Overview: Autonomic sleep disruption occurs when the body’s involuntary regulation — heart rate, stress hormones, and neural inhibition — fails to transition properly into sleep mode. Three mechanisms can drive it:
- Weakened vagal tone — parasympathetic activation doesn’t engage at sleep onset, leaving heart rate elevated through the night
- Reduced GABA — the brain’s primary inhibitory neurotransmitter is insufficient, preventing entry into deep sleep
- Overactive HPA axis — cortisol surges too early, producing the characteristic 2-3am wakeup with a racing mind
Chronic stress, neuroinflammation, gut microbiome disruption, and hormonal changes can each weaken these pathways. The hallmark experience is waking up alert — not groggy — often with a racing heart or the “wired but tired” feeling. Heart rate variability during sleep and cortisol patterns are measurable indicators.
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 26 references cited
What Is Autonomic Dysregulation?
Your autonomic nervous system runs in the background. It manages the functions you don’t consciously control — your heart rate, your breathing rhythm, your stress response, and the physiological transition into sleep.
It has two branches.
- The sympathetic branch drives arousal: it raises heart rate, sharpens alertness, and mobilizes energy.
- The parasympathetic branch drives recovery: it slows heart rate, deepens breathing, and supports the transition into sleep.
Healthy sleep depends on the parasympathetic branch taking over as the night begins. Heart rate drops. Breathing slows. Core body temperature falls. The brain moves from high-frequency waking activity into the slower rhythms of deep sleep.
When that handoff doesn’t happen cleanly — when sympathetic tone remains elevated or parasympathetic activation is insufficient — sleep fragments. You may fall asleep but wake too early. You may lie awake with a racing heart despite being exhausted. The body is stuck between two competing states: one trying to sleep, the other still in sympathetic activation — elevated heart rate, heightened alertness, increased cortisol output.
Several mechanisms contribute to autonomic sleep disruption: weakened vagal tone, reduced GABAergic inhibition, and an overactive stress-hormone axis.
Here are 3 common examples:
How Does Autonomic Dysregulation Disrupt Sleep?
How Does Vagal Tone Affect Sleep?
The vagus nerve is the main conduit of parasympathetic control. It runs from the brainstem to the heart, lungs, and gut, and its activity — measured indirectly through heart rate variability — reflects how effectively the body can transition from alertness to rest.
During healthy sleep, vagal activity increases. Heart rate drops. The body enters a measurably different physiological state. A 2024 study in Stress and Health (Joubert et al.) found that individuals who showed minimal increases in high-frequency heart rate variability at sleep onset — consistent with reduced parasympathetic activation at sleep onset — reported worse sleep quality and greater difficulty with stress the following day.
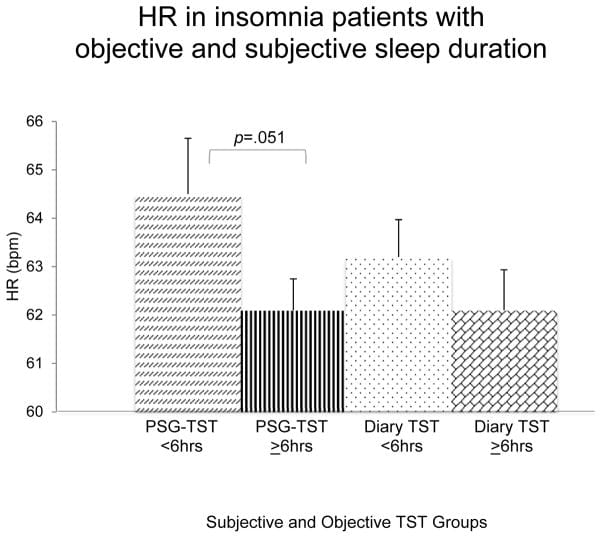
When vagal tone is low, this transition is incomplete. The body enters sleep with sympathetic activity still elevated — meaning heart rate, blood pressure, and arousal-related neural firing remain higher than they should be during rest. A 2020 study using overnight pulse wave analysis (Laharnar et al., PLoS ONE) found that people with insomnia had higher median overnight heart rate of 66-67 bpm throughout the night, compared to 58 bpm in healthy sleepers. The elevated rate was present as an overnight average, not isolated to moments of waking.
