Insulin resistance is one of the central drivers of accelerated aging, and its effect on sleep is often invisible. Total sleep time can look normal while sleep architecture — the internal composition of each sleep stage — degrades underneath. A person sleeping seven or eight hours per night can still have inadequate slow-wave sleep, the stage responsible for overnight glucose regulation, tissue repair, and growth hormone release.
This matters for long-term brain health, cognitive protection, and metabolic resilience. The relationship between insulin resistance and sleep runs in both directions: insulin resistance degrades sleep quality, and degraded sleep quality worsens insulin resistance, compounding the damage over time.
This article covers the bidirectional relationship between insulin resistance and sleep quality, the evidence for a self-reinforcing loop, and why sleep patterns established in younger decades contribute to metabolic risk later in life. For a broader view of how metabolic health affects sleep, see Metabolic Sleep Disruption: The Full Picture.
How Does Insulin Resistance Suppress Deep Sleep?
The connection between insulin resistance and deep sleep was demonstrated directly in a 2008 crossover experiment by Tasali and colleagues. Nine healthy young adults had their slow-wave sleep selectively suppressed using acoustic stimulation for three consecutive nights — without reducing their total sleep time. The acoustic tones were calibrated to move sleep out of the deepest stages without waking the participants.
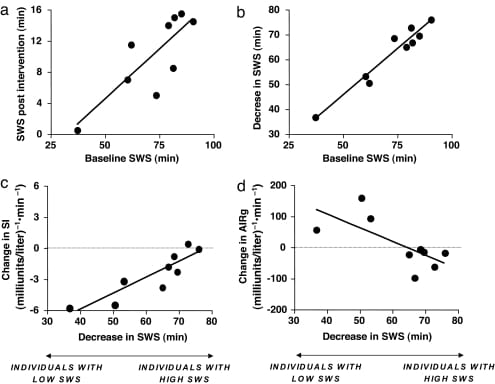
The metabolic consequences were measurable within days. Glucose tolerance declined by approximately 23%. Insulin sensitivity dropped, but insulin secretion did not increase to compensate (p = 0.73 for change in insulin secretion), creating a net metabolic deficit. The correlation between the degree of slow-wave sleep reduction and the degree of insulin sensitivity loss was r = 0.89 (p = 0.001) — a strong dose-response relationship (Tasali et al., 2008).
The participants who lost the greatest amount of deep sleep experienced the largest metabolic deterioration. And because total sleep time remained unchanged, standard sleep metrics would not have flagged anything wrong.
A 2022 meta-analysis by Sondrup and colleagues synthesized 35 randomized controlled trials examining five categories of sleep manipulation: sleep restriction, slow-wave sleep suppression, REM sleep disturbance, sleep fragmentation, and circadian misalignment. Two categories — slow-wave sleep suppression and circadian misalignment — each independently reduced insulin sensitivity. REM sleep disturbance and sleep fragmentation, in isolation, did not produce measurable effects on insulin sensitivity markers (Sondrup et al., 2022).
This distinction matters. Not all sleep disruption affects glucose metabolism equally. The metabolic impact is concentrated in deep sleep loss and in mistimed sleep — not in lighter-stage interruptions. Someone sleeping seven to eight hours but spending insufficient time in slow-wave sleep can still develop metabolic deterioration that does not show up in total sleep time tracking.
The Sondrup meta-analysis also found that the primary site of insulin sensitivity impairment from sleep manipulation appears to be hepatic and whole-body, rather than skeletal muscle — pointing toward a central metabolic effect rather than a peripheral one.
Does Poor Sleep Cause Insulin Resistance, or Does Insulin Resistance Cause Poor Sleep?
Observational studies can show that insulin resistance and poor sleep co-occur, but they cannot determine which came first.
A 2023 study by Wang and colleagues used genetic variants from the UK Biobank as causal inference instruments to test whether insomnia causally elevates insulin resistance traits. This method uses genetic variants as natural experiments — because genes are assigned at conception, they bypass the confounding and reverse-causation problems of observational designs. Across three analytical methods (observational, one-sample, and two-sample causal inference), frequent insomnia was associated with elevated triglyceride-glucose index (a validated surrogate for insulin resistance) and elevated TG/HDL-C ratio. Mediation analysis estimated that insulin resistance mediates approximately 25% of the total association between insomnia and incident type 2 diabetes (Wang et al., 2023).
