Melatonin is widely recognized as a sleep molecule. Fewer people know about its counterpart: orexin, the neuropeptide that keeps the brain locked in a waking state. Newer sleep medications — suvorexant (Belsomra), lemborexant (Dayvigo), daridorexant (Quviviq) — work by blocking orexin receptors, but the mechanism they target has broader implications for anyone whose sleep fragments during the night without an obvious cause.
This article covers what orexin does, how the circadian clock controls when it fires, and what happens when that control weakens. It does not cover specific medications or approaches to modulate orexin — sibling articles in this cluster address those topics.
Orexin timing is one component of circadian sleep regulation. For the full picture of how circadian mechanisms can fragment sleep, see the parent pillar on Circadian Rhythm Disruption and Sleep.
How Does the Brain Decide Whether to Sleep or Stay Awake?
The transition between sleep and wakefulness is not gradual. The brain behaves more like a switch with two stable positions — awake or asleep — and it resists spending time in between. This behavior emerges from the anatomy of two opposing neuron populations.
On the sleep side: the ventrolateral preoptic area (VLPO) contains neurons that release GABA and galanin, both inhibitory neurotransmitters. When these neurons fire, they suppress the brain’s arousal centers — the locus coeruleus (norepinephrine), the tuberomammillary nucleus (histamine), the dorsal raphe (serotonin), and the lateral hypothalamic orexin neurons.
On the wake side: these same arousal centers fire during wakefulness and, in turn, inhibit the VLPO. Each side suppresses the other when active.
Saper et al. (2010) described this arrangement as a flip-flop switch. In electrical engineering, a flip-flop circuit has two stable states and transitions rapidly between them — it does not rest in a middle position. The mutual inhibition between the VLPO and the arousal network produces the same behavior in the brain: once one side gains enough advantage, it suppresses the other and locks into that state. This is why healthy sleep onset tends to happen within minutes rather than over an hour-long fade, and why waking up is usually a rapid transition rather than a slow climb toward consciousness.
Orexin’s role in this circuit is not to initiate wakefulness — it is to stabilize it. The orexin neurons in the lateral hypothalamus project to every major arousal center and reinforce their activity. Without orexin, the switch still functions, but it becomes unstable. It flips back and forth between states at inappropriate times.
The strongest evidence for this comes from narcolepsy type 1, a condition caused by the autoimmune destruction of orexin-producing neurons. People with narcolepsy do not lack the ability to wake up — they wake frequently. What they lack is stable state maintenance. They fall into sleep from wakefulness unpredictably (sleep attacks), and their sleep is fragmented by frequent awakenings. The problem is not too much sleep or too little — it is state instability.
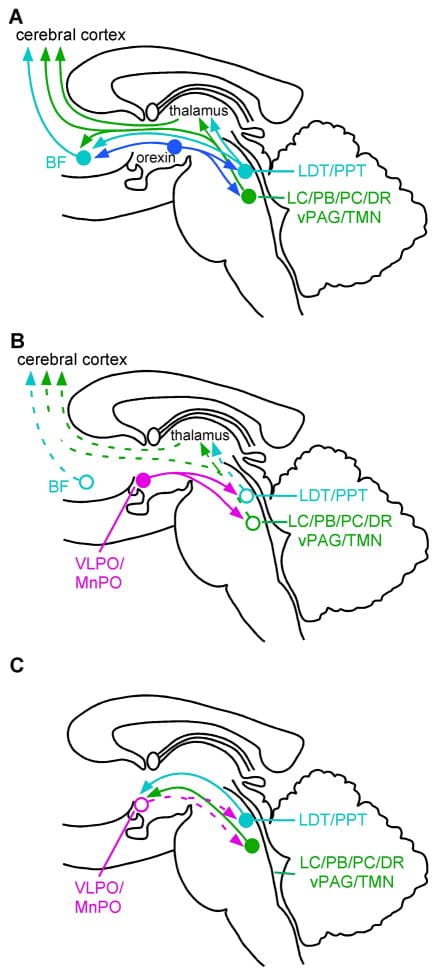
De Luca et al. (2022) added specificity to this model in mice. Using optogenetics — a technique that uses light to activate specific neurons — the researchers showed that orexin neurons projecting to the VLPO do not inhibit sleep-promoting neurons directly. Instead, orexin activates a population of GABAergic interneurons within the VLPO that do not express galanin (VLPO-GABA neurons). These interneurons then inhibit the galanin-expressing sleep-promoting neurons (VLPO-GABA/Gal neurons) through a feed-forward inhibition circuit. The result: orexin suppresses the sleep side of the switch through a relay, not a direct connection.
