Testosterone, growth hormone, and DHEA all decline as men age — starting as early as the late 20s and accelerating after 40. These hormonal changes don’t reduce sex drive and muscle mass alone; they directly alter the brain’s ability to produce and maintain deep sleep. Declining testosterone function involves three concurrent changes: falling production from the testes, reduced bioavailability as sex hormone-binding globulin rises, and decreased androgen receptor sensitivity in target tissues including the brain. The result is that men can have “normal” lab values while experiencing measurable sleep disruption.
- Testosterone production depends on sleep — one week of restricted sleep lowers daytime testosterone by 10-15% in healthy young men (Leproult & Van Cauter, 2011)
- Deep sleep drops roughly 80% between young adulthood and midlife in men, and growth hormone secretion falls in parallel (Van Cauter et al., 2000)
- Rising cortisol suppresses both testosterone production and sleep continuity in men
- Visceral fat accelerates the decline by converting testosterone to estradiol via the enzyme aromatase
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 35 references cited
What Are the Hormonal Changes in Aging Men?
A 1997 paper in Science named three parallel hormonal declines that define male aging: andropause (testosterone), somatopause (growth hormone), and adrenopause (DHEA) (Lamberts et al., 1997). Each involves a different gland and a different downstream effect, but they share a timeline — all three accelerate between the late 30s and 60s, and their combined impact is greater than any one decline alone.
Testosterone has the broadest evidence base of the three. Production from the testes drops roughly 1-2% per year starting in the late 20s (Harada, 2018). But testosterone function is not about production alone. Sex hormone-binding globulin — the protein that binds testosterone in the bloodstream — rises with age, reducing the fraction of free, bioavailable testosterone that reaches tissues. And androgen receptor sensitivity itself decreases, meaning tissues respond less effectively to the testosterone that does arrive. A study of 882 men from the Massachusetts Male Aging Study found that androgen receptor gene variants predicted the rate of age-related testosterone decline — men with more active androgen receptors experienced steeper declines, suggesting the receptors themselves are part of the aging equation (Krithivas et al., 1999). The practical consequence: a man can have total testosterone within the “normal” lab range while his functional testosterone — what his brain and body can use — is low (Harada, 2018).
Growth hormone secretion declines through midlife in parallel with a separate process described in Section 2.
DHEA (dehydroepiandrosterone) is the highest-concentration steroid hormone in the body and serves as a raw material for testosterone production. Adrenal DHEA output drops to 10-20% of its young-adult peak by age 70-80. Because DHEA feeds testosterone synthesis, its decline compounds the andropause.
The health effects of these combined declines in men are broad: increased cardiovascular risk, metabolic syndrome, reduced lean muscle mass, increased visceral fat accumulation, cognitive changes, and mood decline. A 2025 review examined hormonal milestones in relation to insomnia prevalence across life stages (Baldi et al., 2025).
But hormonal decline in men doesn’t only affect metabolism, muscle, and mood. It also directly disrupts sleep.
How Do Hormonal Changes in Men Disrupt Sleep?
Five mechanisms connect hormonal changes to sleep disruption in men. The first — declining testosterone function — acts at multiple levels and has the strongest evidence base.
How Does Declining Testosterone Function Disrupt Sleep in Men?
Production decline. Testosterone peaks during REM sleep onset in men, and the majority of daily testosterone production occurs during sleep rather than during waking hours (Andersen & Tufik, 2008). This makes sleep architecture a direct input to testosterone output — not just a consequence of it.
The sensitivity of this relationship is measurable. In a controlled study of young healthy men, one week of sleep restricted to five hours per night reduced daytime testosterone levels by 10-15% (Leproult & Van Cauter, 2011). At a natural decline rate of 1-2% per year, that represents roughly 5 to 15 years of normal aging compressed into seven days.
A 2024 review documented the bidirectional nature of this relationship in men: sleep deprivation, circadian disruption, and obstructive sleep apnea can each independently cause hypogonadism, while low testosterone itself worsens sleep quality — creating a feedback loop where each side amplifies the other (Khripun & Beliaeva, 2024). Men with lower testosterone show lower sleep efficiency, more nocturnal awakenings, and reduced time in both slow-wave and REM stages.
Bioavailability decline. Even when total testosterone production remains within laboratory reference ranges, the amount of testosterone that can reach tissues changes with age. Sex hormone-binding globulin rises as men age, binding more circulating testosterone and reducing the free fraction available to the brain and other target organs. SHBG is a key determinant of bioactive testosterone levels in men — and standard testosterone tests measure total testosterone, which includes the bound fraction that tissues cannot use (Harada, 2018).
A man with total testosterone of 450 ng/dL and elevated SHBG may have less bioavailable testosterone than a man with total testosterone of 350 ng/dL and low SHBG. This is one reason standard lab ranges often miss hormonal sleep disruption — the number looks adequate while the functional reality is not.
