The experience of waking at 3am with hunger or food cravings reflects measurable hormonal changes that happen when sleep and metabolic regulation fall apart. This is not a willpower problem — it is a neuroendocrine one.
Understanding the mechanism matters beyond the 3am wake-up itself. The same hormonal disruption that produces nighttime hunger also drives insulin resistance and weight gain over time, with downstream consequences for metabolic health, brain function, and longevity. This article covers how leptin, ghrelin, and blood sugar interact during sleep — and what goes wrong when that interaction breaks down. For the full picture of how metabolic processes disrupt sleep, see Metabolic Sleep Disruption: How Blood Sugar, Cortisol, and Hunger Hormones Fragment Your Sleep.
How Do Hunger Hormones Change During Sleep?
Two hormones regulate appetite across the 24-hour cycle. Leptin, produced by fat cells, acts as a satiety cue — it tells the brain that energy stores are adequate and suppresses the drive to eat. Ghrelin, produced primarily in the stomach, does the opposite — it drives hunger and food-seeking behavior.
During a full night of sleep, leptin concentrations rise and ghrelin stays low. This is why healthy sleepers can go 7-9 hours without eating and not wake up hungry. Appetite regulation stays suppressed while the body directs resources toward repair, memory consolidation, and hormonal restoration.
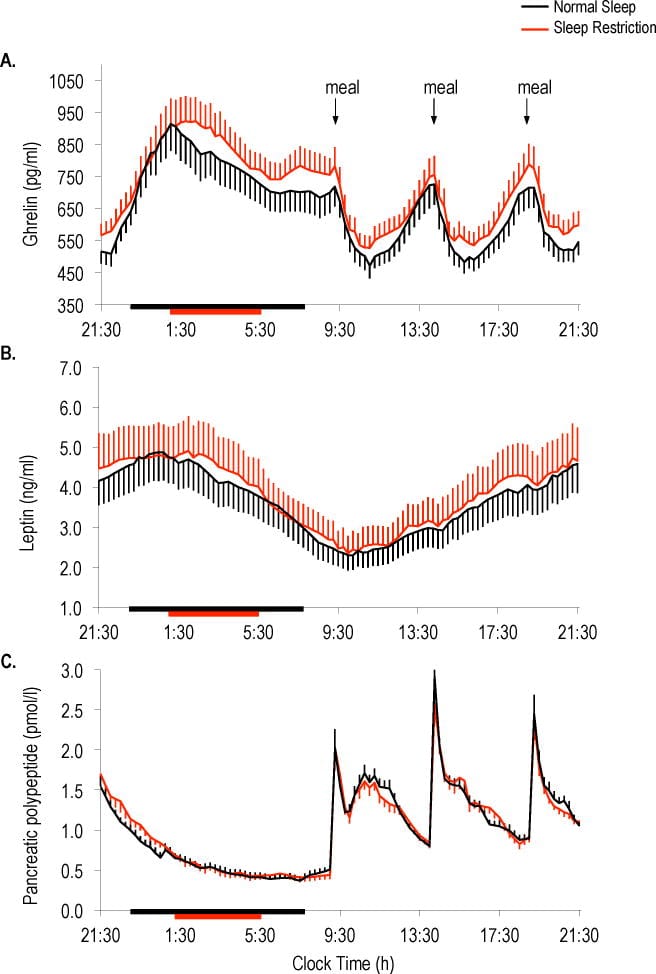
Sleep restriction disrupts this balance. In a controlled crossover study of 12 healthy young men (mean age 22, BMI 23.6), Spiegel et al. (2004) found that two nights of approximately 4 hours of sleep — compared to two nights of approximately 10 hours — produced an 18% decrease in leptin (p = 0.04), a 28% increase in ghrelin (p < 0.04), and a 24% increase in subjective hunger (p < 0.01) [1]. Cravings for calorie-dense, carbohydrate-rich foods rose by 33-45%.
These hormonal changes are not a consequence of weight gain. Taheri et al. (2004) measured leptin and ghrelin in 1,024 participants from the Wisconsin Sleep Cohort and found that participants sleeping 5 hours per night had 15.5% lower leptin and 14.9% higher ghrelin compared to those sleeping 8 hours — independent of BMI (p = 0.01 and p = 0.008, respectively) [2]. Short sleep predicted the hormonal disruption regardless of body composition.
