounders in any age group (Hernandez-Perez et al., 2024).
The proposed self-reinforcing loop works as follows: reduced sleep duration raises IL-6 and TNF-alpha. These cytokines suppress GnRH pulsatility in the hypothalamus and StAR protein in Leydig cells — reducing testosterone output. Lower testosterone degrades sleep continuity, because testosterone helps maintain consolidated sleep architecture (covered in detail in the low testosterone and sleep article). Fragmented sleep means more time spent in lighter sleep stages, less restorative slow-wave sleep, and more inflammation. Each element of this loop has been demonstrated individually, though no single study has tracked the complete cycle.
Can Reducing Inflammation Improve Testosterone Levels?
StAR protein expression and GnRH pulsatility are not permanently damaged by inflammatory exposure — they are actively suppressed by the presence of cytokines. When the cytokine burden decreases, the suppression can reverse.
The TNFAIP3 evidence supports this in cell models. Leydig cells produce TNFAIP3 (A20) as a defense against TNF-alpha, and this defense becomes more effective when inflammatory load decreases. In the 2024 study, overexpressing TNFAIP3 reversed TNF-alpha-induced testosterone suppression and reduced Leydig cell death in culture. The molecular target — StAR protein expression — recovered when TNF-alpha activity was inhibited. This indicates that the inflammatory block on cholesterol transport into the steroidogenic pathway is reversible when the inflammatory stimulus is reduced, at least in cell models (Xing et al., 2024b).
Sleep improvement addresses the inflammation-testosterone loop at two points simultaneously. Normalizing sleep duration can reduce IL-6, TNF-alpha, and CRP — removing the inflammatory suppression of the HPG axis. At the same time, adequate sleep supports testosterone production: the Leproult trial showed that the 10-15% testosterone suppression from one week of short sleep was a function of sleep duration, not cumulative damage (Leproult & Van Cauter, 2011). Restoring sleep duration can restore both the sleep-testosterone relationship and the indirect inflammation-mediated pathway.
The graded relationship in the Rotter 2025 data is relevant here. Even in men without testosterone deficiency, a negative correlation between hsCRP and testosterone existed (R = -0.182, p = 0.018). This suggests that reducing inflammatory load at any baseline level may contribute to higher testosterone output — the benefit is not limited to men who already have low testosterone (Rotter et al., 2025).
The two accessible points for addressing this loop are sleep duration and inflammatory load. Diet composition — particularly ultra-processed food intake and its endocrine effects — and visceral fat accumulation both contribute to the inflammatory burden that suppresses testosterone.
Inflammation-driven testosterone suppression rarely stands alone. The cytokine pathways covered in this article can compound with visceral fat accumulation, cortisol dysregulation, GABA deficiency, and circadian disruption — each capable of independently fragmenting sleep. The 3AM Decoder can help identify which of these contributors might be involved.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Does Interleukin-6 Suppress Testosterone?
IL-6 is one of the three primary inflammatory cytokines — alongside TNF-alpha and IL-1-beta — that converge on NF-kB activation in the hypothalamus. Its role in testosterone suppression is mediated through the HPG axis: by reducing GnRH pulsatility, IL-6 lowers LH release, which reduces the stimulus that Leydig cells need to produce testosterone. IL-6 levels rise with both aging and sleep deprivation, making it a consistent contributor to the inflammaging-testosterone connection (Xing et al., 2024a).
How Does Tumor Necrosis Factor Alpha Affect Leydig Cells?
The Mauduit et al. (1998) study defined this mechanism using cultured porcine Leydig cells. TNF-alpha acts through the P55 receptor on Leydig cells, reducing StAR protein in both the cytosol and the mitochondrial fraction. Maximal suppression occurred at 48 hours of TNF-alpha exposure. The effect was localized to cholesterol transport — when the researchers bypassed StAR by providing a pre-converted intermediate, downstream steroidogenic enzymes (including P450scc) functioned normally. Leydig cells do produce TNFAIP3 as a defense against this suppression, but in aging testes, the rising TNF-alpha load from senescent immune cells and testicular macrophages may outpace this endogenous protection (Xing et al., 2024b).
Does Inflammation Cause Insomnia in Men?
Inflammation can degrade sleep through at least two routes. The first involves cytokine effects on sleep architecture — IL-6 and TNF-alpha increase time spent in lighter sleep stages and reduce deep slow-wave sleep. The second, which this article covers, runs through testosterone: inflammatory cytokines suppress testosterone via GnRH and StAR protein, and the resulting testosterone deficiency fragments sleep continuity. Both routes can be active simultaneously, and both are worsened by sleep deprivation itself, which raises the same inflammatory markers. For how declining testosterone fragments sleep architecture in men, see the low testosterone and sleep article (Xing et al., 2024a).
