Waking up at 3am is a frequent sleep complaint, and impaired GABAergic function is a primary mechanism behind it.
This article explains how GABAergic inhibition maintains sleep continuity, what happens when GABA and cortisol interact in the second half of the night, and why receptor responsiveness matters more than GABA levels alone. It does not cover GABA supplements (that is a separate article) or behavioral approaches to improving GABAergic tone.
GABA-related sleep disruption is one of several mechanisms behind hormonal sleep disruption in men. The full picture — including how testosterone, cortisol, and growth hormone interact with GABAergic tone — is in Why Men’s Hormones Disrupt Sleep.
What Is the Role of GABA in Keeping You Asleep?
To stay asleep, your brain has to continuously suppress the circuits that promote wakefulness. The locus coeruleus (which drives norepinephrine-based alertness), the histaminergic tuberomammillary nucleus (which maintains waking arousal), and the ascending reticular formation (which regulates consciousness) all need to stay suppressed through the night. GABA is what keeps them suppressed.
It does this through two distinct receptor mechanisms (Winsky-Sommerer, 2009):
Phasic inhibition occurs at synapses — brief, precise bursts of GABA-A receptor activation that suppress individual neural firing events. This is useful for moment-to-moment regulation, but it is not what maintains sleep across hours.
Tonic inhibition is the mechanism that matters for staying asleep. Extrasynaptic GABA-A receptors — particularly those containing the delta subunit — respond to low ambient concentrations of GABA that are present in the extracellular space. This creates a continuous suppressive tone across large populations of neurons, holding the brain’s excitability below the threshold where arousal circuits would activate.
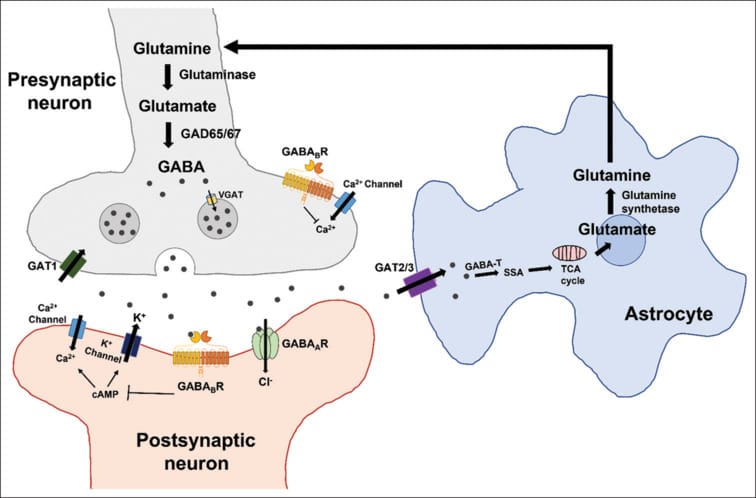
The preoptic area and ventral tegmental area are the two primary GABAergic hubs that coordinate this (Oishi et al., 2023). GABAergic neurons in the preoptic area actively silence the locus coeruleus and histaminergic tuberomammillary nucleus during sleep. When these preoptic neurons lose their inhibitory strength — whether from age-related decline, receptor downregulation, or competing excitatory input — the arousal circuits they are supposed to suppress begin firing. You wake up.
The distinction between phasic and tonic inhibition is directly relevant to why you might fall asleep without difficulty but wake up at 3am. Sleep onset can be initiated with relatively brief bursts of inhibition. Maintaining sleep for 7–8 hours requires the sustained tonic conductance that only extrasynaptic GABA-A receptors provide.
What Is the Relationship Between GABA and Cortisol at 3am?
Cortisol follows a circadian rhythm. It reaches its lowest point around midnight, then begins a steep rise between 2am and 4am, peaking shortly after waking. This is normal physiology — every healthy person experiences this pre-dawn cortisol increase.