Receptor sensitivity decline. The third component is the androgen receptor itself. In the Massachusetts Male Aging Study (882 men), androgen receptor CAG repeat length — a genetic variant that determines how responsive the receptor is to testosterone — predicted the rate of age-related testosterone decline. Men with shorter CAG repeats (more receptor activity) experienced steeper declines over time (Krithivas et al., 1999). In a separate study of 3,369 men, those with longer CAG repeats (less responsive receptors) compensated by producing more testosterone, but the residual effects of weaker receptor response were mediated through estrogen via aromatization — converting testosterone to estradiol rather than using it directly (Huhtaniemi et al., 2009).
The combined importance of all three components — production, bioavailability, and receptor function — was underscored by a small randomized controlled trial (62 men) in which testosterone replacement gel raised serum testosterone from 223 to 775 ng/dL in men with opioid-induced androgen deficiency, yet insomnia severity did not improve compared to placebo over 14 weeks (Huang et al., 2017). This was a specific population — men on chronic opioids — but the finding illustrates the broader principle. Restoring serum levels alone, without addressing receptor function, cortisol balance, or growth hormone, may be insufficient to normalize sleep.
How Does Cortisol Disrupt Testosterone Function, and What Does That Mean for Sleep in Men?
Cortisol and testosterone in men are mechanistically coupled through shared hypothalamic pathways. Sleep restriction simultaneously lowers 24-hour testosterone and raises afternoon cortisol, imbalancing the two primary anabolic and catabolic hormones in men’s physiology. A proof-of-concept study using a dual-hormone clamp — artificially fixing both testosterone and cortisol levels — demonstrated that this mitigated the insulin resistance normally caused by sleep restriction, showing that the testosterone-cortisol imbalance mediates metabolic harm from sleep loss (Liu & Reddy, 2022).
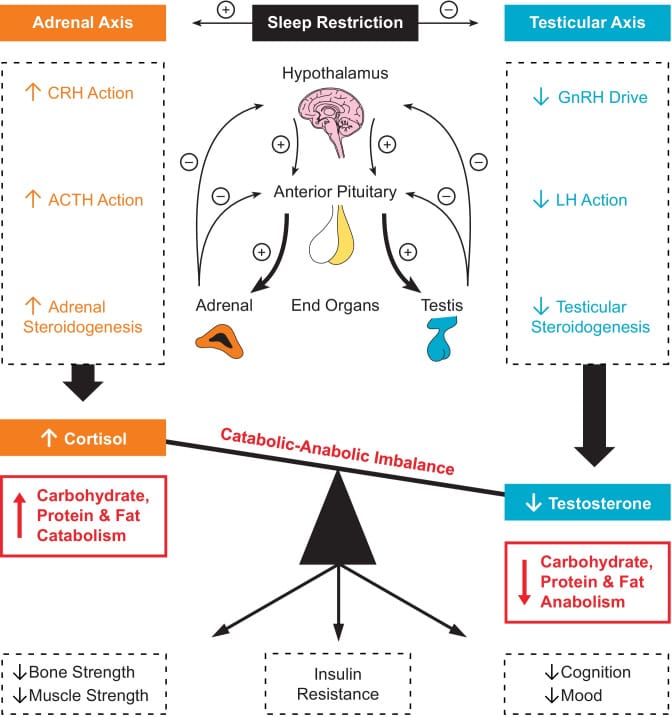
The protective value of prior sleep is limited. A randomized crossover study found that after 24 hours of total wakefulness, both testosterone and cortisol fell compared to morning baseline — and six nights of extended sleep beforehand did not protect against this decline (Arnal et al., 2016). This acute response differs from chronic sleep restriction, where cortisol tends to rise; the shared finding is that prior sleep extension does not build hormonal reserves that protect men during subsequent sleep loss.
Cortisol elevation during sleep deprivation also predicts how well men recover afterward. In 32 healthy adults aged 27-53, each 1 microgram per deciliter increase in morning cortisol during sleep deprivation predicted 87 fewer minutes of recovery sleep duration. Cortisol and cognitive measures together explained 35-41% of the variance in recovery sleep timing and duration (Goel & Pasetes, 2025). The cortisol level at the time of deprivation shapes the subsequent sleep the body can produce — higher cortisol during the deficit predicts a shorter, less complete recovery.
What Is Growth Hormone, How Does It Change in Men After Midlife, and How Does That Affect Sleep?
Growth hormone in men does its primary work during sleep. It drives muscle protein synthesis, bone density maintenance, fat metabolism, and tissue repair — processes that depend on the concentrated bursts of growth hormone released during deep sleep episodes. Approximately 70% of the total daily growth hormone output in men occurs during the first slow-wave sleep episode of the night, and this relationship is causal: pharmacological stimulation of slow-wave sleep increases growth hormone secretion (Van Cauter et al., 1998).
The age-related decline is among the steepest of any hormone in men. In a study of 149 healthy men aged 16 to 83, slow-wave sleep dropped from 18.9% of total sleep time at ages 16-25 to 3.4% at ages 36-50 — a decline of roughly 80%. Growth hormone secretion fell in parallel, declining by 372 micrograms per decade. The association between slow-wave sleep and growth hormone was statistically independent of age itself, meaning the sleep decline was driving the hormonal decline, not aging independently (Van Cauter et al., 2000). The same study found that evening cortisol rose by 19.3 nanomoles per liter per decade after age 50 — connecting growth hormone decline to the cortisol pathway described above.
