NAD+ declines with age, and both NMN (nicotinamide mononucleotide) and NR (nicotinamide riboside) are marketed as ways to restore it. The supplement industry frames them as interchangeable NAD+ precursors, but their research evidence for sleep is not equivalent. That distinction matters when the choice means spending $40-80/month on something that may not address the sleep problem.
This article covers what each precursor does in controlled trials measuring sleep outcomes — not general longevity claims or NAD+ blood levels alone. It does not cover dosing timing optimization, which is addressed in a separate article in this cluster.
NAD+ decline is one of several metabolic causes of sleep disruption. For the full overview of how metabolic changes drive 3am wakeups, see Metabolic Sleep Disruption.
What Does Nicotinamide Mononucleotide Do for Sleep?
The strongest NMN sleep data comes from Kim et al. (2022), a randomized, double-blind, placebo-controlled trial of 108 older Japanese adults. Participants were divided into four groups: NMN taken in the morning, NMN taken in the afternoon, and matching placebo groups for each timing. The dose was 250 mg NMN daily for 12 weeks.
The afternoon NMN group showed the largest effect sizes across sleep-related outcomes. Drowsiness reduction reached d=0.64 (a moderate effect), and physical performance on the 5-times sit-to-stand test reached d=0.72. Morning NMN dosing showed smaller effects. The researchers suggested that afternoon dosing may align with natural fatigue patterns that occur later in the day — a finding that positions dosing time as a variable, not just dose size.
A second RCT from Morifuji et al. (2024) enrolled 60 older adults receiving 250 mg/day beta-NMN or placebo for 12 weeks. Sleep quality improved on the PSQI daytime-function subscale (p=0.010) and Global PSQI score (p=0.013) in the NMN group. Blood NAD+ and its downstream metabolites (2-PY and 4-PY) were elevated throughout the study, indicating that oral NMN at this dose reaches the NAD+ pool. No adverse effects were reported.
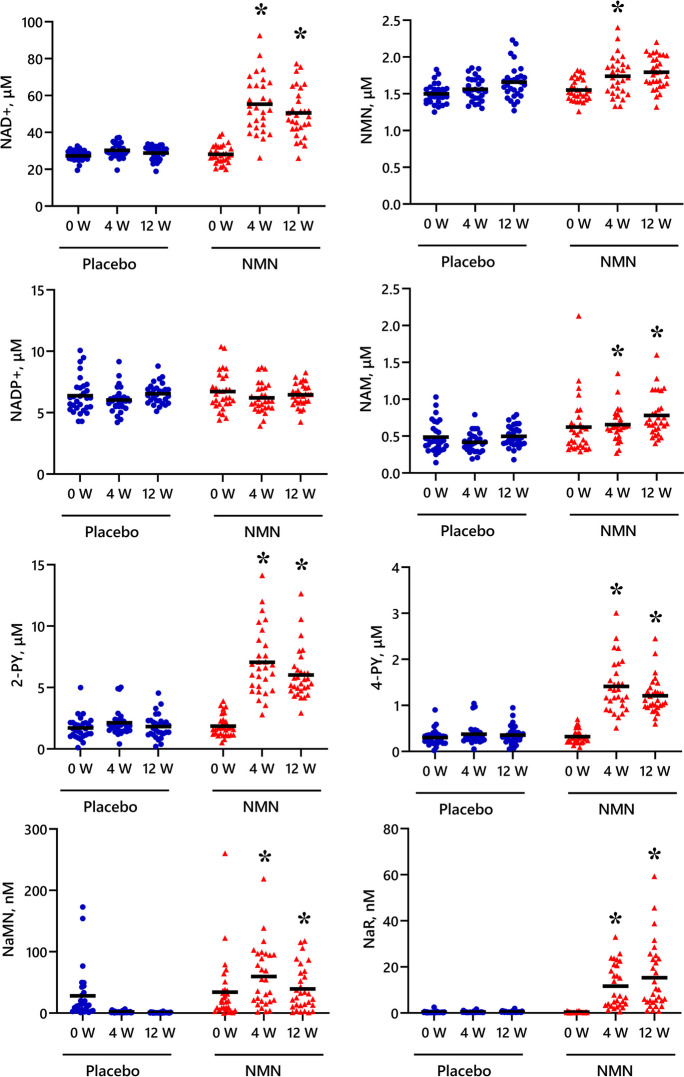
A larger trial is underway. Gao et al. (2023) published the design for a 400-participant multicenter RCT testing NMN at 320 mg/day for 60 days in adults with chronic insomnia, using PSQI as the primary endpoint. When completed, this will be the first large-scale trial positioning NMN as a supplement for chronic insomnia rather than general wellness. Results have not yet been published.
