The 3am waking pattern is one of the more common sleep complaints, and cortisol is frequently cited as the cause. The relationship is supported by evidence, but more nuanced than “cortisol wakes you up.” Cortisol’s circadian rise begins around 2-3am, and that rise interacts with testosterone, GABA function, and sleep architecture in ways that determine whether the rise stays below the arousal threshold or becomes a full awakening.
This article covers how cortisol behaves during sleep, what happens when cortisol and testosterone interact at night, what the cortisol awakening response means, and the pattern of cortisol-driven sleep disruption in men. It does not cover GABA-cortisol interplay (covered in the GABA mechanism article) or blood sugar-cortisol interactions (covered in the metabolic cluster). Cortisol-testosterone disruption is one of several hormonal mechanisms that can fragment sleep in men; the full picture is in Hormonal Sleep Disruption in Men.
What Causes Cortisol to Spike in the Middle of the Night?
Under normal conditions, cortisol reaches its lowest point (nadir) around midnight, then begins a steep circadian rise between 2am and 4am. This rise continues through the early morning hours and peaks approximately 30 minutes after waking (Balbo et al., 2010).
The critical finding from decades of HPA axis research is that sleep onset inhibits cortisol secretion, while every awakening stimulates it. This means each time you wake during the night, even briefly, there is a discrete cortisol cost — the HPA axis activates in response to the arousal itself (Balbo et al., 2010). Waking at 3am doesn’t just coincide with rising cortisol; it adds to it.
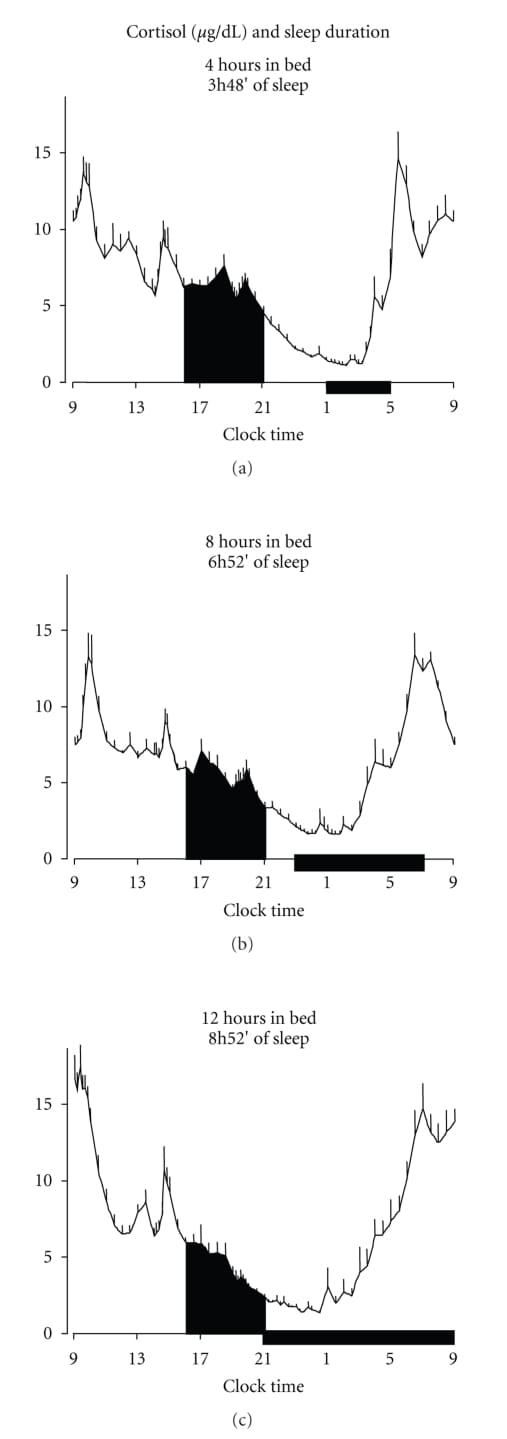
The carry-forward effect is measurable. In a controlled within-subject study, young men underwent continuous cortisol monitoring across normal 8-hour sleep, partial sleep deprivation (~4 hours), and total sleep deprivation. Partial deprivation produced a 37% elevation in evening cortisol the following day (p = 0.03), and total deprivation produced a 45% elevation (p = 0.003). The normal late-afternoon cortisol nadir was abolished, and the onset of the nocturnal cortisol low-activity period was delayed by at least one hour (Leproult et al., 1997).
Waking at 3am raises cortisol. Elevated cortisol at bedtime the next evening delays the low-activity window. That delayed window makes the following night’s sleep more fragmented — a self-perpetuating feedback loop.