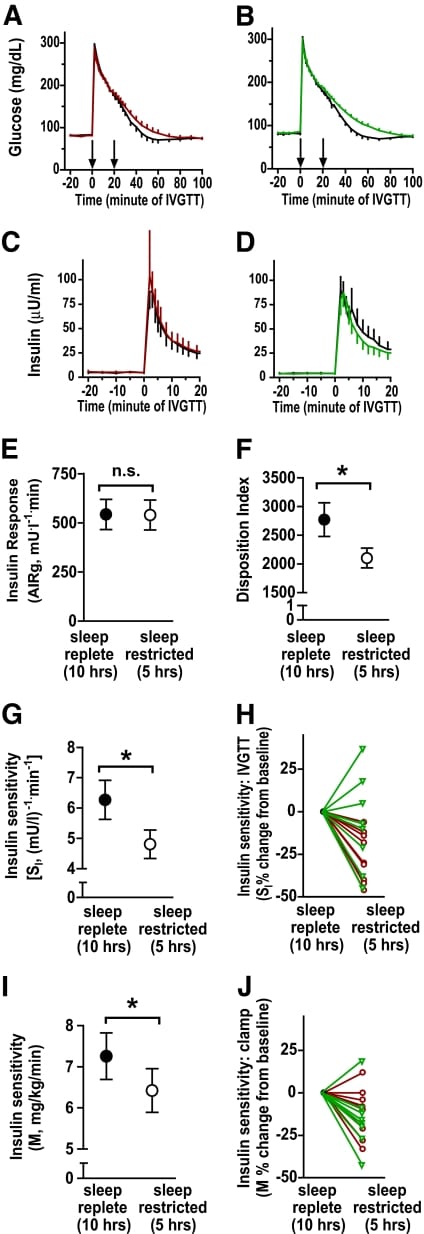
On the experimental side, Buxton and colleagues demonstrated in 2010 that restricting healthy men to five hours of sleep per night for one week — an amount many working adults regularly experience — was sufficient to reduce insulin sensitivity by 20% (p = 0.001) as measured by oral glucose tolerance test. Evening cortisol concentrations rose 51% during sleep restriction (p < 0.02), indicating activation of the stress axis. Half the cohort received modafinil to counteract sleepiness, yet metabolic deterioration was equivalent between groups — showing that the insulin resistance was not driven by subjective fatigue alone (Buxton et al., 2010).
A 2025 meta-analysis by Chen and colleagues, the largest participant pool assembled on this question (91 studies, 2,217,521 participants), quantified the population-level association. Poor sleep quality was associated with adverse glycemic control with an odds ratio of 1.63 (95% CI: 1.30–2.04). A formal insomnia condition carried a higher odds ratio of 1.87 (95% CI: 1.19–2.93). The odds of developing diabetes were elevated with poor sleep quality (OR = 2.28, 95% CI: 1.84–2.82) and with an insomnia condition (OR = 1.32, 95% CI: 1.12–1.56) (Chen et al., 2025).
The bidirectional loop works as follows: insulin resistance suppresses slow-wave sleep (as the Tasali 2008 data demonstrates). Reduced slow-wave sleep further impairs insulin sensitivity (as the Buxton 2010 and Sondrup 2022 data show). That worsened insulin sensitivity then further degrades sleep architecture, and the cycle continues.
Does Sleep Duration in Younger Adults Predict Insulin Resistance Later?
The relationship between sleep and insulin resistance is not limited to adults who already have metabolic impairment. Sleep patterns established during younger decades appear to set metabolic trajectories that compound over time.
A 2023 longitudinal study by Jansen and colleagues followed 362 adolescents enrolled in the ELEMENT birth cohort in Mexico City, with actigraphy-measured sleep assessed at two timepoints approximately two years apart during the peri-pubertal period. Adolescents sleeping one or more hours below age-appropriate recommendations had 2.74-fold higher odds of developing insulin resistance (95% CI: 1.01–7.43; p-trend = 0.05). Those with the latest sleep midpoints (after 4:33 AM) had 2.63-fold higher odds of insulin resistance (95% CI: 1.03–6.72; p-trend = 0.04) compared to those with the earliest sleep midpoints (Jansen et al., 2023).
Two findings stand out from this data. First, sleep timing was an independent contributor — late sleepers had elevated insulin resistance odds regardless of how many hours they slept. Second, adiposity change did not mediate the association. The sleep-to-insulin-resistance link ran through circadian and neuroendocrine pathways independent of weight gain.
At baseline, 51% of participants were already insulin resistant. By follow-up, prevalence had risen to 57%, with 17% of the total sample newly transitioning into insulin resistance during the observation window. Social jetlag and night-to-night variability in sleep duration were not associated with insulin resistance categories — the relevant exposures were chronic short duration and persistent late timing.
For adults focused on long-term metabolic health, this data carries an implication: the sleep habits that shape insulin sensitivity are not confined to the present. They accumulate. A pattern of insufficient or mistimed sleep during formative metabolic years can establish an insulin resistance trajectory that persists into adulthood — even without accompanying weight gain.
Frequently Asked Questions
Can Improving Insulin Resistance Improve Sleep?
The Tasali 2008 data runs in both directions. If experimentally suppressing slow-wave sleep worsens insulin sensitivity, then conditions that restore or protect slow-wave sleep would be expected to improve it. Dietary changes, physical activity, and weight management have each been associated with improvements in both insulin sensitivity and sleep architecture. These are not independent outcomes — they share overlapping physiological pathways including cortisol regulation, inflammatory load, and autonomic balance.
Addressing the metabolic side (reducing insulin resistance through dietary changes, exercise, or weight management) can also improve sleep architecture by reducing the overnight glucose instability that contributes to awakenings and by lowering the sympathetic nervous activation that suppresses deep sleep.
