Diet and sleep quality are linked — and ultra-processed food sits at the intersection of both. The first randomized controlled trial on UPF and male reproductive health, published in Cell Metabolism in 2025, demonstrated that UPF drives hormonal disruption independent of caloric intake (Preston et al., 2025). Understanding how ultra-processed food affects testosterone matters for hormonal health, brain health, and long-term sleep quality.
This article covers the endocrine-disrupting chemical pathway from UPF to testosterone suppression, the bidirectional relationship between UPF and sleep quality, and how these two pathways feed into a self-reinforcing loop. For the broader picture of how hormones disrupt sleep in men, see the full hormonal sleep disruption guide. Diet-driven testosterone suppression is one of several factors that can disrupt sleep — other contributors include visceral fat accumulation, cortisol dysregulation, inflammatory cytokines, and GABA deficiency.
Do Ultra-Processed Foods Lower Testosterone in Men?
The 2025 Preston et al. trial in Cell Metabolism used a randomized crossover design, meaning each participant served as his own control. During the ultra-processed diet phase, men experienced elevated phthalate metabolites (specifically cxMINP), decreased follicle-stimulating hormone (FSH), declining sperm motility, increased LDL:HDL cholesterol ratio, and higher body weight — all independent of caloric intake. Because calorie counts were matched between the ultra-processed and unprocessed phases, these changes point to the composition of UPF, not its energy content, as the driver of reproductive harm (Preston et al., 2025).
The phthalate-to-testosterone pathway has been quantified in earlier population data. In the 2011-2012 NHANES dataset, Meeker and Ferguson (2014) found that an interquartile range increase in DEHP metabolites (di-2-ethylhexyl phthalate, a plasticizer common in food packaging) was associated with up to a 29% reduction in serum testosterone in boys aged 6-12 years. In men aged 40-60, inverse associations between multiple phthalate metabolite classes and testosterone were also present. The mechanism: phthalates inhibit CYP17, an enzyme required for androgen synthesis, and reduce steroidogenic acute regulatory (StAR) protein in Leydig cells. StAR protein transports cholesterol into mitochondria — the rate-limiting step of testosterone production. When phthalates suppress StAR, cholesterol cannot reach the enzymes that convert it into testosterone (Meeker & Ferguson, 2014).
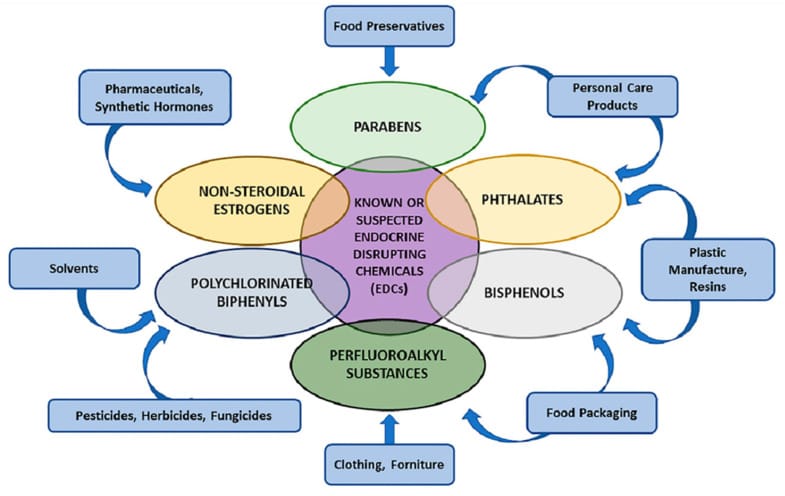
Ultra-processed food delivers more than phthalates alone. A 2025 integrative review by Fajkic et al. catalogued the full range of endocrine-disrupting chemicals in UPF: phthalates, bisphenols (from can linings and plastic containers), PFAS (from grease-resistant wrappers), and acrylamide (formed during high-heat processing). These chemicals interfere with the HPG axis — the hypothalamic-pituitary-gonadal axis that regulates testosterone — through multiple routes: receptor binding competition with natural hormones, suppression of GnRH and LH pulsatility (the brain-level hormones that instruct Leydig cells to produce testosterone), Leydig cell death, and interference with cytochrome P450 enzymes involved in steroidogenesis. UPF accounts for 25-67.8% of daily caloric intake in industrialized populations, making it a primary and ongoing source of EDC exposure (Fajkic et al., 2025).