Sleep fragmentation appears to reshape the entire cortisol curve. In a crossover trial with healthy premenopausal women (the HPA fragmentation response is mechanistically shared across sexes), three nights of experimentally induced sleep fragmentation raised bedtime cortisol by 27% (P = .03) while simultaneously collapsing the cortisol awakening response by 57% (P = .01). Time spent awake after sleep onset was positively associated with bedtime cortisol (P = .047) — each additional minute of nocturnal wakefulness both increased nighttime HPA drive and reduced the morning cortisol awakening response (Cohn et al., 2023).
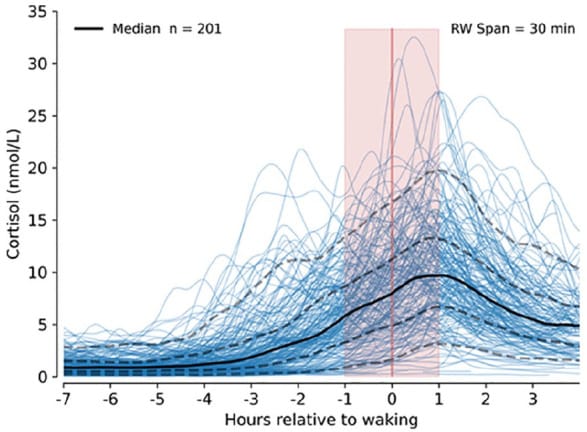
A 2025 microdialysis study of 201 healthy volunteers found that the rate of cortisol increase did not change when participants woke compared to the preceding hour while asleep — suggesting the post-waking cortisol rise may be a continuation of the circadian rhythm rather than a response to the act of waking itself (Velazquez Sanchez & Dalley, 2025). For men waking at 3am, the cortisol rise is likely circadian — already underway before they became conscious.
Does High Cortisol Lower Testosterone in Men?
The hypothalamic-pituitary-adrenal (HPA) axis and the hypothalamic-pituitary-gonadal (HPG) axis share the hypothalamus as their regulatory origin. When the HPA axis is activated — by stress, sleep loss, or sleep fragmentation — it suppresses GnRH pulsatility, which reduces LH, which reduces testosterone production in the testes. This is a direct suppressive pathway, documented across both epidemiological and interventional data (Liu & Reddy, 2022).
Sleep loss and reduced sleep duration are consistently associated with lower morning, afternoon, and 24-hour testosterone, alongside higher afternoon cortisol. Testosterone’s circadian peak occurs during sleep and requires at least three hours of uninterrupted sleep with normal architecture — making nocturnal fragmentation particularly damaging to the testosterone/cortisol ratio (Liu & Reddy, 2022).
In 178 Japanese male workers (mean age 49.1 years), both testosterone and cortisol independently showed negative associations with time in bed. Testosterone showed a positive association with sleep efficiency (standardized beta = 0.15, p < 0.05) — men with higher testosterone achieved more restorative sleep per hour in bed. But a testosterone-by-cortisol interaction effect on sleep efficiency was identified (beta = -0.22, p = 0.012): at low cortisol concentrations, testosterone was negatively correlated with time in bed (r = -0.23, p < 0.05), but this protective association was abolished at high cortisol concentrations (beta = 0.10, p = 0.42). Elevated cortisol nullified testosterone’s protective effect on sleep (Hirokawa et al., 2022).
A 2024 study of 225 men characterized a testosterone awakening response — a morning testosterone rise measured alongside the cortisol awakening response. Two phenotypes emerged: 69% of men showed a typical cortisol awakening response, while 31% showed a blunted or absent one. Only in men with a typical cortisol response did testosterone show age-dependent associations with late-onset hypogonadism scores. Men with a blunted cortisol response showed no testosterone-age or testosterone-complaint associations, suggesting that the HPA and HPG axes are co-regulated during morning awakening, with cortisol gating the testosterone response (Park et al., 2024).
What Are the Signs of High Cortisol and Low Testosterone in Men?
The distinguishing characteristic of cortisol-driven 3am waking is the quality of the arousal. Cortisol-mediated waking often includes physical activation — elevated heart rate, increased body temperature, a sense of being alert or “wired” — because cortisol drives sympathetic nervous activation. This differs from GABA-related waking, which tends to be more cognitive: racing thoughts, an inability to stop the mind from running, a sense of mental activation without the same degree of physical arousal.