The question of whether these findings apply beyond lean young men has been addressed. Van Egmond et al. (2023) studied 44 participants — including 20 women and approximately 19-20 participants with obesity — and found that one night of total sleep deprivation reduced leptin and elevated ghrelin in both sexes and across weight categories [4]. Ghrelin increases were more pronounced in participants with obesity, and leptin reductions were more pronounced in women. The appetite-disrupting hormonal response to sleep loss extends across demographics.
Does the Timing of Sleep Loss Affect Nocturnal Hunger?
People searching for “waking up at 3am hungry” are often struck by the consistency of the timing. The answer involves both circadian ghrelin regulation and conditioned metabolic rhythms.
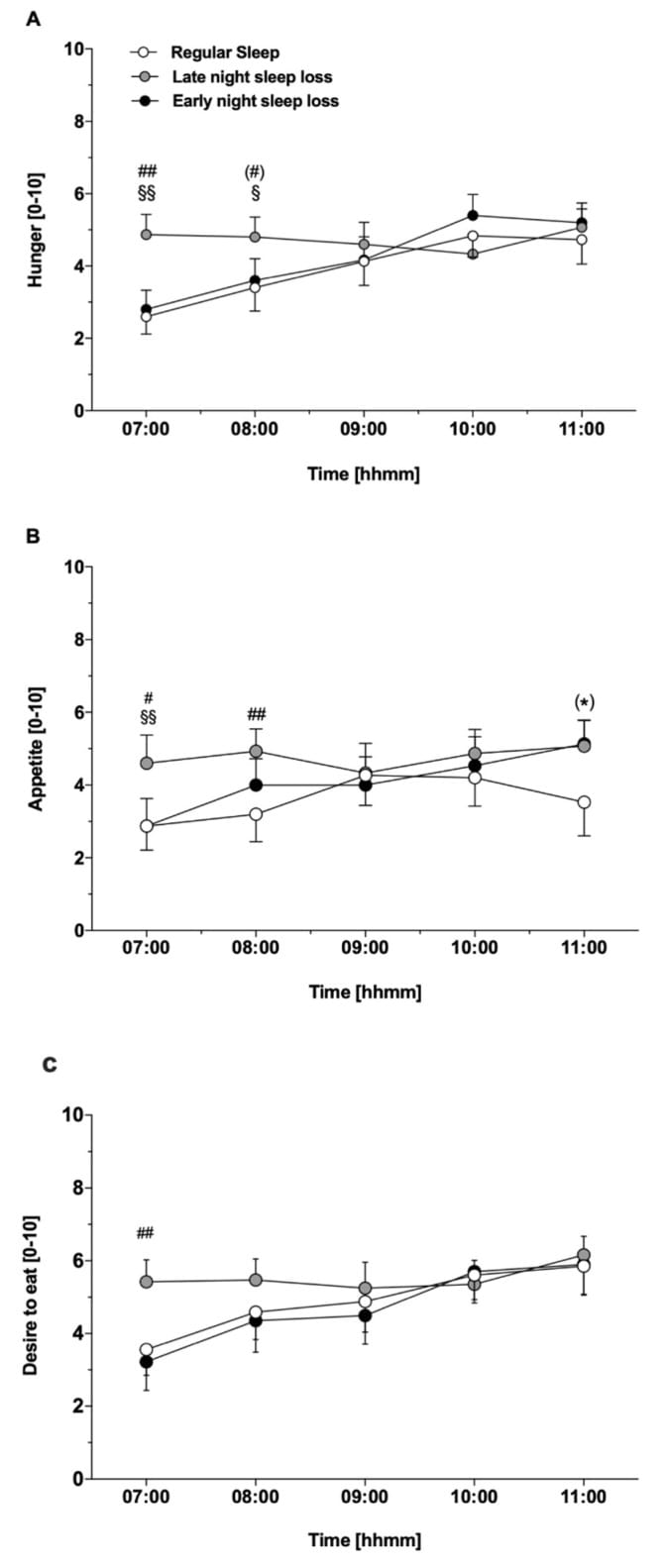
Meyhöfer et al. (2023) ran a crossover study with 15 healthy men comparing three conditions: 4 hours of early-night sleep (10pm-2am), 4 hours of late-night sleep (2am-6am), and 8 hours of full-night sleep [5]. The total hours of sleep lost were identical in the two restriction conditions. But ghrelin concentrations, hunger ratings, and desire for food increased only after late-night sleep loss — not after early-night sleep loss.