The mechanism behind this age-related change involves two competing peptides in the hypothalamus. Growth hormone-releasing hormone promotes both slow-wave sleep and growth hormone secretion. Corticotropin-releasing hormone — which drives the cortisol pathway — opposes both. As men age, the balance between these two peptides tilts toward corticotropin-releasing hormone, producing shallower sleep and blunted growth hormone output simultaneously (Steiger, 2007). This is not two separate declines happening in parallel — it is a single regulatory change that degrades both sleep depth and hormonal output through the same pathway.
How Does DHEA Change with Aging in Men, How Does It Affect Testosterone Function, and What Does That Mean for Sleep?
DHEA contributes to testosterone function in men through direct conversion. The adrenal glands produce DHEA, which is converted to testosterone via androstenedione — making it a raw material for testosterone synthesis that works independently of testicular production. When DHEA declines with age, men lose this supplementary source of testosterone, compounding the direct testicular decline.
The magnitude of this contribution is measurable. In a study of individualized DHEA replacement in men (doses of 15-100 mg per day calibrated to restore physiological DHEA-sulfate levels), free testosterone rose from 10.9 to 14.7 picograms per milliliter — a 35% increase in bioavailable testosterone from DHEA supplementation alone (Rommler, 2003).
DHEA also has a direct relationship with sleep architecture. Administration of DHEA to healthy young men increased REM sleep duration — an effect the authors attributed to GABA-related mechanisms rather than sex steroid conversion (Friess et al., 1995). The implication: DHEA may support sleep through neurosteroid pathways that include but are not limited to its role as a testosterone precursor.
Beyond its role as a precursor, the ratio between cortisol and DHEA-sulfate has emerged as a marker for tracking sleep disruption. In a pilot study of rotating-schedule healthcare professionals, a combined model including reduced cortisol and increased DHEA-sulfate explained 69.8% of the variance in sleep disturbance scores over six months (Salahuddin et al., 2025). In this occupational population, declining cortisol paired with rising DHEA-sulfate tracked worsening sleep — the opposite direction from the chronic stress pattern described above, where cortisol rises. The shared principle is that the balance between these hormones, not either hormone’s absolute level, appears to track sleep disruption.
DHEA and testosterone also have distinct effects on immune function. While testosterone tends to suppress inflammatory responses, DHEA promotes them — meaning their simultaneous decline during aging produces a different immunological outcome than either decline alone (Buendia-Gonzalez & Legorreta-Herrera, 2022). This distinction is relevant because immune activation and inflammation are independent contributors to sleep disruption.
The co-occurrence of adrenopause (DHEA), andropause (testosterone), and somatopause (growth hormone) means that men experience a compound hormonal decline affecting sleep architecture through multiple independent pathways simultaneously (Lamberts et al., 1997).
How Does Visceral Fat Disrupt Testosterone Function and What Does That Mean for Sleep in Men?
Visceral fat in men is not a passive storage tissue. When researchers induced testosterone deficiency in miniature pigs, visceral fat showed more than 700 differentially expressed genes — predominantly in inflammation and lipid metabolism pathways. Testosterone replacement reversed the majority of these metabolic changes, demonstrating that visceral fat actively reshapes its molecular profile in response to testosterone status (Zhang et al., 2016).
The practical consequence for sleep is mediated through two pathways. First, aromatase activity in visceral fat converts testosterone to estradiol, reducing the testosterone available for sleep-regulatory functions in the brain while elevating estradiol levels. Second, visceral fat accumulation increases the risk of obstructive sleep apnea — and obstructive sleep apnea itself suppresses testosterone production in men independently of age or body weight.
The obstructive sleep apnea risk from visceral fat is large and well-documented. In 8,284 participants from the National Health and Nutrition Examination Survey, a visceral fat score in the highest quartile carried an odds ratio of 2.436 for obstructive sleep apnea compared to the lowest quartile. The prevalence of obstructive sleep apnea rose from 32.8% in the lowest visceral fat group to 62.1% in the highest (Xu et al., 2024).
The overlap between visceral fat and testosterone suppression was documented in an analysis of NHANES 2011-2016 data (8,748 adults), which noted that prior research on the 2015-2016 subsample found a high frequency of snoring (five or more nights per week) was associated with lower testosterone concentrations in men aged 40-59 with a waist circumference of 102 centimeters or greater (Hernandez-Perez et al., 2024).
The resulting cycle in men is self-reinforcing: declining testosterone promotes visceral fat accumulation, visceral fat increases aromatase conversion of testosterone to estradiol, lower functional testosterone further degrades sleep architecture, and degraded sleep further suppresses testosterone production.
How Does Low Testosterone Increase Stress Reactivity, and What Does That Mean for Sleep in Men?
