Wearable devices now show men their deep sleep percentages nightly, and many notice the decline after 40. The growth hormone connection explains why that decline matters beyond “feeling rested” — growth hormone drives tissue repair, body composition, and metabolic function, and its secretion depends on deep sleep.
This article covers how growth hormone secretion is linked to slow-wave sleep, what happens to both as men age, the evidence for whether the relationship is causal, and what the research shows about raising growth hormone through sleep. It does not cover testosterone-sleep interactions (Does Low Testosterone Cause Sleep Problems in Men?) or GABA’s role in sleep maintenance (covered in a separate article). Growth hormone decline during sleep is one of several hormonal mechanisms that affect men after 40; the full picture is in Hormonal Sleep Disruption in Men.
When Is Growth Hormone Released During Sleep?
The coupling between delta wave EEG activity and growth hormone secretion is well-documented at the level of individual sleep cycles. In a crossover study of eight healthy men, Gronfier et al. (1996) used ritanserin (a serotonin receptor antagonist that enhances slow-wave sleep) to increase delta power by 24% — and growth hormone secretory rates rose 29% simultaneously. The within-subject correlation between delta power and growth hormone was r = 0.803 during pulsatile peaks (p < 0.0001). EEG delta power tracks the hypothalamic state that drives growth hormone release on a moment-to-moment basis.
The neural circuit underlying this coupling was mapped in 2025. Ding et al. (2025), published in Cell, identified the hypothalamic GHRH/somatostatin (SST) circuit responsible for sleep-state-dependent growth hormone secretion. During NREM sleep, GHRH neuron activity increases while SST neuron activity decreases — creating a permissive window for growth hormone release. The study also found an inherent negative feedback loop: growth hormone itself activates locus coeruleus noradrenergic neurons, promoting wakefulness and self-limiting further secretion within a sleep cycle.
The GHRH/SST permissive window explains why fragmented sleep reduces growth hormone output. Each time a NREM episode is cut short, the permissive window closes before the full growth hormone pulse can complete. Van Cauter et al. (2000) documented that the first slow-wave sleep episode produces the dominant growth hormone pulse of the night; when sleep architecture is fragmented, only smaller, distributed pulses occur across the remaining sleep period.
Does Growth Hormone Decline With Age?
The Van Cauter et al. (2000) study in JAMA remains the largest dataset quantifying the parallel decline of slow-wave sleep and growth hormone across the male lifespan. The trajectory is front-loaded: the steepest decline in both slow-wave sleep and growth hormone happens between young adulthood and midlife (ages 16–43), representing approximately a 75% reduction in growth hormone output. After age 50, the rate of decline slows to −43 micrograms per decade — there is less left to lose.
The medical term for this age-related growth hormone decline is somatopause. It co-occurs with andropause (testosterone decline) and adrenopause (DHEA decline) — a convergence of hormonal reductions that compound each other’s effects on sleep, body composition, and recovery.
A measurement distinction matters here. Ishii et al. (2024) reviewed the difference between delta power (1–4 Hz), which decreases with aging, and slow-oscillatory power (< 1 Hz), which may increase with age. This resolves an apparent contradiction in the literature: some studies report "preserved slow waves" in older adults while growth hormone continues to decline. The frequency band matters — it is the 1–4 Hz delta activity, not the slower oscillations, that is coupled to growth hormone secretion.
The relationship also runs in the other direction. Prinz et al. (1995) measured IGF-1 (growth hormone’s downstream marker) in 30 healthy older men and found it explained 28% of the residual variance in delta sleep after removing age-related effects (semi-partial r = 0.53, p = 0.003). Higher IGF-1 predicted more delta sleep, supporting a bidirectional relationship between the somatotropic axis and slow-wave sleep capacity.
Can Poor Sleep Lower Growth Hormone Levels?