A separate NHANES analysis of 3,354 adolescents found that UPF intake was marginally inversely associated with sex hormone-binding globulin (SHBG), the protein that transports testosterone in the blood. This suggests that UPF may also affect testosterone bioavailability — how much testosterone is free and active — in addition to total production (Zhao et al., 2024).
Does Eating Ultra-Processed Food Affect Sleep Quality?
Delpino et al. (2023) conducted a meta-analysis synthesizing 15 cross-sectional studies on UPF consumption and sleep. After adjusting for confounders, the association between high UPF intake and poor sleep outcomes remained present across all age groups studied. The proposed mechanisms include three pathways: blood glucose dysregulation (UPF-driven glucose spikes and crashes can impair melatonin release), gut microbiota disruption from food additives (altering the gut-brain-sleep axis), and endocrine-disrupting chemicals from UPF packaging interfering with circadian hormonal patterns (Delpino et al., 2023).
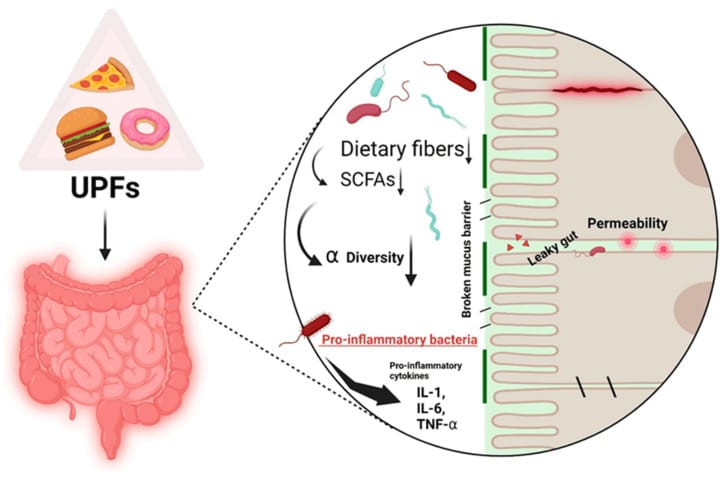
The relationship runs in both directions. Andreeva et al. (2023) conducted a separate review of 15 adult cohort studies and found that both studies modeling sleep as the exposure variable — testing whether poor sleep predicts higher UPF intake — demonstrated that it does. Sleep restriction activates appetite-related pathways: elevated ghrelin (which increases hunger), suppressed leptin (which decreases satiety), and suppressed peptide YY (which reduces fullness after eating). These hormonal changes increase cravings for high-fat, high-sugar ultra-processed foods, not for whole foods (Andreeva et al., 2023).
This creates a feedback loop: UPF consumption is associated with degraded sleep, and degraded sleep increases UPF cravings through appetite hormone dysregulation. A man eating a UPF-heavy diet may find that his sleep worsens, and as his sleep worsens, his cravings for more UPF increase — making dietary change more difficult without also addressing sleep quality. The bidirectional nature of this relationship is one reason why diet and sleep need to be considered together, not in isolation.
How Does the Ultra-Processed Food and Sleep Combination Suppress Testosterone?
The first pathway is chemical. Phthalates and bisphenols from UPF packaging enter the bloodstream and suppress StAR protein and CYP17 enzyme activity in Leydig cells, reducing testosterone production regardless of sleep quality. The 2025 RCT demonstrated this: even with matched caloric intake, men on the ultra-processed diet had elevated phthalate metabolites and reduced FSH (Preston et al., 2025).