Can an Anti-Inflammatory Diet Improve Testosterone and Sleep?
The relevant framing is inflammatory load rather than any single dietary pattern. The same cytokines — TNF-alpha, IL-6, CRP — that are elevated in testosterone-deficient men (Rotter et al., 2025) are responsive to changes in diet composition, body composition, and sleep duration. Ultra-processed food intake is one contributor to this inflammatory burden, both through its inflammatory effects and through endocrine-disrupting chemicals — covered in the ultra-processed food and testosterone article. Sleep improvement is itself anti-inflammatory: normalizing sleep duration can lower IL-6 and TNF-alpha, addressing the testosterone suppression pathway at its source while also supporting testosterone production.
Related Reading
- Hormonal Sleep Disruption in Men — How testosterone, cortisol, growth hormone, and other hormones disrupt sleep in men
- Does Low Testosterone Cause Sleep Problems in Men? — How declining testosterone fragments sleep architecture in men
- Can a Cortisol Spike Wake You Up at 3am? — The cortisol-testosterone axis and nocturnal arousal patterns
- Does Growth Hormone Decline Affect Your Sleep After 40? — How age-related growth hormone loss disrupts slow-wave sleep
- Does Andropause Cause Insomnia? What Men Over 50 Need to Know — Compound hormonal aging and sleep disruption in men over 50
- Can Low GABA Cause Waking Up at 3am? — How insufficient GABA undermines sleep maintenance through the night
- What Are the Signs of Low GABA at Night? — Recognizing GABA deficiency that disrupts sleep
- Do GABA Supplements Help You Stay Asleep Through the Night? — Evaluating GABA supplementation evidence for sleep maintenance
- Does GABA Affect Testosterone and Sleep in Men? — The GABA-testosterone connection in male sleep disruption
- How Do You Increase GABA Levels Naturally for Better Sleep? — Lifestyle and dietary approaches to support GABA function
- Can Belly Fat Lower Your Testosterone and Disrupt Your Sleep? — How visceral fat suppresses testosterone through aromatase and disrupts sleep
- Can Ultra-Processed Food Lower Testosterone and Disrupt Sleep? — How endocrine-disrupting chemicals in processed food suppress testosterone and degrade sleep
- Does Testosterone Replacement Therapy Affect Sleep? — How TRT affects sleep quality, its interaction with sleep apnea, and the role of body weight
References
1. Bobjer, J., Katrinaki, M., Tsatsanis, C., Giwercman, Y. L., & Giwercman, A. (2013). Negative association between testosterone concentration and inflammatory markers in young men: A nested cross-sectional study. PLOS ONE, 8(4), e61466. https://doi.org/10.1371/journal.pone.0061466
2. Hernandez-Perez, J. G., Taha, S., Torres-Sanchez, L. E., Villasante-Tezanos, A., Milani, S. A., Baillargeon, J., Canfield, S., & Lopez, D. S. (2024). Association of sleep duration and quality with serum testosterone concentrations among men and women: NHANES 2011-2016. Andrology, 12(3), 539-549. https://pubmed.ncbi.nlm.nih.gov/37452666/
3. Leproult, R., & Van Cauter, E. (2011). Effect of 1 week of sleep restriction on testosterone levels in young healthy men. JAMA, 305(21), 2173-2174. https://pubmed.ncbi.nlm.nih.gov/21632481/
4. Mauduit, C., Gasnier, F., Rey, C., Chauvin, M. A., Stocco, D. M., Louisot, P., & Benahmed, M. (1998). Tumor necrosis factor-alpha inhibits Leydig cell steroidogenesis through a decrease in steroidogenic acute regulatory protein expression. Endocrinology, 139(6), 2863-2868. https://pubmed.ncbi.nlm.nih.gov/9607795/
5. Rotter, I., Ciosek, Z., Syroka, A., & Ryl, A. (2025). A cross-sectional study of testosterone deficiency and inflammatory markers in older men. Frontiers in Endocrinology, 16, 1606949. https://pubmed.ncbi.nlm.nih.gov/40822957/
6. Xing, D., Jin, Y., & Jin, B. (2024a). A narrative review on inflammaging and late-onset hypogonadism. Frontiers in Endocrinology, 15, 1291389. https://doi.org/10.3389/fendo.2024.1291389
7. Xing, D., Jin, Y., Shen, J., & Jin, B. (2024b). Protective effect of TNFAIP3 on testosterone production in Leydig cells under an aging inflammatory microenvironment. Archives of Gerontology and Geriatrics, 118, 105274. https://pubmed.ncbi.nlm.nih.gov/37995648/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