The Japanese workers study provides quantitative context: men with elevated cortisol and lower testosterone had the worst sleep efficiency scores. The testosterone-cortisol ratio, not either hormone in isolation, predicted sleep quality outcomes (Hirokawa et al., 2022).
A U.S. claims database analysis of sleep disorders and testosterone deficiency in men aged 40-70 found that insomnia was associated with testosterone deficiency with an odds ratio of 1.74. Circadian rhythm disorders had an even higher odds ratio of 2.63. The association held after controlling for age and comorbidities, meaning the sleep problem itself is a marker of hormonal disruption — not just a consequence of it (Agrawal et al., 2024).
Beyond sleep, the cortisol-testosterone imbalance manifests as: central adiposity that does not respond to caloric restriction (cortisol promotes visceral fat deposition), slower exercise recovery (the catabolic-to-anabolic ratio favors catabolism), reduced libido, and a pattern of daytime fatigue paired with nighttime alertness (cortisol’s circadian curve is flattened — not low enough at night, not high enough in the morning).
Chronic stress is the most common upstream driver of cortisol-testosterone imbalance. Sustained stress maintains HPA activation, which maintains cortisol elevation, which maintains testosterone suppression — a self-reinforcing loop that worsens with each night of disrupted sleep.
Why Do Men Wake Up at 3am?
The 3am waking pattern has a physiological basis. It corresponds to a period when multiple sleep-maintenance mechanisms are at their weakest — and when cortisol’s circadian rise creates the first sustained arousal pressure since sleep onset.
By 2-3am, the first and typically largest cycles of slow-wave sleep have been completed. The remaining sleep is lighter, more dominated by REM, and more susceptible to disruption. At the same time, melatonin — which peaked in the early night — has begun its decline. And cortisol, which reached its nadir around midnight, is now accelerating upward toward its morning peak.
Under this converging pressure, any additional arousal stimulus — whether from cortisol, blood sugar regulation, GABA insufficiency, or bladder activation — is more likely to produce a full awakening than it would at 11pm or 1am, when deep sleep and melatonin are at their strongest.
Each awakening at 3am triggers additional cortisol release (Balbo et al., 2010). That cortisol suppresses testosterone. Lower testosterone reduces neurosteroid-mediated sleep maintenance. The next night, the same 3am window arrives — but with higher baseline cortisol from the previous night’s fragmentation and lower testosterone from the suppressive effect.
Multiple causes can converge on this window. Cortisol-testosterone imbalance is one mechanism, but GABA insufficiency (Can Low GABA Cause Waking Up at 3am?), blood sugar instability, and circadian misalignment also target the 2-4am period. In the U.S. claims database analysis, circadian rhythm disorders had the highest odds ratio (2.63) for testosterone deficiency — even higher than insomnia at 1.74 (Agrawal et al., 2024). The 3am waking may reflect cortisol alone, but it may also reflect several of these mechanisms compounding.
Cortisol-driven 3am waking often overlaps with other mechanisms. Impaired GABA function, testosterone decline, metabolic instability, and circadian disruption might all be contributing to the same pattern. Men experiencing regular 3am waking often have more than one of these at work.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Is Waking Up at 3am a Sign of High Cortisol?
Cortisol’s circadian rise begins around 2-3am in everyone — waking during this window may reflect cortisol, but it may also reflect declining melatonin, GABA insufficiency, or a blood sugar drop that triggers a counter-regulatory cortisol response. What distinguishes cortisol-driven waking is physical activation (heart rate, body temperature, alertness) rather than purely mental restlessness. If the pattern includes daytime fatigue, central weight gain, and reduced recovery alongside the 3am waking, cortisol-testosterone imbalance becomes a more likely contributor.
How Do You Reduce Cortisol-Driven 3am Waking?
Melatonin supplementation targets sleep onset, not maintenance — it does not change the cortisol curve. Reducing the cortisol load requires addressing the upstream drivers: chronic stress (the primary HPA activator), sleep debt itself (since short sleep raises the following evening’s cortisol by 37-45%, per Leproult et al., 1997), and the testosterone-cortisol ratio.
What Happens When Cortisol Is High and Testosterone Is Low?
The HPA axis suppresses the HPG axis when activated, meaning cortisol elevation directly reduces testosterone output. Lower testosterone reduces sleep quality, which fragments sleep further. Fragmented sleep raises cortisol (27% bedtime increase after three nights of fragmentation, per Cohn et al., 2023), completing a feedback loop. In the randomized hormone-clamp experiment, simultaneously normalizing both hormones during sleep restriction reversed approximately half of the associated insulin resistance — evidence that the imbalance mediates measurable metabolic consequences (Liu & Reddy, 2022).