The second pathway runs through sleep. UPF consumption degrades sleep quality through glucose dysregulation, gut disruption, and circadian interference (as covered in the meta-analyses above). Poor sleep then independently suppresses nocturnal GnRH pulsatility — the rhythmic release of gonadotropin-releasing hormone from the hypothalamus that drives LH release and, downstream, Leydig cell testosterone production. NHANES data from 8,748 adults supports this connection: in middle-aged men (41-64 years), sleep duration is associated with testosterone levels. Men in this age group sleeping nine or more hours had 2.03-fold elevated odds of low testosterone (OR = 2.03; 95% CI: 1.10-3.73), though this likely reflects that excessive sleep need may itself be a consequence of testosterone deficiency rather than a cause (Hernandez-Perez et al., 2024).
These two pathways are independent of each other. A man eating a UPF-heavy diet faces both chemical suppression of testosterone and sleep-mediated suppression of the GnRH-LH cascade. Removing one pathway still leaves the other active. Switching to unprocessed food would reduce EDC exposure (partially addressing the chemical pathway) but would not automatically restore sleep architecture that has already been disrupted. Conversely, improving sleep quality would support nocturnal GnRH pulsatility but would not reduce the phthalate and bisphenol load from a diet still dominated by UPF.
The 2025 RCT illustrates one side of this: when men ate an unprocessed diet (same calorie count), their hormonal markers improved during that phase, demonstrating that the chemical pathway responds to dietary composition changes (Preston et al., 2025). But the sleep pathway requires its own attention — and because the UPF-sleep relationship is bidirectional, addressing it may require changes to both diet and sleep simultaneously.
Diet-driven testosterone suppression rarely acts alone. The endocrine disruption and sleep degradation covered in this article often co-occur with visceral fat accumulation, cortisol dysregulation, and inflammatory cytokines — each capable of independently suppressing testosterone and fragmenting sleep. Identifying which of these causes might be involved is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Do Processed Foods Contain Endocrine Disruptors?
Phthalates leach from plastic packaging and flexible tubing used in food manufacturing. Bisphenol A (BPA) and its substitutes migrate from epoxy-lined cans and polycarbonate containers. PFAS (per- and polyfluoroalkyl substances) come from grease-resistant food wrappers and microwave popcorn bags. Acrylamide forms when starchy foods are heated above 120 degrees Celsius during industrial frying, baking, or extrusion.
The more processing steps a food undergoes — and the more contact it has with packaging materials — the higher the EDC concentration in the final product. A 2025 review catalogued the full range of these chemicals and noted that UPF introduces a mix of endocrine-disrupting compounds that interfere with HPG axis function through multiple concurrent pathways (Fajkic et al., 2025).
Does Sugar Before Bed Affect Sleep Quality?
This blood sugar mechanism is one component of how ultra-processed food affects sleep. UPF delivers both sugar and endocrine-disrupting chemicals, meaning its sleep-disrupting effects are not limited to glucose volatility alone. The Delpino et al. (2023) meta-analysis identified blood glucose dysregulation as one of several pathways through which UPF consumption is associated with adverse sleep outcomes. For individuals whose sleep is disrupted by overnight cortisol spikes, blood sugar-driven cortisol release may be a contributing factor alongside UPF’s hormonal and gut-related effects.
What Should Men Eat to Improve Sleep Quality?
The 2025 RCT demonstrated that when men ate unprocessed food at the same calorie level, their hormonal markers moved in a favorable direction compared to the UPF phase — lower phthalate metabolites, more stable FSH (Preston et al., 2025). This supports a reduction in UPF as directionally beneficial rather than prescribing a particular diet plan.
Because the UPF-sleep relationship is bidirectional, improving diet may also improve sleep, which in turn can reduce UPF cravings — creating a positive feedback loop that works in the opposite direction from the one described above. Andreeva et al. (2023) noted that appetite hormone changes from better sleep (lower ghrelin, higher leptin and peptide YY) reduce the drive toward high-fat, high-sugar processed foods. Starting with dietary changes may make subsequent sleep improvements easier to sustain.
Does Protein Intake Affect Sleep Quality in Men?