The mechanism: testosterone as a cortisol regulator. Testosterone suppresses cortisol output through at least three documented pathways in men.
First, testosterone directly reduces the cortisol response to corticotropin-releasing hormone — the hypothalamic peptide that initiates the stress cascade. In a controlled study using leuprolide to suppress endogenous hormones followed by testosterone add-back, testosterone suppressed CRH-stimulated cortisol output in men (Rubinow et al., 2005). The study design — suppression of all gonadal hormones followed by selective replacement — isolated testosterone as the active agent.
Second, testosterone attenuates adrenal sensitivity to ACTH, the pituitary hormone that instructs the adrenal glands to release cortisol. A longitudinal study of transgender men receiving testosterone therapy found a 58% reduction in cortisol output and a 15% reduction in ACTH output after three months. The change in the ACTH-to-cortisol ratio indicated that testosterone was dampening the adrenal gland’s responsiveness to pituitary instructions, not just reducing the pituitary instructions themselves (Fuss et al., 2019). Conversely, individuals receiving estrogen and antiandrogen therapy showed cortisol increases of 15% and ACTH increases of 18% — the mirror image, demonstrating that testosterone removal amplifies the stress response.
Third, testosterone modulates glucocorticoid receptor expression in the brain. In a rat model, dihydrotestosterone (the active metabolite of testosterone) reduced glucocorticoid receptor mRNA in the hippocampal CA1 region — a brain area rich in androgen receptors and central to stress regulation — to 69% of intact levels. This effect was region-specific: it occurred only in CA1 where androgen receptors are concentrated, and did not affect mineralocorticoid receptors (Kerr et al., 1996). While this was demonstrated in an animal model, the hippocampal CA1 region expresses androgen receptors in humans as well, and the finding is consistent with the human evidence from Rubinow and Fuss above. Fewer glucocorticoid receptors in the hippocampus means the brain’s stress-response feedback loop is less sensitive to cortisol — effectively raising the threshold at which cortisol triggers further stress activation. When testosterone declines, glucocorticoid receptor density may rise, lowering that threshold and making the brain more reactive to the same cortisol levels.
The consequence: low testosterone amplifies every stressor. When testosterone declines in aging men, all three dampening mechanisms weaken simultaneously. The adrenal glands become more responsive to pituitary instructions, producing more cortisol per unit of ACTH (as demonstrated by the Fuss et al. data). The brain’s hippocampal feedback loop may become more sensitive to cortisol as glucocorticoid receptor density rises (as suggested by the Kerr et al. animal data). And testosterone’s direct suppression of the CRH-stimulated stress response weakens (Rubinow et al.).
The practical result is measurable: a man with declining testosterone produces a larger cortisol spike from the same stressor — a work deadline, a financial worry, a poor stretch of sleep — than he would have produced a decade earlier. The cortisol spike lasts longer because the feedback mechanisms that would normally shut it down are themselves testosterone-dependent.
This amplification has a bidirectional partner. In animal models, elevated glucocorticoids directly suppress testosterone production by inhibiting Leydig cell function in the testes (Witorsch, 2016). So low testosterone amplifies cortisol reactivity, and amplified cortisol further suppresses testosterone — each side accelerating the other.
What this means for sleep in men. Amplified stress reactivity translates directly to fragmented sleep through two pathways. First, elevated evening and nocturnal cortisol — driven by a stress response that men with low testosterone cannot adequately suppress — disrupts the transition into and maintenance of deep sleep. Second, each episode of sleep fragmentation itself becomes a stressor that triggers further cortisol release, which suppresses the pulsatile testosterone secretion that normally occurs during the first hours of sleep.
The Fuss et al. (2019) finding is particularly relevant here: a 58% reduction in cortisol output from testosterone therapy represents a large change in how much cortisol the body produces in response to stressors. The Fuss study measured the effect of a full hormonal transition, not the partial decline of aging — but the direction and magnitude suggest that even a partial testosterone decline in aging men could meaningfully increase cortisol reactivity, with each nocturnal arousal producing a larger cortisol response than it would have a decade earlier.
Research in pain populations adds a further dimension: in a study of 33 patients with temporomandibular disorder, greater sleep disturbance amplified cortisol reactivity to acute pain — though not to psychological stress — while longer sleep duration attenuated cortisol reactivity to both (Altier et al., 2024). This suggests that once the testosterone-cortisol imbalance begins fragmenting sleep, the fragmented sleep can make cortisol reactivity to physical stressors worse, which fragments sleep further — adding a third loop on top of the testosterone-cortisol and sleep-cortisol cycles.
Why this matters for men specifically. The organizational effects of testosterone on the HPA axis appear to be partly sex-specific. Even when researchers induced equivalent gonadal suppression in men and women — removing the influence of current testosterone levels — men still showed greater ACTH and cortisol responses to CRH and exercise stress than women (Roca et al., 2005). This suggests that men’s HPA axis is calibrated differently from women’s at a developmental level, and that testosterone’s regulatory role may be more consequential in men because the baseline HPA reactivity it needs to dampen is higher.