A 2025 international expert consensus in Nature Reviews Cardiology (Menuet et al.) argued that respiratory-related HRV is primarily vagally mediated and should be interpreted in relation to breathing. Consumer wearables may help track nighttime HRV trends as a useful indicator, though this observation is not from the Menuet paper.
Medications can also weaken vagal and cholinergic activity. Anticholinergic drugs — a class that includes certain antihistamines, bladder medications, and some antidepressants — work against the same cholinergic pathways the vagus nerve uses. Their effect on sleep is covered in the triggers section below.
What Role Does GABA Play in Sleep Disruption?
Several major classes of sleep medication — from barbiturates to benzodiazepines to newer non-benzodiazepine hypnotics — act through GABA receptors. That convergence is not coincidental. GABA is a key mechanism through which the brain reduces its own activity to allow sleep.
A 2024 review in Tzu Chi Medical Journal (Varinthra et al.) mapped how GABAergic impairment directly produces insomnia through multiple receptor subtypes. A companion review in Heliyon (Zhu et al., 2024) identified five GABA-related mechanisms involved in insomnia: GABA-A receptor modulation, GABA-B receptor modulation, neuroinflammation, oxidative damage, and circadian melatonin regulation.
In a study published in Sleep (Winkelman et al., 2008), people with primary insomnia had approximately 30% lower whole-brain GABA levels than healthy sleepers, measured using magnetic resonance spectroscopy. The lower the GABA, the more time spent awake after sleep onset.
GABAergic tone is not fixed.
It is vulnerable to multiple disruptors — neuroinflammation, gut microbiome changes, and hormonal changes among them. A 2025 study in Frontiers in Bioscience (Jiang et al.) reported that a GABA-producing bacterial strain altered sleep-related behavior through a microbiota-gut-brain pathway and supports a possible gut-GABA-sleep link. A 2023 study in Nature (Sun et al.) resolved native GABA-A receptor structures and found that allopregnanolone — a neurosteroid derived from progesterone — was bound to the receptor even without external administration, supporting an endogenous hormonal role in maintaining GABAergic function.
When GABA is low, the brain cannot calm itself. Neural circuits that should be suppressed during sleep remain active, producing the experience of lying in bed with a mind that won’t stop running.
How Do Cortisol and Adrenaline Wake You Up?
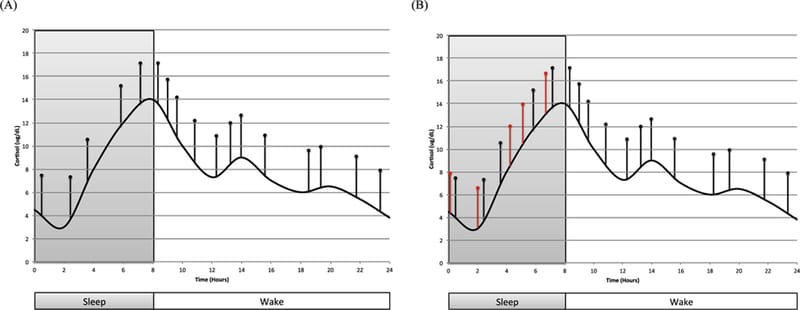
The HPA axis follows a predictable 24-hour pattern.
Cortisol drops after sleep onset, reaches its nadir in the first few hours of the night, then begins a gradual rise that peaks shortly after waking. This cortisol awakening response is tightly coordinated with sleep architecture — deep sleep is associated with lower HPA output, and the rise toward morning helps the brain transition back to wakefulness.
When cortisol stays elevated or the rhythm flattens, the result is measurable: more nighttime awakenings, less deep sleep, and the characteristic early-morning waking that people describe as “my brain turns on at 3am.”