Whole-food protein sources — fish, poultry, eggs, legumes — deliver tryptophan without the EDC load that comes with heavily processed protein products (protein bars in plastic packaging, processed deli meats, shelf-stable protein shakes). In the context of UPF and sleep, choosing minimally processed protein sources serves two functions: providing the amino acid precursors that support melatonin production and reducing exposure to the endocrine-disrupting chemicals that interfere with both hormonal health and sleep quality.
Related Reading
- Hormonal Sleep Disruption in Men — How testosterone, cortisol, growth hormone, and other hormones disrupt sleep in men
- Does Low Testosterone Cause Sleep Problems in Men? — How declining testosterone fragments sleep architecture in men
- Can a Cortisol Spike Wake You Up at 3am? — The cortisol-testosterone axis and nocturnal arousal patterns
- Does Growth Hormone Decline Affect Your Sleep After 40? — How age-related growth hormone loss disrupts slow-wave sleep
- Does Andropause Cause Insomnia? What Men Over 50 Need to Know — Hormonal aging and sleep disruption in men over 50
- Can Belly Fat Lower Your Testosterone and Disrupt Your Sleep? — How visceral fat suppresses testosterone through aromatase and disrupts sleep
- Can Inflammation Suppress Testosterone and Disrupt Your Sleep? — How inflammatory cytokines suppress testosterone production at the brain and testicular level
- Can Low GABA Cause Waking Up at 3am? — How insufficient GABA leaves sleep unprotected
- What Are the Signs of Low GABA at Night? — Recognizing GABA deficiency that disrupts sleep
- Do GABA Supplements Help You Stay Asleep Through the Night? — Evaluating GABA supplementation evidence for sleep maintenance
- Does GABA Affect Testosterone and Sleep in Men? — The GABA-testosterone connection in male sleep disruption
- How Do You Increase GABA Levels Naturally for Better Sleep? — Lifestyle and dietary approaches to support GABA function
- Does Testosterone Replacement Therapy Affect Sleep? — How TRT affects sleep quality, its interaction with sleep apnea, and the role of body weight
References
1. Preston, J. M., et al. (2025). Effect of ultra-processed food consumption on male reproductive and metabolic health: A randomized controlled crossover trial. Cell Metabolism. https://pubmed.ncbi.nlm.nih.gov/40882621/
2. Meeker, J. D., & Ferguson, K. K. (2014). Urinary phthalate metabolites are associated with decreased serum testosterone in men, women, and children from NHANES 2011-2012. Journal of Clinical Endocrinology & Metabolism, 99(11), 4346-4352. https://pubmed.ncbi.nlm.nih.gov/25121464/
3. Fajkic, A., et al. (2025). Ultra-Processed Diets and Endocrine Disruption: Explanation of Missing Link in Rising Cancer Incidence Among Young Adults. Cancers, 17(13), 2196. https://pubmed.ncbi.nlm.nih.gov/40647494/
4. Zhao, H., et al. (2024). Ultra-processed foods intake and sex hormone levels among children and adolescents aged 6-19 years: A cross-sectional study. Frontiers in Nutrition, 11, 1451481. https://pubmed.ncbi.nlm.nih.gov/39309139/
5. Delpino, F. M., et al. (2023). Intake of ultra-processed foods and sleep-related outcomes: A systematic review and meta-analysis. Nutrition, 106, 111908. https://pubmed.ncbi.nlm.nih.gov/36470114/
6. Andreeva, V. A., Perez-Jimenez, J., & St-Onge, M. P. (2023). A Systematic Review of the Bidirectional Association Between Consumption of Ultra-processed Food and Sleep Parameters Among Adults. Current Obesity Reports, 12(4), 457-467. https://pubmed.ncbi.nlm.nih.gov/37477854/
7. Hernandez-Perez, J. G., et al. (2024). Association of sleep duration and quality with serum testosterone concentrations among men and women: NHANES 2011-2016. Andrology, 12(3), 598-609. https://pubmed.ncbi.nlm.nih.gov/37452666/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