What Triggers or Worsens Hormonal Sleep Disruption in Men?
Does Chronic Sleep Restriction Lower Testosterone in Men?
The Leproult and Van Cauter (2011) finding that one week of restricted sleep lowers testosterone by 10-15% in young healthy men established chronic sleep restriction as an independent trigger of hormonal decline — not a secondary consequence. Men who habitually restrict sleep are actively accelerating their testosterone decline on top of the age-related trajectory.
The bidirectional nature of this relationship makes it self-reinforcing. Once testosterone drops below a threshold where sleep quality is affected, the resulting sleep disruption lowers testosterone further, which degrades sleep architecture further. Without a change on either side of the cycle, the trajectory is downward.
How Do Diet and Body Composition Affect Testosterone Function and Sleep in Men?
A 2026 review in Reviews in Endocrine and Metabolic Disorders examined the molecular mechanisms by which chronic sleep deprivation and ultra-processed food intake interact to suppress testosterone in men. The review identified these as a dual-hit on the hypothalamic-pituitary-gonadal axis — sleep deprivation impairs the central hormonal instruction while dietary factors impair the metabolic environment that testosterone production depends on (Schimenes et al., 2026).
Visceral fat functions as the amplifier. Aromatase in visceral fat tissue converts testosterone to estradiol, and men aged 40-59 with frequent snoring and elevated waist circumference showed lower testosterone concentrations (Hernandez-Perez et al., 2024).
Does Chronic Stress Accelerate Hormonal Sleep Decline in Men?
The hypothalamic-pituitary-adrenal axis and the hypothalamic-pituitary-gonadal axis share regulatory machinery at the hypothalamic level. Sustained activation of one suppresses the other. In men under chronic stress, this means elevated cortisol output paired with suppressed gonadotropin-releasing hormone — the upstream instruction for testosterone production.
The prevalence of this pattern is not rare. In a study of 3,966 individuals, 20.6% showed flattened diurnal cortisol slopes — a marker of chronic hypothalamic-pituitary-adrenal dysregulation (Kreitzberg et al., 2022). A flattened cortisol slope means that evening cortisol remains elevated when it should be declining, which both fragments sleep directly and suppresses the pulsatile testosterone release that normally occurs during the first hours of sleep.
How Do You Know If Hormonal Changes Are Disrupting Your Sleep as a Man?
The sleep pattern associated with hormonal disruption in men has several recognizable features. Falling asleep is often not the problem — the disruption appears in the second half of the night, when cortisol begins its pre-dawn rise and testosterone production, which depends on sustained sleep architecture, is already declining. Reduced dream recall can indicate diminished REM sleep. Waking feeling unrested despite seven or more hours in bed can indicate reduced slow-wave sleep depth.
These sleep changes often co-occur with other signs of hormonal decline in men: increased abdominal fat, reduced muscle mass or strength, lower libido, mood changes, and slower recovery from exercise. The co-occurrence of sleep disruption with these physical changes does not prove hormonal causation, but it raises the probability that hormonal pathways are involved.
Biomarkers men can investigate: Standard testosterone testing measures total testosterone, which includes the fraction bound to SHBG that tissues cannot use. Testing total testosterone, free testosterone, and SHBG together gives a more complete picture of actual testosterone function. Additional markers include morning and evening cortisol (to assess diurnal pattern and detect flattened slopes), DHEA-sulfate (to assess adrenopause), and IGF-1 (as a proxy for growth hormone status). The cortisol-to-testosterone ratio is emerging as a composite biomarker that may capture the interaction between these axes better than either hormone measured alone (Liu & Reddy, 2022).
Many men experiencing hormonal sleep disruption have two or three causes working together — hormonal disruption often coexists with autonomic, inflammatory, or metabolic causes. Identifying the hormonal component is one step in understanding the full picture.
Find out which causes are driving your 3am wakeups: Take the 3AM Decoder
Frequently Asked Questions
Can Hormonal Sleep Disruption in Men Be Reversed?
The evidence that modifiable factors drive a measurable portion of hormonal sleep disruption comes from controlled studies. Testosterone replacement reversed the majority of metabolic changes in visceral fat tissue that were caused by testosterone deficiency (Zhang et al., 2016). A dual-hormone clamp that corrected the testosterone-cortisol imbalance mitigated metabolic harm from sleep restriction (Liu & Reddy, 2022). These findings indicate that the hormonal pathways involved in sleep disruption respond to change — the decline is not a fixed trajectory.
The entry point with the broadest downstream effect is sleep itself. Sleep fragmentation directly suppresses next-morning testosterone secretion. Improving sleep continuity and architecture can restore more favorable hormone patterns within one to two weeks. The relationship is bidirectional — either side can be the entry point for improvement.
Does Testosterone Replacement Therapy Fix Sleep Problems in Men?
A 14-week randomized controlled trial raised serum testosterone from 223 to 775 ng/dL in men with opioid-induced androgen deficiency, yet insomnia severity scores did not improve compared to placebo (Huang et al., 2017). A review of testosterone therapy in aging men found that high-dose testosterone can induce or worsen obstructive sleep apnea, while improving cardiometabolic function and sexual health — creating a trade-off that remains unresolved (Liu & Reddy, 2022).