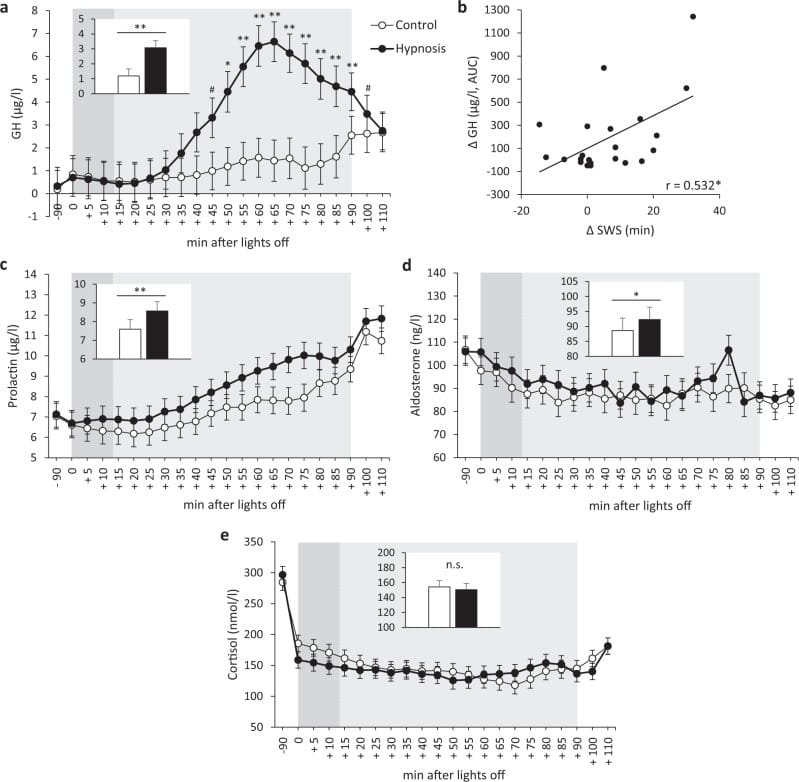
The causal evidence comes from Besedovsky et al. (2022), who used hypnotic suggestion to enhance slow-wave sleep during afternoon naps in 23 healthy young men. Slow-wave sleep duration increased by 49%, and growth hormone levels rose to more than 400% of control values at 75 minutes after sleep onset. The correlation between individual slow-wave sleep gains and growth hormone elevation was r = 0.532 (p = 0.013). Growth hormone concentrations achieved under enhanced slow-wave sleep were comparable to peak levels normally observed only during nighttime sleep — demonstrating that the slow-wave sleep state itself, not circadian time, drives the growth hormone pulse.
Slow-wave sleep suppression has practical implications for men over 40. Several common factors reduce slow-wave sleep and, by extension, growth hormone output:
- Alcohol suppresses slow-wave sleep in the second half of the night, reducing the available window for growth hormone secretion
- Untreated sleep apnea fragments sleep architecture, repeatedly interrupting NREM episodes before the permissive window for growth hormone can open
- Chronic stress elevates cortisol, which interferes with delta wave generation — the same delta activity that drives growth hormone release
- Aging itself reduces slow-wave sleep, but the other factors on this list accelerate the decline
The Ding et al. (2025) circuit work explains why: anything that shortens NREM episodes or fragments them closes the hypothalamic permissive window before the growth hormone pulse can complete. And because slow-wave sleep suppression also reduces testosterone (a relationship documented separately in the testosterone-sleep literature), losing deep sleep reduces both growth hormone and testosterone simultaneously.
Does Growth Hormone Therapy Improve Sleep in Older Men?
Fernández-Garza et al. (2025) reviewed the evidence for GHRH and growth hormone approaches in aging. Pulsatile GHRH administration in healthy older adults reduced nocturnal awakenings and extended the first NREM sleep period. Intranasal GHRH increased both REM sleep and slow-wave sleep. GHRH stimulates the natural secretion pathway, which aligns with the circuit architecture Ding et al. (2025) described.
Exogenous growth hormone therapy tells a different story. In men aged 70–85, reviewed trials showed a 4.3% increase in lean body mass and a 13.1% decrease in fat mass over 6 months. One 6-year longitudinal study reported a 15.9% improvement in lumbar spine bone mineral density. But adverse effects were common: fluid retention in 11–100% of participants and carpal tunnel in 7–50%. Current guidelines restrict growth hormone therapy to pathological deficiency — not somatopause.