A 2022 meta-analysis in Sleep Medicine Reviews (Dressle et al.) pooled data from 20 case-control studies involving over 800 participants and found that people with chronic insomnia had moderately elevated cortisol levels compared to healthy sleepers (standardized mean difference 0.50). When restricted to blood-sample studies, the effect was larger (0.67). The elevation was not confined to the morning — it reflected a 24-hour pattern of HPA overactivation.
Cortisol doesn’t just follow a single 24-hour curve — it also pulses in shorter bursts every 60-120 minutes throughout the day and night. A 2018 review in Sleep Medicine Reviews (Vargas et al.) proposed that these ultradian cortisol pulses can contribute to nocturnal awakenings when they become amplified or mistimed. Normally these pulses are less frequent during the night. When deep sleep is reduced, nocturnal cortisol pulsing may increase, contributing to awakenings.
A 2023 study in The Journal of Clinical Endocrinology and Metabolism (Cohn et al.) demonstrated this relationship experimentally: sleep fragmentation increased bedtime cortisol by 27% and decreased the cortisol awakening response by 57%. The more fragmented the sleep, the higher the nighttime cortisol and the flatter the morning response. This creates a self-reinforcing loop — elevated cortisol fragments sleep, and fragmented sleep elevates cortisol.
Sympathetic arousal, including elevated norepinephrine, compounds the effect.
A 2019 review in Autonomic Neuroscience (Grimaldi et al.) examined how sympathetic hyperactivity — persistent autonomic overactivation — may help explain the link between insomnia and cardiovascular risk, noting that the hyperarousal model has attracted support but that evidence remains mixed. The arousal doesn’t only happen at the moment of waking. A 2013 study (de Zambotti et al., Clinical Autonomic Research) showed that cardiac sympathetic activation was elevated throughout the entire night in people with insomnia — not just during awakenings, but continuously.
What Triggers or Worsens Autonomic Sleep Disruption?
How Does Chronic Stress Affect Autonomic Sleep Regulation?
The HPA axis is designed for acute activation — a short burst of cortisol and adrenaline in response to a specific demand, followed by a return to baseline. Under chronic stress, the return doesn’t fully happen. Baseline cortisol rises. The 24-hour rhythm flattens.
The Dressle 2022 meta-analysis found this pattern across 20 studies: people with insomnia showed consistent cortisol elevation, supporting the hyperarousal model — the idea that insomnia is a state of multi-level overactivation, not just poor sleep habits.
The relationship between cortisol and sleep is bidirectional. Elevated cortisol fragments sleep. Fragmented sleep, in turn, amplifies cortisol output. The Cohn 2023 experimental data showed a 27% cortisol increase from sleep fragmentation alone, independent of any psychological stressor. For example, a person whose sleep fragmented under sustained work stress can still have elevated nighttime cortisol after the stressor resolves — because the cortisol-sleep loop, once established, can persist for some time (Vargas et al., 2018).
Aging can compound this pattern — cortisol rhythms tend to flatten with age, and the early-morning cortisol rise may begin earlier. But age is one input among many. Chronic work stress, sustained caregiving demands, or prolonged physiological load can produce the same flattening at any adult stage.
What Disrupts GABAergic Tone?
Neuroinflammation. When inflammatory activity is elevated in the brain, it can impair the inhibitory circuits that depend on GABA. Zhu et al. (2024) identified neuroinflammation as one of five GABA-related mechanisms involved in insomnia. A 2024 study in Nutrients (Lee et al.) showed that stress-induced neuroinflammation — measured by NF-kB-positive cells in the prefrontal cortex and colon and elevated serum TNF-alpha — was accompanied by downregulation of GABA, GABA-A receptors, and serotonin. Reducing the inflammation restored GABAergic activity and sleep.