This is not coincidental. Ghrelin secretion follows a circadian rhythm that endogenously peaks in the late-night and early-morning hours. Losing sleep during this window appears to amplify or extend this ghrelin surge. For anyone whose sleep ends prematurely — new parents, night-schedule workers, people who wake at 3am and cannot fall back asleep — the hormonal cost is disproportionately concentrated in appetite disruption.
There is a second mechanism at work. Isherwood et al. (2023) demonstrated in a controlled 8-day laboratory study with 24 male participants that the body develops anticipatory glucose dips and hunger surges timed to habitual meal windows [6]. Participants who ate on a two-meal schedule developed circadian glucose troughs and hunger peaks that fired precisely when meals had previously occurred — and these rhythms persisted even when all external time cues and food were removed.
This has direct relevance to nighttime hunger. People who habitually eat at night — whether in response to a genuine glucose drop, stress, or habit — develop persistent circadian hunger and glucose rhythms that continue to drive food-seeking behavior even in the absence of an external trigger. The 3am wake-up-and-eat pattern, once established, can become self-reinforcing through circadian entrainment.
Is Nighttime Hunger a Blood Sugar Problem or a Hormone Problem?
The blood sugar pathway works like this: overnight glucose drops below the brain’s comfort threshold, the body releases cortisol and adrenaline to mobilize stored glucose, and these stress hormones produce arousal — waking you up. Hunger follows secondarily, as the brain registers low glucose availability. This pathway is covered in detail in Why Does Your Blood Sugar Drop at 3am and Wake You Up?.
The hunger hormone pathway is different. Elevated ghrelin directly drives hunger and food-seeking behavior, independent of glucose levels. You wake up because ghrelin activates reward-related brain circuits that make food — particularly calorie-dense carbohydrate foods — acutely appealing.
Broussard et al. (2016) quantified this in a crossover study of 19 healthy lean men: sleep restriction (~4.5 hours vs. ~8.5 hours) elevated 24-hour ghrelin levels and predicted an additional 328 kcal of snack intake (p = 0.03), concentrated in carbohydrates (p = 0.02) [3]. Elevated evening ghrelin correlated with sweet-food consumption (r = 0.48, p = 0.04). The excess intake was concentrated at snack occasions rather than main meals, consistent with ghrelin’s known role in reward-driven eating.
The distinction between the two pathways matters in principle, but in practice they often coexist. Insulin resistance worsens both glucose instability (making blood sugar drops more frequent and more severe) and leptin resistance (making the brain less responsive to satiety cues). A person with developing insulin resistance may experience both the blood sugar crash and the ghrelin surge — sometimes on the same night.
Tajiri et al. (2023) demonstrated in a free-living crossover trial with 24 young adults (13 women, 11 men) that sleep restriction increased hunger, desire for fatty foods, and snack carbohydrate intake under everyday conditions — not just in the lab [7]. The effect emerged autonomously in daily life without forced caloric restriction or controlled feeding schedules.
If you are waking up at 3am hungry and not sure which mechanism is driving the pattern, the 3AM Decoder can help you map the pattern to the underlying metabolic and hormonal inputs.
Should You Eat If You Wake Up Hungry at Night?
When overnight glucose drops low enough to trigger a counterregulatory hormone response, eating a small amount of food — particularly something with protein and fat rather than pure carbohydrate — can stabilize glucose and reduce the cortisol and adrenaline release that keeps you awake.
But if the 3am hunger is driven by a conditioned circadian eating rhythm — the kind Isherwood et al. (2023) demonstrated, where the body learns to expect food at a given time and produces anticipatory glucose dips and hunger surges — then eating at that time reinforces the cycle [6]. The circadian entrainment deepens, and the 3am hunger becomes more entrenched.
There is no universal eat-or-don’t-eat answer. The question to investigate is whether the hunger reflects acute glucose instability or a conditioned pattern — because the two require different responses.
Does Sleep Deprivation Make You Crave Sugar and Carbs?