The reason people sleep through it is that GABAergic tonic inhibition is strong enough to hold the arousal threshold above the level the cortisol rise can reach. The cortisol goes up. GABA keeps the brain’s excitability below the point where that cortisol rise would produce a waking event.
When GABAergic tone is weakened, the math changes. The same cortisol rise that healthy GABAergic tone absorbs now crosses the arousal threshold. You go from asleep to awake — often abruptly, often with a wired-rather-than-tired quality that makes returning to sleep difficult.
A study by Chapotot et al. (2001) measured this interaction directly in 10 healthy young men. They found that cortisol secretory rate tracks EEG-measured brain arousal with a consistent 10-minute lag (average R = 0.458, p < 0.001). Cortisol does not initiate the waking — it follows it. The arousal event comes first, and cortisol amplifies and sustains it.
This is an important distinction. In a GABA-driven 3am waking:
- GABAergic tonic inhibition weakens, allowing arousal circuits to activate
- Cortical arousal increases (measurable on EEG within minutes)
- Cortisol secretion follows approximately 10 minutes later, reinforcing and prolonging the waking state
The 2–4am window is where this tends to happen because two curves converge: cortisol is rising steeply, and GABAergic demand is at its peak. The second half of your sleep is when GABAergic inhibition faces its heaviest load — and where any weakness in receptor function or inhibitory tone becomes consequential.
After sleep deprivation, this coupling between cortisol and arousal was attenuated (R = 0.276) even as both cortisol levels and waking EEG power increased (Chapotot et al., 2001). Sleep loss itself raised cortisol pulse amplitude, suggesting a compounding relationship: poor sleep weakens GABAergic regulation, which produces more cortisol-amplified waking, which further degrades sleep.
Is GABA Deficiency the Cause of Primary Insomnia?
The intuitive assumption — that insomnia means your brain does not make enough GABA — does not hold up under the evidence.
Morgan et al. (2012) used proton magnetic resonance spectroscopy (MRS) to measure occipital cortex GABA in 16 adults with primary insomnia and 17 matched controls. The insomnia group had GABA levels that were 12% higher than controls — yet they averaged 72 fewer minutes of total sleep time per night and spent approximately 40 more minutes awake after initially falling asleep.
Within both groups, higher GABA did correlate with less time awake after sleep onset (p < 0.05). But at the group level, the insomnia group had more GABA and worse sleep. The elevated GABA may represent a compensatory upregulation — the brain producing more of the neurotransmitter to compensate for receptors that are no longer responding to it adequately.
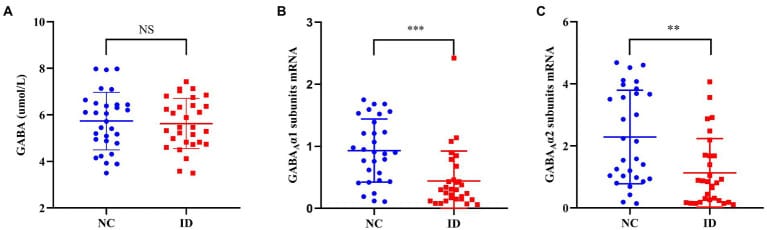
Xiang et al. (2023) confirmed this at the receptor level. In 30 adults with insomnia and 30 controls, serum GABA concentrations showed no difference between groups (p = 0.733). But GABA-A receptor alpha-1 subunit mRNA was reduced in the insomnia group (p < 0.001), and alpha-2 subunit mRNA was similarly downregulated (p = 0.001). The alpha-1 subunit expression negatively correlated with sleep quality and total sleep time; the alpha-2 subunit expression inversely correlated with daytime impairment.
Your brain may produce enough GABA. But if the receptors that respond to it are downregulated, the GABA cannot do its job. The inhibitory tone that should suppress arousal circuits through the night is reduced — not because the neurotransmitter is absent, but because the receptors are less responsive to it.