This feed-forward circuit has a practical implication. If orexin neurons remain active when they should be suppressed — during the circadian night — they hold the switch in the wake position even when sleep pressure (adenosine accumulation from prolonged wakefulness) is high. The person feels exhausted but remains awake, or falls asleep briefly and then wakes because the switch is not being held firmly in the sleep position.
How Does the Circadian Clock Control When Orexin Fires?
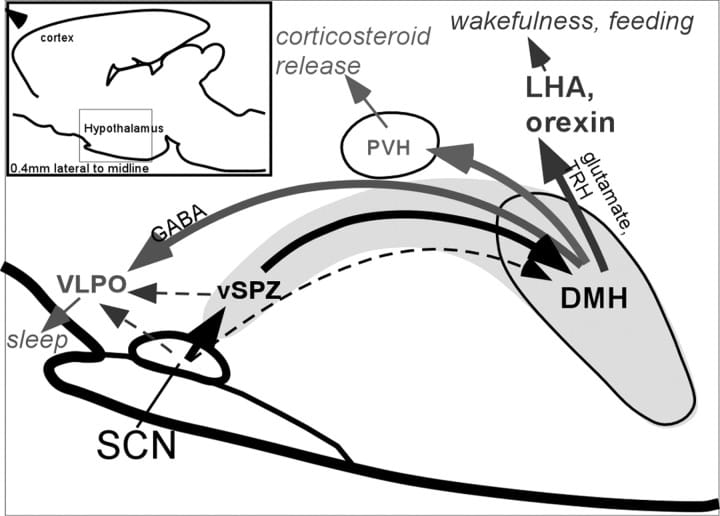
The suprachiasmatic nucleus (SCN) — the brain’s master circadian pacemaker, located in the anterior hypothalamus — does not project directly to orexin neurons. The circadian timing of orexin activity depends on a relay through the dorsomedial hypothalamus (DMH).
Chou et al. (2003) mapped this relay using lesion studies in rats. When the researchers destroyed 80% or more of DMH neurons, the animals lost 78–89% of the circadian rhythm in their sleep-wake cycles. The rats still slept and woke, but the timing became disorganized — sleep and wakefulness occurred in 3–4 hour ultradian bouts scattered across the 24-hour period instead of consolidating into a day-active, night-sleeping pattern. The DMH lesions also abolished the circadian rhythm of cortisol secretion while leaving melatonin rhythms intact, confirming the DMH’s role as a specific relay rather than a general loss of all circadian output.
The anatomical pathway runs: SCN sends projections to the subparaventricular zone (SPZ), which projects to the DMH. The DMH then sends excitatory (glutamate/thyrotropin-releasing hormone) projections to orexin neurons in the lateral hypothalamus, promoting wakefulness during the circadian day. Simultaneously, the DMH sends inhibitory (GABAergic) projections to the VLPO, suppressing sleep during the same period. At night, when SCN output changes, the DMH reduces its excitatory drive to orexin neurons and releases the VLPO from inhibition, permitting sleep.
Zeitzer et al. (2003) confirmed the timing of this rhythm in primates by measuring orexin-A concentrations in cerebrospinal fluid (CSF) sampled every two hours from squirrel monkeys. Orexin levels followed a circadian pattern: lowest during the early sleep period and peaking in the late active phase — the hours just before the habitual sleep onset. This late-day peak is consistent with orexin’s role as a wake stabilizer: orexin output rises through the day to counteract increasing sleep pressure (Process S, driven by adenosine accumulation), then drops at night to permit sleep.