Gut microbiome disruption. The gut can be a site of microbial GABA production, and gut microbes can influence sleep-related signaling through neural, immune, endocrine, and metabolic pathways. A March 2026 study in Biomedicine & Pharmacotherapy (Tung et al.) demonstrated that GABA-producing postbiotics from Levilactobacillus brevis reversed the effects of chronic sleep deprivation in an experimental model — restoring intestinal barrier integrity, reducing serum inflammatory markers, and suppressing neuroinflammatory gene expression in the brain. The implication is that gut dysbiosis may alter sleep-related signaling.
Hormonal changes. Progesterone converts into allopregnanolone, a neurosteroid that potentiates GABA-A receptor function. The Sun et al. (2023) cryo-EM study in Nature found allopregnanolone bound to native GABA-A receptors even without external administration — meaning this is not a pharmacological effect but an endogenous regulatory mechanism. When progesterone declines — whether through hormonal transitions, chronic stress, or other factors — allopregnanolone levels can also decline, which may reduce GABA-A receptor modulation.
These three pathways — inflammation, gut microbiome, and hormonal changes — often coexist and compound each other.
Can Medications Disrupt Autonomic Sleep Regulation?
Anticholinergic drugs block acetylcholine receptors. Acetylcholine is one of the key neurotransmitters the vagus nerve uses to exert parasympathetic control, and it plays a direct role in REM sleep maintenance.
The impact scales with dose and the number of anticholinergic medications taken concurrently — a concept called anticholinergic burden. For adults taking multiple medications — common in the 40+ age range — the cumulative effect on sleep can be meaningful even when individual drug doses are low.
This is explanatory, not prescriptive. If a medication is serving a necessary function, that conversation belongs with your prescribing physician. The point here is that anticholinergic effects on sleep are a recognized mechanism, and awareness of it can inform that conversation.
How Do You Know If Autonomic Dysregulation Is Disrupting Your Sleep?
What Does Autonomic Sleep Disruption Feel Like?
- Waking alert, not groggy. If you wake at 2 or 3am and your mind is active right away — not confused or disoriented — that’s more consistent with a hyperarousal pattern than with a nonspecific awakening.
- Racing heart at rest. Elevated heart rate when you’re lying still, especially at sleep onset or during nighttime awakenings, reflects sympathetic activation that hasn’t subsided.
- “Wired but tired.” Exhaustion paired with an inability to calm down. The body wants sleep; the autonomic state won’t permit it. This is what simultaneous sympathetic activation and parasympathetic insufficiency feels like.
- Difficulty winding down before sleep. If the transition from activity to rest takes an unusually long time — if lying down doesn’t feel like the body is winding down — that may reflect low vagal tone and insufficient parasympathetic engagement.
What Can Be Measured?
Several markers are relevant:
- Heart rate variability during sleep. Lower high-frequency or respiratory-related HRV during sleep can reflect reduced vagally mediated cardiac modulation. Some consumer wearables can track HRV trends, but device methods and accuracy vary.
- Cortisol patterns. The Dressle 2022 meta-analysis found cortisol to be a useful biomarker distinguishing insomnia from healthy sleep. Hair cortisol provides a longer-term window (weeks to months) of HPA axis activity.
- Sleep architecture. Reduced deep sleep and increased light-sleep fragmentation can be consistent with autonomic interference with normal sleep staging.
Can Multiple Causes Contribute at Once?
Find out which causes are driving your 3am wakeups with the 3AM Decoder
Frequently Asked Questions
Does Autonomic Dysregulation Mean Something Is Wrong With My Nervous System?
In the context of sleep, “dysregulation” means the timing and intensity of autonomic responses are mismatched — cortisol rises too early, vagal tone doesn’t increase at sleep onset, or GABA levels are insufficient to suppress waking neural activity. These are functional imbalances, not diagnoses of nerve damage.
Can Heart Rate Variability From a Wearable Tell Me If I Have Autonomic Sleep Disruption?
Consumer wearables measure HRV through photoplethysmography (PPG) at the wrist. The accuracy varies by device, but the trend over weeks and months is more informative than any single night. If your overnight HRV is consistently low or shows little of the usual overnight rise relative to your own baseline, that’s worth noting — it suggests parasympathetic activation may not be increasing during sleep as it should.