Regional specificity also matters. MRS data reviewed by Varinthra et al. (2024) show that individuals sleeping fewer than 6 hours per night had lower GABA concentrations in the anterior cingulate cortex and medial prefrontal cortex compared to normal sleepers. These are the regions that govern rumination suppression and top-down arousal control — which is consistent with the racing-thoughts quality that characterizes many 3am waking episodes.
This receptor-level understanding explains why “taking more GABA” — whether through supplements or diet — may not resolve the problem. If the receptors are downregulated, increasing the amount of GABA available does not restore the inhibitory tone that keeps you asleep.
What Is the Difference Between Falling Asleep and Staying Asleep?
This distinction matters because the supplement and behavioral strategies that help with sleep onset do not address sleep maintenance — and vice versa.
Sleep onset is driven by two converging processes. Adenosine accumulates during wakefulness and creates homeostatic sleep pressure — the longer you have been awake, the stronger the drive to sleep. Melatonin provides the circadian timing component, rising in the evening to open the sleep window. Together, they initiate the transition from wakefulness to sleep.
Sleep maintenance requires something different: continuous GABAergic tonic inhibition that suppresses arousal circuits for 7–8 hours against increasing excitatory pressure. Adenosine is metabolized during sleep (its pressure decreases as the night progresses). Melatonin declines in the second half of the night. By 3am, the neurochemical conditions that helped you fall asleep have largely dissipated — and GABA-mediated inhibition is what remains between you and a waking event.
This is why melatonin does not fix maintenance insomnia. Melatonin has no effect on GABAergic tone. It does not suppress the arousal circuits that become active in the second half of the night. If you fall asleep within a reasonable time but wake at 3am and cannot return to sleep, GABAergic inhibition — not melatonin — is the mechanism to investigate.
Pharmacological research supports this distinction directly. Wang et al. (2024) reviewed the development of partial GABA-A receptor agonists with half-lives of approximately 4 hours, designed to provide sustained GABAergic augmentation through the 2am–6am window when endogenous tonic inhibition is weakest. These compounds reduce wakefulness after sleep onset without producing next-day sedation — a pharmacokinetic profile that would be irrelevant if GABA were not the rate-limiting factor in second-half-of-night sleep maintenance.
If you fall asleep without difficulty but consistently wake in the second half of the night, the mechanism driving the waking is different from what caused any initial difficulty falling asleep. Addressing sleep onset (melatonin, sleep hygiene, relaxation techniques) without addressing GABAergic tone leaves the maintenance problem untouched.
Impaired GABAergic function rarely acts alone. In men, declining testosterone reduces neurosteroid-mediated GABA-A receptor support, while elevated cortisol from chronic stress or poor sleep further challenges GABAergic inhibition. Metabolic disruption, inflammatory load, and circadian misalignment might also be compounding the problem.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Does Low GABA Cause Waking Up at 3am?
The 2–4am window is where the cortisol circadian rise intersects with the period of peak demand on GABAergic tonic inhibition. When GABAergic function is impaired at either the neurotransmitter or receptor level, this is the window where sleep continuity breaks. The full mechanism — including the 10-minute lag between arousal and cortisol response — is covered above in What Is the Relationship Between GABA and Cortisol at 3am?.
Is Waking at 3am a GABA Deficiency?
The Xiang et al. (2023) study found no difference in serum GABA between people with insomnia and controls, while GABA-A receptor alpha-1 and alpha-2 subunit expression was reduced in the insomnia group. Morgan et al. (2012) found cortical GABA was higher in insomniacs. The receptor-level impairment is covered in detail in Is GABA Deficiency the Cause of Primary Insomnia? above.
What Neurotransmitter Keeps You Asleep?
Other neurotransmitters contribute to different aspects of sleep. Adenosine builds sleep pressure during wakefulness. Melatonin provides circadian timing for sleep onset. But sustained sleep maintenance through the night — particularly through the second half when arousal pressure increases — depends on GABAergic tonic inhibition. This is why virtually all prescription sleep-maintenance medications work by enhancing GABA-A receptor activity (Winsky-Sommerer, 2009).