This interaction between orexin timing (Process C, the circadian component) and sleep pressure (Process S, the homeostatic component) explains why healthy individuals experience a period of peak alertness in the early evening despite having been awake for 12+ hours. Orexin output is at its circadian maximum, counteracting the accumulated sleep drive.
Liu et al. (2023) identified a molecular mechanism within the DMH of mice that actively suppresses arousal at night. A population of GABAergic neurons in the DMH — expressing a protein called mWAKE (encoded by the ANKFN1 gene) — fires more at night than during the day. When the researchers deleted mWAKE in these mice, the DMH neurons became hyperexcitable specifically at night, leading to increased nighttime locomotor activity and heightened startle responses. The mWAKE protein cycles under control of the core circadian clock gene BMAL1 — when BMAL1 was knocked out, the mWAKE cycling was abolished.
This finding reframes the circadian control of arousal. The clock does not just withdraw the wake-promoting input at night. It actively generates a brake on arousal through mWAKE-expressing neurons. When that molecular brake weakens — because of circadian disruption, aging-related reduction in clock gene expression, or irregular schedules that weaken BMAL1 cycling — the result is excessive arousal during the circadian night. (This mechanism has been demonstrated in mice; whether mWAKE functions identically in humans is not yet confirmed.)
What Happens When Circadian Gating of Orexin Breaks Down?
The circadian gate over orexin can weaken through several mechanisms. Irregular light exposure, rotating work schedules, and late-night screen use can weaken SCN output, reducing the strength of the circadian input to the DMH relay. Aging reduces the amplitude of circadian rhythms in SCN firing, which in turn weakens the suppression of orexin at night. Chronic stress elevates corticotropin-releasing hormone (CRH), which directly excites orexin neurons independent of circadian input — stress can override the gate.
Muehlan et al. (2023) reviewed evidence that people with chronic insomnia show elevated orexin activity consistent with a hyperarousal phenotype. The hallmark of this state is not difficulty falling asleep per se — many people with insomnia fall asleep initially — but difficulty maintaining consolidated sleep through the night. At sleep cycle transitions, where the brain naturally comes close to wakefulness every 90–120 minutes, inappropriately active orexin tips the balance toward a full awakening rather than a smooth transition into the next cycle.
This mechanism explains a common experience: waking at 2–3 AM feeling alert despite being exhausted. At that time of night, sleep pressure remains high (adenosine has not fully dissipated), but if orexin neurons are firing when they should be suppressed, the wake-promoting drive competes with sleep pressure. The person wakes at a natural cycle transition, and the active orexin prevents rapid return to sleep. The mind races — not because of anxiety per se, but because orexin promotes cortical arousal. The subjective experience is being trapped between exhaustion and wakefulness.
Dual orexin receptor antagonists (DORAs) — the drug class that includes suvorexant, lemborexant, and daridorexant — work by blocking both OX1R and OX2R receptors. Their effectiveness in people with this hyperarousal phenotype supports the orexin-timing hypothesis: if the problem is orexin activity at the wrong time, blocking the orexin receptors during the sleep window addresses the timing mismatch without sedating the entire brain.
Braga et al. (2024) identified a metabolic vulnerability in the orexin circuit. Orexin neurons depend on lactate supplied by surrounding astrocytes (a type of glial cell) through monocarboxylate transporters — specifically MCT4 on astrocytes and MCT2 on orexin neurons. When the researchers deleted MCT4 from astrocytes in the lateral hypothalamus of mice, orexin neuron firing decreased and wakefulness fragmented. When they deleted MCT2 from orexin neurons, the same fragmentation occurred. Supplying exogenous lactate rescued wakefulness in the MCT4-deleted mice but not in the MCT2-deleted mice — confirming that the lactate must enter the orexin neuron through MCT2 to sustain firing.