These findings are consistent with the three-component view of testosterone function: raising serum levels addresses production but does not change SHBG binding or androgen receptor sensitivity. It also does not address the cortisol pathway, the growth hormone decline, or the DHEA deficit that contribute independently to sleep disruption in men.
Can Men in Their 30s Experience Hormonal Sleep Disruption?
The 1-2% per year decline rate means a man at 35 has already experienced 5-10% cumulative testosterone decline from his peak (Harada, 2018). For men with additional risk factors — elevated visceral fat, chronic work stress, habitual sleep restriction — the functional decline can be steeper. The Leproult and Van Cauter (2011) study demonstrated measurable testosterone suppression from sleep restriction in young healthy men, showing that the bidirectional relationship between sleep and testosterone is active in men decades before midlife.
Why Do Standard Lab Tests Sometimes Miss Hormonal Sleep Problems in Men?
SHBG is a key variable that standard testing often omits. As SHBG rises with age, it binds more circulating testosterone, reducing the free fraction that reaches the brain and other tissues (Harada, 2018). Two men with identical total testosterone can have different free testosterone levels depending on their SHBG concentration.
Androgen receptor sensitivity adds a further dimension that no blood test can capture directly. Genetic variation in the androgen receptor CAG repeat length influences how effectively tissues respond to the free testosterone that does reach them (Krithivas et al., 1999). This means that even free testosterone measurement, while more informative than total testosterone alone, does not fully capture functional testosterone status.
How Is Hormonal Sleep Disruption Different from Insomnia in Men?
The distinction matters for men because the presentation can look identical. A man waking at 3am and unable to return to sleep may be experiencing cortisol-mediated arousal, testosterone-dependent sleep architecture degradation, or both — none of which respond to behavioral sleep approaches alone. A 2025 review of gender-specific insomnia patterns found that declining testosterone may contribute to increased insomnia prevalence in older men as a biological factor, not just a correlate (Baldi et al., 2025). And cortisol levels during sleep deprivation predict how well the body can recover sleep afterward — each 1 microgram per deciliter increase in cortisol during a deficit predicted 87 fewer minutes of recovery sleep (Goel & Pasetes, 2025).
Related Reading
- Can Low GABA Cause Waking Up at 3am?
- Does Low Testosterone Cause Sleep Problems in Men?
- Can Belly Fat Lower Your Testosterone and Disrupt Your Sleep?
- Does Testosterone Replacement Therapy Affect Sleep?
- Autonomic Sleep Disruption
- Circadian Sleep Disruption
- Hormonal Sleep Disruption in Women
- Inflammatory Sleep Disruption
- Metabolic Sleep Disruption
References
Axis 1: Testosterone Function and Sleep in Men
- Andersen, M. L., & Tufik, S. (2008). The effects of testosterone on sleep and sleep-disordered breathing in men: Its bidirectional interaction with erectile function. Sleep Medicine Reviews, 12(5), 365-379. https://pubmed.ncbi.nlm.nih.gov/18519168/
- Leproult, R., & Van Cauter, E. (2011). Effect of 1 week of sleep restriction on testosterone levels in young healthy men. JAMA, 305(21), 2173-2174. https://pubmed.ncbi.nlm.nih.gov/21632481/
- Khripun, I. A., & Beliaeva, E. V. (2024). Sleep disorders and testosterone deficiency in men. Vestnik Urologii, 12(6), 52-58. https://doi.org/10.21886/2308-6424-2024-12-6-52-58
- Harada, N. (2018). Role of androgens in energy metabolism affecting on body composition, metabolic syndrome, type 2 diabetes, cardiovascular disease, and longevity: Lessons from a meta-analysis and rodent studies. Bioscience, Biotechnology, and Biochemistry, 82(10), 1667-1682. https://pubmed.ncbi.nlm.nih.gov/29957125/
- Krithivas, K., Yurgalevitch, S. M., Mohr, B. A., Wilcox, C. J., Batter, S. J., Brown, M., Longcope, C., McKinlay, J. B., & Kantoff, P. W. (1999). Evidence that the CAG repeat in the androgen receptor gene is associated with the age-related decline in serum androgen levels in men. Journal of Endocrinology, 162(1), 137-142. https://pubmed.ncbi.nlm.nih.gov/10396030/