Is Waking Up at 3am a Sign of Anxiety?
The HPA axis does not distinguish between psychological stress and physiological stress. Anxiety activates the same cortisol pathway as sleep deprivation, chronic illness, or caloric deficit (Balbo et al., 2010). If anxiety is the upstream driver, cortisol elevation is downstream. If sleep fragmentation or circadian misalignment is the driver, the cortisol pattern may be present without any anxious experience. Both pathways produce similar 3am waking patterns but respond to different approaches.
Related Reading
- Hormonal Sleep Disruption in Men — How testosterone, cortisol, growth hormone, and GABA interact to fragment sleep in men
- Can Low GABA Cause Waking Up at 3am? — The GABAergic mechanism behind 3am waking
- What Are the Signs of Low GABA at Night? — How to recognize impaired GABAergic function
- Do GABA Supplements Help You Stay Asleep Through the Night? — Trial evidence for oral GABA and the blood-brain barrier question
- Does GABA Affect Testosterone and Sleep in Men? — How testosterone-derived neurosteroids modulate GABA-A receptors
- How Do You Increase GABA Levels Naturally for Better Sleep? — Exercise, yoga, fermented foods, and gut health for GABA support
- Does Low Testosterone Cause Sleep Problems in Men? — The bidirectional testosterone-sleep feedback loop
- Does Growth Hormone Decline Affect Your Sleep After 40? — GH-SWS coupling and age-related decline
- Does Andropause Cause Insomnia? What Men Over 50 Need to Know — Compound hormonal decline in aging men
- Can Belly Fat Lower Your Testosterone and Disrupt Your Sleep? — How visceral fat suppresses testosterone through aromatase and disrupts sleep
- Can Ultra-Processed Food Lower Testosterone and Disrupt Sleep? — How endocrine-disrupting chemicals in processed food suppress testosterone and degrade sleep
- Can Inflammation Suppress Testosterone and Disrupt Your Sleep? — How inflammatory cytokines suppress testosterone production at the brain and testicular level
- Does Testosterone Replacement Therapy Affect Sleep? — How TRT affects sleep quality, its interaction with sleep apnea, and the role of body weight
References
1. Leproult, R., Copinschi, G., Buxton, O., & Van Cauter, E. (1997). Sleep loss results in an elevation of cortisol levels the next evening. Sleep, 20(10), 865-870. https://pubmed.ncbi.nlm.nih.gov/9415946/
2. Balbo, M., Leproult, R., & Van Cauter, E. (2010). Impact of sleep and its disturbances on hypothalamo-pituitary-adrenal axis activity. International Journal of Endocrinology, 2010, 759234. https://pubmed.ncbi.nlm.nih.gov/20628523/
3. Liu, P. Y., & Reddy, R. T. (2022). Sleep, testosterone and cortisol balance, and ageing men. Reviews in Endocrine and Metabolic Disorders, 23, 1323-1339. https://pubmed.ncbi.nlm.nih.gov/36152143/
4. Hirokawa, K., Sakamoto, T., Kasahara, S., & Murata, C. (2022). Associations of testosterone and cortisol concentrations with sleep quality in Japanese male workers. Comprehensive Psychoneuroendocrinology, 12, 100159. https://pubmed.ncbi.nlm.nih.gov/36148025/
5. Cohn, A. Y., Goel, N., Engeda, J. C., Grinspoon, S. K., & Klibanski, A. (2023). Effects of sleep fragmentation and estradiol decline on cortisol in a human experimental model of menopause. Journal of Clinical Endocrinology & Metabolism, 108(11), e1347-e1356. https://pubmed.ncbi.nlm.nih.gov/37207451/
6. Agrawal, S., Bhatt, N., Engel, A. J., & Dobs, A. S. (2024). Sleep disorders are associated with testosterone deficiency and erectile dysfunction — a U.S. claims database analysis. Andrology, 12(3), 589-596. https://pubmed.ncbi.nlm.nih.gov/36473958/
7. Park, J. Y., Sakamoto, T., Sato, S., & Hirokawa, K. (2024). Association of salivary testosterone levels during the post-awakening period with age and symptoms suggestive of late-onset hypogonadism in men. Annals of Medicine, 56(1), 2354891. https://pubmed.ncbi.nlm.nih.gov/38776237/
8. Velazquez Sanchez, C., & Dalley, J. W. (2025). The cortisol awakening response: Fact or fiction? Psychoneuroendocrinology, 175, 107404. https://pubmed.ncbi.nlm.nih.gov/40297522/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 8 references cited