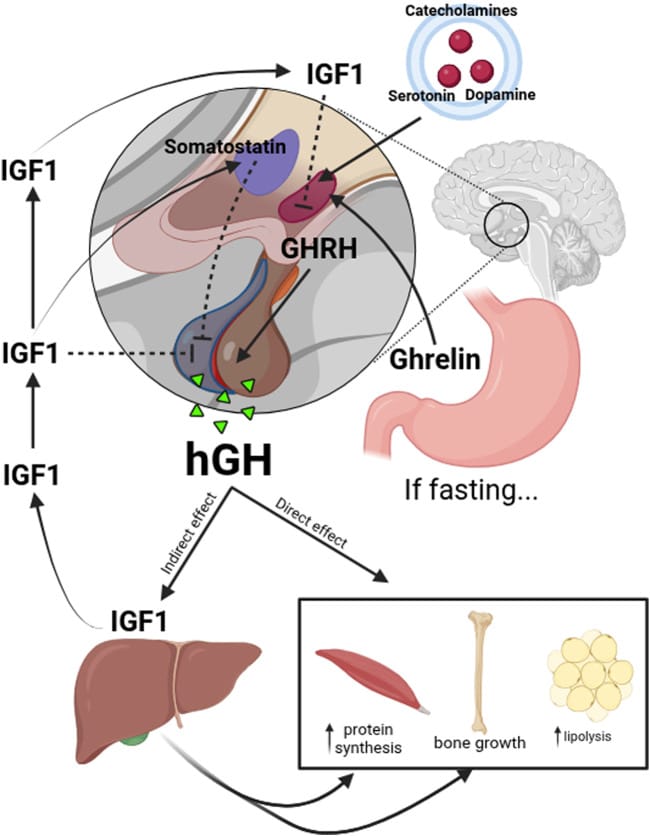
The more evidence-based question for men experiencing age-related growth hormone decline is: what restores slow-wave sleep? Because slow-wave sleep drives growth hormone through the natural GHRH pathway, addressing the causes of slow-wave sleep loss — cortisol elevation, sleep architecture fragmentation, hormonal decline, inflammation — raises growth hormone output without exogenous therapy. The Besedovsky et al. (2022) result is proof of concept: enhance slow-wave sleep by 49%, and growth hormone rises by more than 400%.
The causes of slow-wave sleep loss overlap with the other hormonal mechanisms covered in this cluster — cortisol elevation, testosterone decline, and GABA all affect the same deep sleep architecture that growth hormone depends on.
Growth hormone decline and slow-wave sleep loss rarely happen in isolation. Testosterone decline reduces slow-wave sleep independently, cortisol elevation fragments sleep architecture, and impaired GABA receptor function weakens the tonic inhibition that sustains deep sleep. These mechanisms might all be contributing to the pattern of declining deep sleep after 40.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
What Are the Signs of Low Growth Hormone During Sleep?
Because growth hormone secretion peaks during slow-wave sleep, a declining deep sleep percentage on a wearable tracker can be an indirect indicator — though wearables measure movement-based estimates of sleep staging, not EEG delta power. The body composition changes associated with somatopause — increased abdominal fat, reduced lean mass, slower post-exercise recovery — develop gradually and overlap with testosterone decline and other age-related changes, making it difficult to attribute them to growth hormone alone (Fernández-Garza et al., 2025).
How Can You Increase Growth Hormone While Sleeping?
Increasing growth hormone during sleep is a sleep architecture question. The Besedovsky et al. (2022) data showed that increasing slow-wave sleep duration by 49% produced a greater-than-400% growth hormone response. The steps that matter are the ones that protect or restore slow-wave sleep: reducing evening cortisol elevation, addressing sleep fragmentation from apnea or environmental disruption, maintaining hormonal health (both testosterone and GABA function affect slow-wave sleep), and regular exercise — particularly resistance training and high-intensity interval training, which are associated with increased slow-wave sleep in subsequent nights.