Can Insulin Resistance Cause Night Sweats?
Night sweats in the context of insulin resistance are often a downstream consequence of nocturnal hypoglycemia rather than a direct effect of insulin resistance itself. Insulin resistance produces exaggerated insulin responses to meals, which can lead to reactive blood sugar drops hours later — including during sleep. When blood glucose falls below a threshold, the body mounts a counter-regulatory response that includes epinephrine release. That epinephrine drives sweating, increased heart rate, and often an awakening.
For a deeper look at how overnight blood sugar drops affect sleep, see [Can a Blood Sugar Drop Wake You Up at 3am?].
What Are Early Indicators That Insulin Resistance May Be Affecting Sleep?
The Tasali 2008 data explains why someone can sleep seven or eight hours and still feel unrefreshed: if slow-wave sleep is suppressed — as it is in insulin resistance — total sleep time looks adequate while the restorative component is degraded. Standard sleep duration tracking would not detect this.
Early morning awakenings (between 2:00 and 4:00 AM) can indicate overnight glucose drops, which are more common in insulin-resistant individuals whose blood sugar regulation is less stable. Daytime fatigue that seems disproportionate to the amount of sleep obtained — especially combined with gradual weight gain or dietary changes — is another observable pattern.
Quantifiable markers provide more direct data. Fasting glucose, fasting insulin, and their ratio (HOMA-IR) can identify insulin resistance before it progresses to prediabetic glucose levels. HbA1c reflects average blood sugar over the preceding two to three months. Iyegha and colleagues (2019) found that 62% of prediabetic individuals report poor sleep quality, suggesting that sleep complaints and insulin resistance often co-occur before metabolic status is formally identified.
Sleep and metabolic health influence each other through multiple overlapping pathways — cortisol, inflammation, autonomic tone, glucose regulation. No single variable accounts for all of it. The 3AM Decoder can help identify which contributors are relevant to your data. Most people have 2-3 causes working together, not just one.
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep — the full metabolic cause overview
- Can a Blood Sugar Drop Wake You Up at 3am? — how nocturnal blood sugar drops trigger the hormonal cascade that wakes you up
- Why Do You Wake Up Hungry at 3am? — how leptin, ghrelin, and blood sugar interact during sleep
- Why Does Cortisol Spike at 3am and Wake You Up? — the cortisol-sleep feedback loop and HPA axis timing
- Can a Calorie Deficit Cause Insomnia? — how energy restriction elevates cortisol and fragments sleep
- Is Waking Up at 3am a Sign of Adrenal Fatigue? — the real HPA axis mechanism behind what people call adrenal fatigue
- Can Prediabetes Cause Sleep Problems? — how prediabetes changes sleep architecture and accelerates metabolic decline
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
References
Buxton, O. M., Pavlova, M., Reid, E. W., Wang, W., Simonson, D. C., & Adler, G. K. (2010). Sleep restriction for 1 week reduces insulin sensitivity in healthy men. Diabetes, 59(9), 2126–2133. https://pubmed.ncbi.nlm.nih.gov/20585000/
Chen, Y., Li, Y., Huang, Y., Tan, X., & Zheng, X. (2025). The association between insomnia (related symptoms) and glycaemic control: A systematic review and meta-analysis. Journal of Global Health, 15, 04016. https://pubmed.ncbi.nlm.nih.gov/39916567/
Iyegha, I. D., Chieh, A. Y., Bryant, B. M., & Li, L. (2019). Associations between poor sleep and glucose intolerance in prediabetes. Psychoneuroendocrinology, 110, 104444. https://pubmed.ncbi.nlm.nih.gov/31546116/
Jansen, E. C., Dunietz, G. L., Matos-Moreno, A., Lajous, M., Batalle-Claret, A., Tellez-Rojo, M. M., & Peterson, K. E. (2023). Sleep duration and timing are prospectively linked with insulin resistance during late adolescence. Obesity, 31(4), 912–922. https://pubmed.ncbi.nlm.nih.gov/36847394/
Sondrup, N., Termannsen, A. D., Eriksen, J. N., Hjorth, M. F., Færch, K., Klingenberg, L., & Quist, J. S. (2022). Effects of sleep manipulation on markers of insulin sensitivity: A systematic review and meta-analysis of randomized controlled trials. Sleep Medicine Reviews, 62, 101594. https://pubmed.ncbi.nlm.nih.gov/35189549/
Tasali, E., Leproult, R., Ehrmann, D. A., & Van Cauter, E. (2008). Slow-wave sleep and the risk of type 2 diabetes in humans. Proceedings of the National Academy of Sciences, 105(3), 1044–1049. https://pubmed.ncbi.nlm.nih.gov/18172212/
Wang, X., Zhao, C., Feng, H., Li, Y., Li, M., & Yang, L. (2023). Associations of insomnia with insulin resistance traits: A cross-sectional and Mendelian randomization study. Journal of Clinical Endocrinology & Metabolism, 108(8), e574–e582. https://pubmed.ncbi.nlm.nih.gov/36794917/