Is Cortisol Testing Useful for Identifying Autonomic Sleep Disruption?
A single morning cortisol draw has limited value — it tells you the level at one point, but sleep disruption is about the shape of the 24-hour curve. Salivary cortisol collected at wake, 30 minutes post-wake, afternoon, and bedtime can reveal whether the curve is flattened, whether the cortisol awakening response is blunted, and whether bedtime cortisol remains elevated. Hair cortisol provides a retrospective window over weeks to months.
Can Autonomic Sleep Disruption Improve Without Medication?
The three mechanisms in this article are not permanent states. Vagal tone can increase with sustained practices that activate the parasympathetic branch (Laborde et al., 2019). GABAergic tone responds to changes in inflammation, gut microbiome composition, and hormonal status. HPA axis activity responds to changes in stress exposure and sleep continuity (Cohn et al., 2023). Identifying which mechanisms are contributing is the first step — which is what the 3AM Decoder is designed for.
Related Reading
- Can Your Nervous System Get Stuck in Fight or Flight and Ruin Your Sleep? — The broad entry point for autonomic hyperarousal and sympathetic activation.
- Why Do Cholinergic Supplements Give You Vivid Dreams? — How acetylcholine can affect REM sleep and dream intensity.
- Why Can You Not Sleep After COVID? What Happened to Your Vagus Nerve — How post-viral dysautonomia can affect sleep continuity.
- Does Polyvagal Theory Explain Insomnia? — How trauma and autonomic state shifts may shape insomnia patterns.
- Which Probiotic Strains Improve Sleep? Vagus Nerve Evidence — How gut microbes can signal through vagal and sleep-related pathways.
- Why Does Inflammation Cause Insomnia? — How vagal inflammatory signaling can bridge immune activation and sleep.
- Inflammatory Sleep Disruption — How cytokines, histamine, and immune activation can fragment sleep.
- Metabolic Sleep Disruption — How blood sugar, insulin resistance, and energy metabolism can drive night waking.
- Circadian Sleep Disruption — How clock timing, cortisol rhythm, temperature, and melatonin shape sleep.
- Hormonal Sleep Disruption in Men — How testosterone, cortisol, and growth hormone affect sleep architecture.
- Hormonal Sleep Disruption in Women — How estrogen, progesterone, FSH, and thermoregulation affect sleep.
References
Vagal Tone and Parasympathetic Sleep Regulation
- Joubert, M., Beilharz, J. E., Fatt, S., Chung, Y. M., Cvejic, E., Vollmer-Conna, U., & Burton, A. R. (2024). Stress reactivity, wellbeing and functioning in university students: A role for autonomic activity during sleep. Stress and Health, 40(6), e3509. https://pubmed.ncbi.nlm.nih.gov/39616430/
- Menuet, C., Ben-Tal, A., Linossier, A., Allen, A. M., Machado, B. H., Moraes, D. J. A., Farmer, D. G. S., Paterson, D. J., Mendelowitz, D., Lakatta, E. G., Taylor, E. W., Ackland, G. L., Zucker, I. H., Fisher, J. P., Schwaber, J. S., Shanks, J., Paton, J. F. R., Buron, J., Spyer, K. M., … Gourine, A. V. (2025). Redefining respiratory sinus arrhythmia as respiratory heart rate variability: An international expert recommendation for terminological clarity. Nature Reviews Cardiology, 22(12), 978-984. https://pubmed.ncbi.nlm.nih.gov/40328963/