Does Growth Hormone Deficiency Cause Sleep Problems?
Adults with growth hormone deficiency report poorer subjective sleep quality in studies reviewed by Fernández-Garza et al. (2025). However, the effect is difficult to isolate from the other hormonal changes that co-occur with aging — testosterone, DHEA, and cortisol rhythms all change during the same period, and each affects sleep architecture independently. The Prinz et al. (1995) finding that IGF-1 predicts residual delta sleep capacity in older men supports the idea that maintaining somatotropic axis function helps preserve deep sleep — but it does not establish that growth hormone deficiency is the primary driver of age-related sleep decline.
Related Reading
- Hormonal Sleep Disruption in Men — How testosterone, cortisol, growth hormone, and GABA interact to fragment sleep in men
- Can Low GABA Cause Waking Up at 3am? — The GABAergic mechanism behind 3am waking
- What Are the Signs of Low GABA at Night? — How to recognize impaired GABAergic function
- Do GABA Supplements Help You Stay Asleep Through the Night? — Trial evidence for oral GABA and the blood-brain barrier question
- Does GABA Affect Testosterone and Sleep in Men? — How testosterone-derived neurosteroids modulate GABA-A receptors
- How Do You Increase GABA Levels Naturally for Better Sleep? — Exercise, yoga, fermented foods, and gut health for GABA support
- Does Low Testosterone Cause Sleep Problems in Men? — The bidirectional testosterone-sleep feedback loop
- Can a Cortisol Spike Wake You Up at 3am? — Cortisol-testosterone axis and nocturnal waking
- Does Andropause Cause Insomnia? What Men Over 50 Need to Know — Compound hormonal decline in aging men
- Can Belly Fat Lower Your Testosterone and Disrupt Your Sleep? — How visceral fat suppresses testosterone through aromatase and disrupts sleep
- Can Ultra-Processed Food Lower Testosterone and Disrupt Sleep? — How endocrine-disrupting chemicals in processed food suppress testosterone and degrade sleep
- Can Inflammation Suppress Testosterone and Disrupt Your Sleep? — How inflammatory cytokines suppress testosterone production at the brain and testicular level
- Does Testosterone Replacement Therapy Affect Sleep? — How TRT affects sleep quality, its interaction with sleep apnea, and the role of body weight
References
Besedovsky, L., Cordi, M. J., Wisst, B., Müller, J., Brehm, L., & Born, J. (2022). Hypnotic enhancement of slow-wave sleep increases sleep-associated hormone secretion and reduces sympathetic predominance in healthy humans. Communications Biology, 5, 747. https://pubmed.ncbi.nlm.nih.gov/35882899/
Ding, X., et al. (2025). Neuroendocrine circuit for sleep-dependent growth hormone release. Cell, 188(3), 567–582. https://pubmed.ncbi.nlm.nih.gov/40562026/
Fernández-Garza, L. E., et al. (2025). Growth hormone and aging: a clinical review. Frontiers in Aging, 6. https://pubmed.ncbi.nlm.nih.gov/40260058/
Gronfier, C., et al. (1996). A quantitative evaluation of the relationships between growth hormone secretion and delta wave electroencephalographic activity during normal sleep and after enrichment in delta waves. Sleep, 19(10), 817–824. https://pubmed.ncbi.nlm.nih.gov/9085491/
Ishii, T., et al. (2024). From macro to micro: slow-wave sleep and its pivotal health implications. Frontiers in Sleep, 3. https://pubmed.ncbi.nlm.nih.gov/41424515/
Prinz, P. N., et al. (1995). Higher plasma IGF-1 levels are associated with increased delta sleep in healthy older men. Journal of Gerontology: Biological Sciences, 50A(4), M222–M226. https://pubmed.ncbi.nlm.nih.gov/7614245/
Van Cauter, E., Leproult, R., & Plat, L. (2000). Age-related changes in slow wave sleep and REM sleep and relationship with growth hormone and cortisol levels in healthy men. JAMA, 284(7), 861–868. https://pubmed.ncbi.nlm.nih.gov/10938176/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