- Huhtaniemi, I. T., Pye, S. R., Limer, K. L., Thomson, W., O’Neill, T. W., Platt, H., Payne, D., John, S. L., Jiang, M., Boonen, S., Borghs, H., Vanderschueren, D., Adams, J. E., Ward, K. A., Bartfai, G., Casanueva, F., Finn, J. D., Forti, G., Giwercman, A., … Wu, F. C. W. (2009). Increased estrogen rather than decreased androgen action is associated with longer androgen receptor CAG repeats. Journal of Clinical Endocrinology & Metabolism, 94(1), 277-284. https://pubmed.ncbi.nlm.nih.gov/18840639/
- Huang, G., Travison, T. G., Edwards, R. R., & Basaria, S. (2017). Effects of testosterone replacement on pain catastrophizing and sleep quality in men with opioid-induced androgen deficiency. Pain Medicine, 18(6), 1070-1076. https://pubmed.ncbi.nlm.nih.gov/27550959/
- Liu, C. C., Lee, Y. C., Wang, C. J., Yeh, H. C., Li, W. M., Wu, W. J., Huang, C. N., Bao, B. Y., Huang, C. H., & Huang, S. P. (2012). The impact of androgen receptor CAG repeat polymorphism on andropausal symptoms in different serum testosterone levels. Journal of Sexual Medicine, 9(9), 2429-2437. https://pubmed.ncbi.nlm.nih.gov/22429282/
Axis 2: Cortisol-Testosterone Interaction and Sleep in Men
- Liu, P. Y., & Reddy, R. T. (2022). Sleep, testosterone and cortisol balance, and ageing men. Reviews in Endocrine and Metabolic Disorders, 23(6), 1323-1339. https://pubmed.ncbi.nlm.nih.gov/36152143/
- Arnal, P. J., Drogou, C., Sauvet, F., Regnauld, J., Dispersyn, G., Faraut, B., Millet, G. Y., Leger, D., Gomez-Merino, D., & Chennaoui, M. (2016). Effect of sleep extension on the subsequent testosterone, cortisol and prolactin responses to total sleep deprivation and recovery. Journal of Neuroendocrinology, 28(2), 12346. https://pubmed.ncbi.nlm.nih.gov/26647769/
- Goel, N., & Pasetes, L. (2025). Cognitive and cortisol measures during sleep deprivation and psychological stress influence recovery sleep metrics. SLEEP, 48(Suppl 1), A111. https://doi.org/10.1093/sleep/zsaf090.0254
Axis 3: Growth Hormone and Sleep in Men
- Van Cauter, E., Leproult, R., & Plat, L. (2000). Age-related changes in slow wave sleep and REM sleep and relationship with growth hormone and cortisol levels in healthy men. JAMA, 284(7), 861-868. https://pubmed.ncbi.nlm.nih.gov/10938176/
- Van Cauter, E., Plat, L., & Copinschi, G. (1998). Interrelations between sleep and the somatotropic axis. Sleep, 21(6), 553-566. https://pubmed.ncbi.nlm.nih.gov/9779515/
- Steiger, A. (2007). Neurochemical regulation of sleep. Journal of Psychiatric Research, 41(7), 537-552. https://pubmed.ncbi.nlm.nih.gov/16777143/
Axis 4: DHEA, Testosterone Function, and Sleep in Men
- Lamberts, S. W. J., van den Beld, A. W., & van der Lely, A. J. (1997). The endocrinology of aging. Science, 278(5337), 419-424. https://pubmed.ncbi.nlm.nih.gov/9334293/
- Rommler, A. (2003). Adrenopause and dehydroepiandrosterone: Pharmacological therapy versus replacement therapy. Gynakologisch-geburtshilfliche Rundschau, 43(2), 79-90. https://pubmed.ncbi.nlm.nih.gov/12649580/
- Friess, E., Trachsel, L., Guldner, J., Schier, T., Steiger, A., & Holsboer, F. (1995). DHEA administration increases rapid eye movement sleep and EEG power in the sigma frequency range. American Journal of Physiology, 268(1 Pt 1), E107-E113. https://pubmed.ncbi.nlm.nih.gov/7840167/
- Salahuddin, M. F., Sukararuji, K., Sharifi, M., Odia, K. A. F., Manzar, M. D., Pandi-Perumal, S. R., & BaHammam, A. S. (2025). Associations between salivary cortisol, DHEA-S, and alpha-amylase and longitudinal sleep disruption in shift-working healthcare professionals: A pilot study. Nature and Science of Sleep, 17, 2611-2623. https://pubmed.ncbi.nlm.nih.gov/41103458/
- Buendia-Gonzalez, F. O., & Legorreta-Herrera, M. (2022). The similarities and differences between the effects of testosterone and DHEA on the innate and adaptive immune response. Biomolecules, 12(12), 1768. https://pubmed.ncbi.nlm.nih.gov/36551196/
Axis 5: Visceral Fat, Testosterone Function, and Sleep in Men
- Zhang, L., Cai, Y., Wei, S., Ling, Y., Zhu, L., Li, D., & Cai, Z. (2016). Testosterone deficiency induces changes of the transcriptomes of visceral adipose tissue in miniature pigs fed a high-fat and high-cholesterol diet. International Journal of Molecular Sciences, 17(12), 2125. https://pubmed.ncbi.nlm.nih.gov/27999286/