The mechanism connecting NMN to sleep runs through NAD+ restoration of SIRT1 activity, which in turn regulates the molecular clock genes that time the sleep-wake cycle. This pathway is covered in detail below.
What Does Nicotinamide Riboside Do for Sleep?
The primary NR human trial is Dellinger et al. (2017), which enrolled 120 healthy adults aged 60-80 in a randomized, double-blind, placebo-controlled design. Participants received either a standard dose or double dose of NRPT (NR combined with pterostilbene) for eight weeks. The standard dose raised whole-blood NAD+ by approximately 40%, and the double dose by approximately 90% at 4 weeks. The standard-dose elevation was sustained through the study period; the double-dose elevation partially declined to approximately 55% by week 8. No serious adverse events were reported. Sleep was not a measured endpoint in this trial, but it established the NAD+ elevation benchmark against which NMN results are routinely compared.
The primary NR sleep data comes from Wu et al. (2025), a 24-week, double-blind, placebo-controlled trial in 58 long-COVID participants. NR was dosed at 2000 mg/day — higher than the NMN trials. NAD+ levels increased 2.6- to 3.1-fold compared to baseline. Despite this NAD+ elevation, the between-group difference in sleep quality did not reach statistical threshold (p=0.69). An exploratory within-group analysis pooling all participants during their first 10 weeks on NR did show improvement, but without placebo control that analysis lacks rigor. The trial also had high dropout — 51.4% by 20 weeks — which limits interpretation of later timepoints.
Weiss (2025) published a narrative review synthesizing NR’s animal and human sleep evidence. In animal models, NR supplementation enhanced expression of the circadian clock genes BMAL1 and PER2, reduced NREM sleep by approximately 17%, increased REM sleep, and reduced oxidative stress in microglia during chronic sleep deprivation. In human data, the review noted that NR improved sleep efficiency in young and middle-aged males with insomnia in one study. The author called for larger RCTs, particularly ones including women and diverse populations, to evaluate these findings.
The pattern here is notable: NR raises NAD+ more reliably than almost any supplement, but NAD+ elevation alone does not appear sufficient to improve sleep in controlled human trials. Something beyond raw NAD+ availability is needed — a question the mechanism section below addresses.
What Is the Difference Between Nicotinamide Mononucleotide and Nicotinamide Riboside for Sleep?
Laid out side by side, the controlled sleep evidence is asymmetric. NMN has two positive sleep RCTs (Kim 2022, Morifuji 2024), with a third large insomnia trial in progress (Gao 2023). NR has one null sleep RCT (Wu 2025, p=0.69), promising animal data (Weiss 2025), and no completed RCT with sleep as a primary endpoint.
Prokopidis et al. (2025) published the strongest head-to-head comparison available: a review and meta-analysis pooling 6 NMN and 4 NR RCTs on shared endpoints including muscle mass and physical function. Neither precursor showed measurable effects on these endpoints. The analysis noted population-specific differences — NR improved walking distance in peripheral artery disease but was associated with lower physical performance scores in adults with mild cognitive impairment. Sleep was not a pooled outcome in this meta-analysis, so the NMN-vs-NR sleep comparison still relies on cross-study inference.
The biochemical difference between the two precursors may help explain the gap. NMN is one enzymatic step from NAD+ — it is converted directly by the enzyme NMNAT. NR requires two steps: first, NRK1 phosphorylates NR into NMN, and then NMNAT converts that NMN into NAD+. Both molecules feed the same NAD+ pool, but tissue-specific conversion rates and bioavailability differ. This may explain why equivalent NAD+ blood elevation does not produce equivalent effects on sleep.