The preference for carbohydrate-rich and sweet foods after sleep loss is not coincidental. Ghrelin activates mesolimbic reward pathways — the same brain circuits involved in motivation and reward — with a preference for palatable, energy-dense foods.
Spiegel et al. (2004) documented this: after two nights of 4 hours of sleep, cravings for cookies, candy, salty snacks, and starchy foods rose by 33-45% (p = 0.02) compared to the sleep extension condition [1]. Broussard et al. (2016) added behavioral evidence: elevated evening ghrelin during sleep restriction predicted not just greater hunger, but greater consumption of sweet foods (r = 0.48, p = 0.04), with excess caloric intake concentrated in snack carbohydrates [3].
This means the 3am sugar craving is a downstream effect of ghrelin elevation — a hormonally driven preference, not a lack of discipline.
Can Improving Your Sleep Reduce Nighttime Hunger?
Tajiri et al. (2023) tested this in 24 young adults under everyday conditions [7]. When participants extended their sleep from approximately 5 hours to approximately 8 hours, hunger ratings dropped, desire for fatty foods decreased, and snack carbohydrate intake fell — without any instructions about what or when to eat.
The implication is that the hormonal environment driving nighttime hunger responds to sleep duration. Leptin and ghrelin normalize when sleep duration is adequate, and the appetite-disrupting cascade reverses. This positions sleep extension — not dietary restriction — as the first-order approach for nocturnal hunger driven by hormonal disruption.
For conditioned circadian hunger rhythms (the Isherwood 2023 mechanism), consistent meal timing may also help. Moving eating windows earlier and maintaining consistency allows the circadian glucose-hunger rhythm to re-entrain to a pattern that does not include 3am food-seeking.
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep — the full metabolic cause overview
- Can a Blood Sugar Drop Wake You Up at 3am? — how nocturnal blood sugar drops trigger the hormonal cascade that wakes you up
- Why Does Cortisol Spike at 3am and Wake You Up? — the cortisol-sleep feedback loop and HPA axis timing
- Can a Calorie Deficit Cause Insomnia? — how energy restriction elevates cortisol and fragments sleep
- Is Waking Up at 3am a Sign of Adrenal Fatigue? — the real HPA axis mechanism behind what people call adrenal fatigue
- Does Insulin Resistance Affect Sleep Quality? — the bidirectional relationship between insulin sensitivity and deep sleep
- Can Prediabetes Cause Sleep Problems? — how prediabetes changes sleep architecture and accelerates metabolic decline
References
1. Spiegel K, Tasali E, Penev P, Van Cauter E. Brief communication: Sleep curtailment in healthy young men is associated with decreased leptin levels, elevated ghrelin levels, and increased hunger and appetite. Annals of Internal Medicine. 2004;141(11):846-850. PMID: 15583226
2. Taheri S, Lin L, Austin D, Young T, Mignot E. Short sleep duration is associated with reduced leptin, elevated ghrelin, and increased body mass index. PLoS Medicine. 2004;1(3):e62. PMID: 15602591
3. Broussard JL, Kilkus JM, Delebecque F, et al. Elevated ghrelin predicts food intake during experimental sleep restriction. Obesity. 2016;24(1):132-138. PMID: 26467988
4. van Egmond LT, Meth EMS, Engstrom J, et al. Effects of acute sleep loss on leptin, ghrelin, and adiponectin in adults with healthy weight and obesity: A laboratory study. Obesity. 2023;31(3):635-643. PMID: 36404495
5. Mehyofer S, Chamorro R, Engeli S, Wilms B, Lehnert H, Schmid SM. Late, but not early, night sleep loss compromises neuroendocrine appetite regulation and the desire for food. Nutrients. 2023;15(9):2035. PMID: 37432152
6. Isherwood CM, Mayneris-Perxachs J, Sherwood WB, et al. Human glucose rhythms and subjective hunger anticipate meal timing. Current Biology. 2023;33(7):1321-1331. PMID: 36822203
7. Tajiri E, Yoshimura E, Hatamoto Y, Tanaka H, Shimoda S. Effects of sleep restriction on food intake and appetite under free-living conditions: A randomized crossover trial. Appetite. 2023;189:106968. PMID: 37562755
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