- Ziegler, K. A., Engelhardt, S., Carnevale, D., McAlpine, C. S., Guzik, T. J., Dimmeler, S., & Swirski, F. K. (2025). Neural mechanisms in cardiovascular health and disease. Circulation Research, 136(11), 1233-1261. https://pubmed.ncbi.nlm.nih.gov/40403111/
- Laborde, S., Hosang, T., Mosley, E., & Dosseville, F. (2019). Influence of a 30-day slow-paced breathing intervention compared to social media use on subjective sleep quality and cardiac vagal activity. Journal of Clinical Medicine, 8(2), 193. https://pubmed.ncbi.nlm.nih.gov/30736268/
- Anderson, C., Forte, G., Hu, W., Zhang, H., Boyett, M. R., & D’Souza, A. (2023). Non-canonical role of the sympathetic nervous system in the day-night rhythm in heart rate. Philosophical Transactions of the Royal Society B, 378(1879), 20220179. https://pubmed.ncbi.nlm.nih.gov/37122216/
GABAergic Mechanisms and Sleep
- Varinthra, P., Anwar, S. N. M. N., Shih, S. C., & Liu, I. Y. (2024). The role of the GABAergic system on insomnia. Tzu Chi Medical Journal, 36(2), 103-109. https://pubmed.ncbi.nlm.nih.gov/38645778/
- Zhu, W., Huang, L., Cheng, H., Li, N., Zhang, B., Dai, W., Wu, X., Zhang, D., Feng, W., Li, S., & Xu, H. (2024). GABA and its receptors’ mechanisms in the treatment of insomnia. Heliyon, 10(23), e40665. https://pubmed.ncbi.nlm.nih.gov/39654705/
- Jiang, Y., Guo, L., He, H., Chen, H., Chen, T., Liu, Y., & Zhao, W. (2025). Lactobacillus reuteri E9 regulates sleep disorders through its metabolite GABA. Frontiers in Bioscience (Landmark edition), 30(6), 39587. https://pubmed.ncbi.nlm.nih.gov/40613302/
- Ye, X., Sun, C., Zhao, Y., Wang, W., Li, Z., Liu, L., & Han, X. (2025). Promotion of sleep by cinnamic acid in parachlorophenylalanine-induced insomnia in rats. International Immunopharmacology, 158, 114852. https://pubmed.ncbi.nlm.nih.gov/40367689/
- Sun, C., Zhu, H., Clark, S., & Gouaux, E. (2023). Cryo-EM structures reveal native GABAA receptor assemblies and pharmacology. Nature, 622(7981), 195-201. https://pubmed.ncbi.nlm.nih.gov/37730991/
- Winkelman, J. W., Buxton, O. M., Jensen, J. E., Benson, K. L., O’Connor, S. P., Wang, W., & Renshaw, P. F. (2008). Reduced brain GABA in primary insomnia: Preliminary data from 4T proton magnetic resonance spectroscopy (1H-MRS). Sleep, 31(11), 1499-1506. https://pubmed.ncbi.nlm.nih.gov/19014069/
- Gottesmann, C. (2002). GABA mechanisms and sleep. Neuroscience, 111(2), 231-239. https://pubmed.ncbi.nlm.nih.gov/11983310/
Cortisol, HPA Axis, and Stress-Related Sleep Disruption
- Dressle, R. J., Feige, B., Spiegelhalder, K., Schmucker, C., Benz, F., Mey, N. C., & Riemann, D. (2022). HPA axis activity in patients with chronic insomnia: A systematic review and meta-analysis of case-control studies. Sleep Medicine Reviews, 62, 101588. https://pubmed.ncbi.nlm.nih.gov/35091194/
- Vargas, I., Vgontzas, A. N., Abelson, J. L., Faghih, R. T., Morales, K. H., & Perlis, M. L. (2018). Altered ultradian cortisol rhythmicity as a potential neurobiologic substrate for chronic insomnia. Sleep Medicine Reviews, 41, 234-243. https://pubmed.ncbi.nlm.nih.gov/29678398/
- Cohn, A. Y., Grant, L. K., Nathan, M. D., Wiley, A., Abramson, M., Harder, J. A., Crawford, S., Klerman, E. B., Scheer, F. A. J. L., Kaiser, U. B., Rahman, S. A., & Joffe, H. (2023). Effects of sleep fragmentation and estradiol decline on cortisol in a human experimental model of menopause. The Journal of Clinical Endocrinology & Metabolism, 108(11), e1347-e1357. https://pubmed.ncbi.nlm.nih.gov/37207451/