No head-to-head RCT has compared NMN versus NR using sleep as a primary outcome. All current comparisons are cross-study. Until a direct comparison trial is completed, the evidence favors NMN for sleep, because NMN is the precursor with positive controlled sleep data. NR may yet show sleep benefits in future trials designed with sleep as a primary endpoint.
How Does Nicotinamide Adenine Dinucleotide Affect the Sleep-Wake Cycle?
The connection between NAD+ and sleep runs through a specific molecular pathway. NAD+ is the required cofactor (a molecule that an enzyme needs to function) for SIRT1, a protein deacetylase. SIRT1 is recruited to the CLOCK:BMAL1 chromatin complex, where it regulates the amplitude and timing of circadian gene expression. When NAD+ drops below sufficient concentrations, SIRT1 cannot maintain clock gene oscillation — and the circadian rhythm that dictates when the body sleeps and wakes loses its precision.

Weiss (2025) identified this NAD+/SIRT1 axis as the central mechanism linking NAD+ precursor supplementation to sleep architecture changes. The animal NR data — enhanced BMAL1 and PER2 expression, reduced NREM sleep, increased REM sleep — all run through this pathway.
NAD+ levels decline progressively after age 40. This decline is one contributor to circadian rhythm degradation in aging, alongside melatonin decline and other mechanisms covered in the Metabolic Sleep Disruption pillar.
But NAD+ elevation alone may not be sufficient to restore sleep. The Wu 2025 NR trial raised NAD+ 2.6- to 3.1-fold with no measurable sleep benefit. This suggests the NAD+-to-sleep pathway involves bottlenecks beyond NAD+ availability. Tissue-specific delivery, SIRT1 expression levels, and downstream clock gene responsiveness may each play a role. Restoring the cofactor does not ensure the enzyme or the genes it targets will respond at the same rate.
NAD+ decline is rarely the only metabolic factor disrupting sleep. In adults over 40, blood sugar instability, cortisol rhythm changes, hormonal fluctuations, and inflammatory pathways might be compounding alongside weakened NAD+ metabolism — and each pathway responds to different approaches.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Does Nicotinamide Adenine Dinucleotide Help With Sleep?
NAD+ does not induce sleep the way melatonin does. Instead, it supports the molecular clock that times when sleep occurs. The SIRT1 enzyme requires NAD+ to regulate the CLOCK:BMAL1 complex, and when NAD+ is insufficient, the precision of that clock degrades. Supplementing NAD+ precursors can restore SIRT1 activity, but as the NR data shows, raising NAD+ blood levels does not ensure sleep improvement — the downstream pathway from SIRT1 to clock genes to sleep architecture has its own constraints (Weiss, 2025).
How Long Does It Take for Nicotinamide Adenine Dinucleotide Precursors to Affect Sleep?
Both the Kim 2022 and Morifuji 2024 NMN trials used 12-week supplementation periods before measuring sleep outcomes. The Dellinger 2017 NR trial showed NAD+ elevation at 4 weeks that sustained through 8 weeks. The gap between NAD+ elevation (weeks) and measurable sleep effects (months) reinforces that NAD+ availability is a necessary but not sufficient condition — the clock gene machinery needs time to respond to restored SIRT1 activity (Morifuji et al., 2024).
Does Timing of Nicotinamide Mononucleotide or Nicotinamide Riboside Supplementation Matter for Sleep?
The Kim 2022 trial is the only RCT to compare morning versus afternoon dosing of an NAD+ precursor. The afternoon NMN group achieved a drowsiness reduction effect size of d=0.64, while morning NMN showed a smaller effect. This is a single study, and the timing question needs replication — but the timing logic is consistent with afternoon fatigue patterns and NAD+ activation kinetics observed in animal studies (Kim et al., 2022). A full analysis of NMN timing and dose considerations is covered in a separate article in this cluster.
Is Nicotinamide Mononucleotide or Nicotinamide Riboside Safer for Long-Term Use?
Morifuji 2024 reported no adverse effects with NMN at 250 mg/day over 12 weeks. Dellinger 2017 reported no serious adverse events with NR over 8 weeks. Wu 2025 used the highest dose (NR 2000 mg/day) for the longest duration (24 weeks) with no serious adverse events attributed to NR, though the trial had a 51.4% dropout rate by 20 weeks — dropouts were attributed to multiple factors including adverse events, COVID-19 reinfection, medication changes, and scheduling constraints, which limit long-term interpretability. Neither precursor has multi-year safety data (Wu et al., 2025).