- Xu, X., Xu, J., & Zhang, M. (2024). Association between metabolic score for visceral fat and obstructive sleep apnea: A cross-sectional study. Frontiers in Medicine, 11, 1480717. https://pubmed.ncbi.nlm.nih.gov/39726679/
- Hernandez-Perez, J. G., Taha, S., Torres-Sanchez, L. E., Villasante-Tezanos, A., Milani, S. A., Baillargeon, J., Canfield, S., & Lopez, D. S. (2024). Association of sleep duration and quality with serum testosterone concentrations among men and women: NHANES 2011-2016. Andrology, 12(3), 518-526. https://pubmed.ncbi.nlm.nih.gov/37452666/
Axis 6: Testosterone, Stress Reactivity, and Sleep in Men
- Rubinow, D. R., Roca, C. A., Schmidt, P. J., Danaceau, M. A., Putnam, K., Cizza, G., Chrousos, G., & Nieman, L. (2005). Testosterone suppression of CRH-stimulated cortisol in men. Neuropsychopharmacology, 30(10), 1906-1912. https://pubmed.ncbi.nlm.nih.gov/15841103/
- Fuss, J., Claro, L., Ising, M., Biedermann, S. V., Wiedemann, K., Stalla, G. K., Briken, P., & Auer, M. K. (2019). Does sex hormone treatment reverse the sex-dependent stress regulation? A longitudinal study on hypothalamus-pituitary-adrenal (HPA) axis activity in transgender individuals. Psychoneuroendocrinology, 104, 228-237. https://pubmed.ncbi.nlm.nih.gov/30897530/
- Kerr, J. E., Beck, S. G., & Handa, R. J. (1996). Androgens modulate glucocorticoid receptor mRNA, but not mineralocorticoid receptor mRNA levels, in the rat hippocampus. Journal of Neuroendocrinology, 8(6), 439-447. https://pubmed.ncbi.nlm.nih.gov/8809674/
- Witorsch, R. J. (2016). Effects of elevated glucocorticoids on reproduction and development: Relevance to endocrine disruptor screening. Critical Reviews in Toxicology, 46(5), 420-436. https://pubmed.ncbi.nlm.nih.gov/26912073/
- Roca, C. A., Schmidt, P. J., Deuster, P. A., Danaceau, M. A., Altemus, M., Putnam, K., Chrousos, G. P., Nieman, L. K., & Rubinow, D. R. (2005). Sex-related differences in stimulated hypothalamic-pituitary-adrenal axis during induced gonadal suppression. Journal of Clinical Endocrinology & Metabolism, 90(7), 4224-4231. https://pubmed.ncbi.nlm.nih.gov/15886244/
- Altier, H., et al. (2024). Cortisol reactivity to experimental pain and psychological stress in facial pain: Moderating effects of sleep. SLEEP, 47(Suppl 1), A390. https://doi.org/10.1093/sleep/zsae067.0909
Section 3: Triggers
- Schimenes, B. C., Alvarenga, T. A., Toricelli, M., Moyses-Oliveira, M., Vasco, M. B., Tufik, S., & Andersen, M. L. (2026). The 24/7 lifestyle and male hormonal health: How sleep deprivation and ultra-processed foods impact testosterone and reproduction. Reviews in Endocrine and Metabolic Disorders. Advance online publication. https://pubmed.ncbi.nlm.nih.gov/41862783/
Section 4: Recognition
- Kreitzberg, D., et al. (2022). At-home sample collection for identification of alterations in HPA-axis activity and cortisol AUC. SLEEP, 45(Suppl 1), A170. https://doi.org/10.1093/sleep/zsac079.376
- Jiao, Y., Butoyi, C., Zhang, Q., Adotey, S. A. A. I., Chen, M., Shen, W., Wang, D., Yuan, G., & Jia, J. (2025). Sleep disorders impact hormonal regulation: Unravelling the relationship among sleep disorders, hormones and metabolic diseases. Diabetology & Metabolic Syndrome, 17, 305. https://pmc.ncbi.nlm.nih.gov/articles/PMC12315459/
Additional Background References
- Barrett-Connor, E., Dam, T. T., Stone, K., Harrison, S. L., Redline, S., & Orwoll, E. (2008). The association of testosterone levels with overall sleep quality, sleep architecture, and sleep-disordered breathing. Journal of Clinical Endocrinology and Metabolism, 93(7), 2602-2609. https://pubmed.ncbi.nlm.nih.gov/18413429/
- Piovezan, R. D., Abucham, J., Dos Santos, R. V. T., Mello, M. T., Tufik, S., & Poyares, D. (2015). The impact of sleep on age-related sarcopenia: Possible connections and clinical implications. Ageing Research Reviews, 23(Pt B), 210-220. https://pubmed.ncbi.nlm.nih.gov/26216211/
- Steiger, A. (2003). Sleep and endocrinology. Journal of Internal Medicine, 254(1), 13-22. https://pubmed.ncbi.nlm.nih.gov/12823639/
- Baldi, E., Cerolini, S., Meneo, D., Baglioni, C., & Palagini, L. (2025). Insomnia disorder: Gender issues over the lifespan. Journal of Sleep Research, 34(5), e70110. https://pubmed.ncbi.nlm.nih.gov/40461101/