What Brain Regions Are Affected by Low GABA in Insomnia?
MRS studies reviewed by Varinthra et al. (2024) show that short sleepers (fewer than 6 hours per night) had lower GABA concentrations in these prefrontal regions compared to normal sleepers. The anterior cingulate cortex is involved in suppressing intrusive thoughts and regulating emotional arousal — functions that, when impaired by low regional GABA, produce the characteristic experience of waking at 3am with a mind that will not settle.
Related Reading
- Hormonal Sleep Disruption in Men — How testosterone, cortisol, growth hormone, and GABA interact to fragment sleep in men
- What Are the Signs of Low GABA at Night? — How to recognize impaired GABAergic function
- Do GABA Supplements Help You Stay Asleep Through the Night? — Trial evidence for oral GABA and the blood-brain barrier question
- Does GABA Affect Testosterone and Sleep in Men? — How testosterone-derived neurosteroids modulate GABA-A receptors
- How Do You Increase GABA Levels Naturally for Better Sleep? — Exercise, yoga, fermented foods, and gut health for GABA support
- Does Low Testosterone Cause Sleep Problems in Men? — The bidirectional testosterone-sleep feedback loop
- Can a Cortisol Spike Wake You Up at 3am? — Cortisol-testosterone axis and nocturnal waking
- Does Growth Hormone Decline Affect Your Sleep After 40? — GH-SWS coupling and age-related decline
- Does Andropause Cause Insomnia? What Men Over 50 Need to Know — Compound hormonal decline in aging men
- Can Belly Fat Lower Your Testosterone and Disrupt Your Sleep? — How visceral fat suppresses testosterone through aromatase and disrupts sleep
- Can Ultra-Processed Food Lower Testosterone and Disrupt Sleep? — How endocrine-disrupting chemicals in processed food suppress testosterone and degrade sleep
- Can Inflammation Suppress Testosterone and Disrupt Your Sleep? — How inflammatory cytokines suppress testosterone production at the brain and testicular level
- Does Testosterone Replacement Therapy Affect Sleep? — How TRT affects sleep quality, its interaction with sleep apnea, and the role of body weight
References
1. Winsky-Sommerer, R. (2009). Role of GABAA receptors in the physiology and pharmacology of sleep. European Journal of Neuroscience, 29(9), 1779–1794. https://pubmed.ncbi.nlm.nih.gov/19473233/
2. Morgan, P. T., et al. (2012). Cortical GABA levels in primary insomnia. Sleep, 35(6), 807–814. https://pubmed.ncbi.nlm.nih.gov/22654200/
3. Chapotot, F., et al. (2001). Hypothalamo-pituitary-adrenal axis activity is related to the level of central arousal. Neuroendocrinology, 73(5), 312–321. https://pubmed.ncbi.nlm.nih.gov/11399904/
4. Xiang, T., et al. (2023). Impairment of GABA inhibition in insomnia disorders: Evidence from the peripheral blood system. Frontiers in Psychiatry, 14, 1134434. https://pubmed.ncbi.nlm.nih.gov/36846238/
5. Oishi, Y., Saito, Y. C., & Sakurai, T. (2023). GABAergic modulation of sleep-wake states. Pharmacology & Therapeutics, 249, 108505. https://pubmed.ncbi.nlm.nih.gov/37541595/
6. Varinthra, P., et al. (2024). The role of the GABAergic system on insomnia. Tzu Chi Medical Journal, 36(3), 232–240. https://pubmed.ncbi.nlm.nih.gov/38645778/
7. Wang, W., et al. (2024). Progress in GABAA receptor agonists for insomnia disorder. Frontiers in Pharmacology, 15, 1462286. https://pubmed.ncbi.nlm.nih.gov/39564117/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