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep — the pillar article for metabolic sleep disruption, covering glucose regulation, cortisol rhythm, mitochondrial stress, NAD+ loss, and fat metabolism
- Does NMN Cause Insomnia? What Timing and Dose Matter — NMN timing, dose, circadian effects, and sleep continuity data
- Does Apigenin Protect Your NAD+ and Improve Sleep Through CD38 Inhibition? — CD38 inhibition, NAD+ depletion with age, and apigenin sleep evidence
- Does Your Body Divert Tryptophan From Melatonin to Make NAD+? — tryptophan allocation between melatonin, kynurenine, inflammation, and NAD+ synthesis
- Why Does Sleep Repair Your DNA — And Burn Through Your NAD+ to Do It? — PARP1, sleep pressure, DNA repair, and NAD+ demand during sleep
- Does Your Gut Decide Whether Your NAD+ Supplement Works? — gut microbiome conversion of NAD+ precursors and why supplement response varies
- Does NAD+ IV Therapy Improve Sleep? What the Evidence Shows — IV NAD+ evidence, safety, infusion effects, and comparison with oral precursors
- Why Does NAD+ Drop Faster in Women After 40 — And What Does That Mean for Sleep? — menopause, ovarian aging, NAD+ decline, and sleep disruption after 40
References
Dellinger, R. W., Santos, S. R., Morris, M., Evans, M., Alminana, D., Guarente, L., & Marcotulli, E. (2017). Repeat dose NRPT (nicotinamide riboside and pterostilbene) increases NAD+ levels in humans safely and sustainably: a randomized, double-blind, placebo-controlled study. NPJ Aging and Mechanisms of Disease, 3, 17. https://pubmed.ncbi.nlm.nih.gov/29184669/
Gao, X., Li, J., Xu, S., Li, X., Wang, X., Li, Y., Huang, Y., Liu, S., & Zeng, Q. (2023). Oral nicotinamide mononucleotide (NMN) to treat chronic insomnia: protocol for the multicenter, randomized, double-blinded, placebo-controlled trial. Trials, 24(1), 340. https://pubmed.ncbi.nlm.nih.gov/37202819/
Kim, M., Seol, J., Sato, T., Fukamizu, Y., Sakurai, T., & Okura, T. (2022). Effect of 12-week intake of nicotinamide mononucleotide on sleep quality, fatigue, and physical performance in older Japanese adults: a randomized, double-blind placebo-controlled study. Nutrients, 14(4), 755. https://pubmed.ncbi.nlm.nih.gov/35215405/
Morifuji, M., Higashi, S., Ebihara, S., & Nagata, M. (2024). Ingestion of beta-nicotinamide mononucleotide increased blood NAD levels, maintained walking speed, and improved sleep quality in older adults in a double-blind randomized, placebo-controlled study. GeroScience, 46(5), 4671-4688. https://pubmed.ncbi.nlm.nih.gov/38789831/
Prokopidis, K., Moriarty, F., Bahat, G., McLean, J., Church, D. D., & Patel, H. P. (2025). The effect of nicotinamide mononucleotide and riboside on skeletal muscle mass and function: a systematic review and meta-analysis. Journal of Cachexia, Sarcopenia and Muscle, 16(3), e13799. https://pubmed.ncbi.nlm.nih.gov/40275690/
Weiss, C. (2025). Vitamin B3 ameliorates sleep duration and quality in clinical and pre-clinical studies. Nutrients, 17(12), 1982. https://pubmed.ncbi.nlm.nih.gov/40573093/
Wu, C. Y., Reynolds, W. C., Abril, I., McManus, A. J., Brenner, C., Gonzalez-Irizarry, G., Gutierrez-Martinez, L., Sun, O., Rosand, J., Tanzi, R. E., Arnold, S. E., & Guzman-Velez, E. (2025). Effects of nicotinamide riboside on NAD+ levels, cognition, and symptom recovery in long-COVID: a randomized controlled trial. EClinicalMedicine, 89, 103633. https://pubmed.ncbi.nlm.nih.gov/41357333/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 7 references cited