This metabolic dependency means that anything affecting local energy availability in the lateral hypothalamus can influence orexin neuron activity. Metabolic conditions that alter brain lactate availability — glucose dysregulation, ketogenic diets, fasting states — may modulate orexin output independent of circadian input. Orexin output is not governed by the clock alone; it integrates metabolic state with circadian timing.
The convergence of these mechanisms — weakened circadian gating, stress-driven CRH excitation, metabolic inputs — means that orexin-mediated sleep fragmentation is often multifactorial. The circadian timing may be weakened rather than absent, while stress or metabolic factors add excitatory input that the weakened gate cannot override. The result is variable: some nights are worse than others, and the pattern can be difficult to identify from subjective experience alone.
Orexin timing is one of several circadian mechanisms that can fragment sleep. Cortisol phase advance, melatonin misalignment, peripheral clock decoupling, and metabolic or inflammatory factors might also be contributing. Identifying which causes might be active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Is Orexin the Same as Hypocretin?
Can Too Much Orexin Cause Insomnia?
What Is the Difference Between Orexin-A and Orexin-B?
Related Reading:
- Circadian Rhythm Disruption and Sleep — The full cause reference page covering all five circadian mechanisms
- How Does Orexin Change With Age? Why Sleep Fragments More After 50
- How Melatonin Suppresses Orexin: The Push-Pull That Decides Whether You Sleep or Stay Awake
- How Stress Hijacks Your Orexin: Why “Wired but Tired” Is a Real Brain State
- Can You Lower Orexin Naturally? What Controls Your Brain’s Wake Drive
- DORAs vs. Ambien: How Orexin-Blocking Sleep Medications Work Differently in Your Brain
References
Braga, A., Chiacchiaretta, M., Pellerin, L., Kong, D., & Haydon, P. G. (2024). Astrocytic metabolic control of orexinergic activity in the lateral hypothalamus regulates sleep and wake architecture. Nature Communications, 15(1), 5979. https://pubmed.ncbi.nlm.nih.gov/39013907/
Chou, T. C., Scammell, T. E., Gooley, J. J., Gaus, S. E., Saper, C. B., & Lu, J. (2003). Critical role of dorsomedial hypothalamic nucleus in a wide range of behavioral circadian rhythms. Journal of Neuroscience, 23(33), 10691–10702. https://pubmed.ncbi.nlm.nih.gov/14627654/
De Luca, R., Nardone, S., Grace, K. P., Venner, A., Cristofolini, M., Bandaru, S. S., … & Arrigoni, E. (2022). Orexin neurons inhibit sleep to promote arousal. Nature Communications, 13(1), 4163. https://pubmed.ncbi.nlm.nih.gov/35851580/
Liu, Q., Bell, B. J., Kim, D. W., Lee, S. S., Keles, M. F., Liu, Q., … & Wu, M. N. (2023). A clock-dependent brake for rhythmic arousal in the dorsomedial hypothalamus. Nature Communications, 14(1), 6381. https://pubmed.ncbi.nlm.nih.gov/37821426/
Muehlan, C., Roch, C., Vaillant, C., & Dingemanse, J. (2023). The orexin story and orexin receptor antagonists for the treatment of insomnia. Journal of Sleep Research, 32(6), e13902. https://pubmed.ncbi.nlm.nih.gov/37086045/
Saper, C. B., Fuller, P. M., Pedersen, N. P., Lu, J., & Scammell, T. E. (2010). Sleep state switching. Neuron, 68(6), 1023–1042. https://pubmed.ncbi.nlm.nih.gov/21172606/
Zeitzer, J. M., Buckmaster, C. L., Parker, K. J., Hauck, C. M., Lyons, D. M., & Mignot, E. (2003). Circadian and homeostatic regulation of hypocretin in a primate model: Implications for the consolidation of wakefulness. Journal of Neuroscience, 23(8), 3555–3560. https://pubmed.ncbi.nlm.nih.gov/12716965/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