Autonomic Hyperarousal and Insomnia
- Lin, Y. K., Huang, Y. C., Huang, W. C., Lee, H. C., Lin, I. M., & Yang, C. M. (2026). Can addressing autonomic hyperarousal with heart rate variability biofeedback enhance CBT-I outcomes in insomnia disorder? Behavioral Sleep Medicine, 24(1), 40-57. https://pubmed.ncbi.nlm.nih.gov/40873182/
- Grimaldi, D., Goldstein, M. R., & Carter, J. R. (2019). Insomnia and cardiovascular autonomic control. Autonomic Neuroscience: Basic & Clinical, 220, 102551. https://pubmed.ncbi.nlm.nih.gov/31331688/
- McCall, W. V., Looney, S. W., Zulfiqar, M., Ketcham, E., Jones, M., Mixson, C., McCloud, L., Miller, B. J., & Rosenquist, P. B. (2023). Daytime autonomic nervous system functions differ among adults with and without insomnia symptoms. Journal of Clinical Sleep Medicine, 19(11), 1885-1893. https://pubmed.ncbi.nlm.nih.gov/37421322/
- Laharnar, N., Grote, L., Zou, D., Hedner, J., Sommermeyer, D., Strassberger, C., Marciniak, A., Potzka, S., Lederer, K., Glos, M., Zimmermann, S., Fietze, I., & Penzel, T. (2020). Overnight pulse wave analysis to assess autonomic changes during sleep in insomnia patients and healthy sleepers. PLoS ONE, 15(5), e0232589. https://pubmed.ncbi.nlm.nih.gov/32379833/
- Jarrin, D. C., Ivers, H., Lamy, M., Chen, I. Y., Harvey, A. G., & Morin, C. M. (2018). Cardiovascular autonomic dysfunction in insomnia patients with objective short sleep duration. Journal of Sleep Research, 27(3), e12663. https://pubmed.ncbi.nlm.nih.gov/29493063/
GABAergic Disruption: Neuroinflammation, Gut-Brain Axis, and Hormonal Factors
- Tung, Y. T., Yang, C. H., Chen, B. C., Hsieh, C. C., Yang, Y. S. H., Yang, C. T., Xu, J. W., & Wu, J. H. (2026). Preventive effects of GABA-producing postbiotics derived from Levilactobacillus brevis against chronic sleep deprivation-induced gut-brain axis dysfunction, neuroinflammation, and behavioral impairments in mice. Biomedicine & Pharmacotherapy, 196, 119093. https://pubmed.ncbi.nlm.nih.gov/41653907/
- Lee, D. Y., Baek, J. S., Shin, Y. J., & Kim, D. H. (2024). Alleviation of immobilization stress or fecal microbiota-induced insomnia and depression-like behaviors in mice by Lactobacillus plantarum and its supplement. Nutrients, 16(21), 3711. https://pubmed.ncbi.nlm.nih.gov/39519543/
- Fang, H., Wang, Y. H., Yang, L., Che, Y. H., Liu, F. L., & Liu, H. (2026). Revealing the mechanism of suanzaoren tang against insomnia via integrated metabolomics and gut microbiota analysis. Journal of Pharmaceutical and Biomedical Analysis, 269, 117231. https://pubmed.ncbi.nlm.nih.gov/41207006/
- Nohesara, S., Mostafavi Abdolmaleky, H., Pirani, A., & Thiagalingam, S. (2025). Therapeutic horizons: Gut microbiome, neuroinflammation, and epigenetics in neuropsychiatric disorders. Cells, 14(13), 1027. https://pubmed.ncbi.nlm.nih.gov/40643545/
Cortisol Awakening Response and Aging
- Heaney, J. L. J., Phillips, A. C., & Carroll, D. (2012). Aging, health behaviors, and the diurnal rhythm and awakening response of salivary cortisol. Experimental Aging Research, 38(3), 295-314. https://pubmed.ncbi.nlm.nih.gov/22540384/
