Most sleep content treats inflammation as a chemical disruptor — cytokines interfere with sleep neurotransmitters, TNF-alpha fragments sleep architecture, interleukin-6 shifts circadian timing. That is real, but incomplete. Inflammation also causes physical nerve damage. When the target is the vagus nerve, the consequences extend beyond a bad night: the nerve that regulates the parasympathetic shift required for sleep onset is structurally compromised.
This article covers how inflammation physically damages vagal nerve fibers, why small fiber neuropathy predicts sleep disruption independently of other symptoms, how vagal damage creates a self-perpetuating inflammatory cycle, and whether the damage can be reversed. The focus is inflammation as a cause of nerve damage — the vagus nerve as the victim, not just the messenger. For a full overview of autonomic sleep disruption mechanisms including vagal tone, GABA, and cortisol, see How Does Autonomic Nervous System Impairment Disrupt Sleep?.
How Does Chronic Inflammation Physically Damage the Vagus Nerve?
The most direct evidence for inflammation causing structural vagal damage comes from postmortem histopathology. Woo et al. (2023) examined vagus nerve tissue from COVID-19 fatalities and detected SARS-CoV-2 RNA inside the nerve alongside inflammatory cell infiltrates composed predominantly of monocytes. This is not indirect evidence of vagal dysfunction — the inflammatory cells were physically present within the nerve tissue itself.
RNA sequencing of the affected vagal tissue revealed something more significant than the presence of immune cells alone. Interferon signaling was strongly upregulated in neurons and endothelial cells, while Schwann cells showed stress responses including upregulation of autophagy, endoplasmic reticulum stress, and proteasomal catabolism pathways. The intensity of this response correlated with local viral load within the nerve, establishing a dose-response relationship between viral presence and tissue-level damage.
The Schwann cell involvement has particular functional significance. Schwann cells are the glial cells responsible for maintaining myelin sheaths and supporting nerve conduction velocity in peripheral nerves. When Schwann cells are stressed and their gene expression is disrupted, the nerve fibers they support lose structural integrity and signal transmission capacity. Schwann cell injury in the vagus nerve directly impairs the speed and reliability of autonomic signals traveling between the brainstem and peripheral organs.
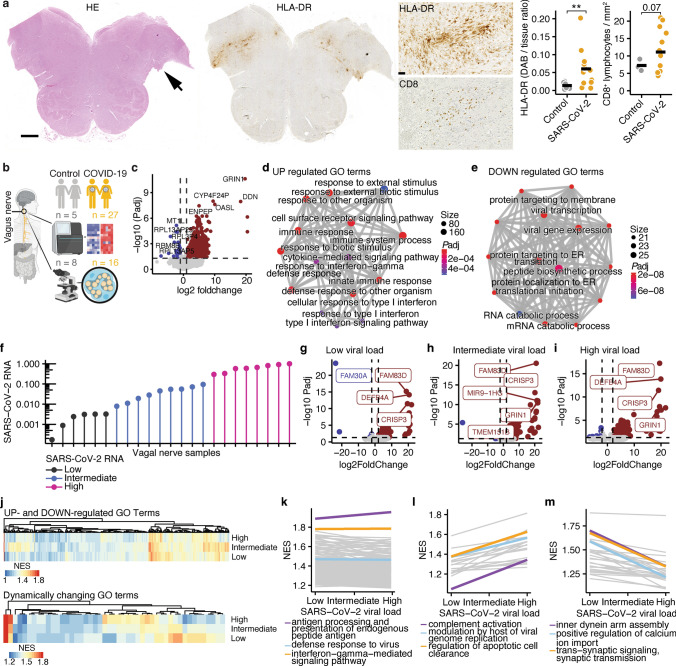
The damage is not limited to the peripheral nerve trunk. Andersson and Tracey (2024) synthesized pathological evidence showing inflammatory infiltration in the vagal brainstem nuclei — specifically the dorsal motor nucleus and nucleus tractus solitarius. The dorsal motor nucleus generates efferent parasympathetic signals that travel down the vagus nerve to regulate cardiac, respiratory, and gastrointestinal function. The nucleus tractus solitarius receives afferent sensory information from peripheral organs and relays that information to higher brain centers. When both nuclei are inflamed alongside the peripheral nerve trunk, the entire vagal signaling pathway — sensory input, central processing, and motor output — is compromised simultaneously.
The implication for sleep is structural, not merely chemical. The vagus nerve contains approximately 75% of all parasympathetic nerve fibers. The parasympathetic shift — the transition from sympathetic dominance during waking to parasympathetic dominance during sleep — depends on intact vagal fibers transmitting signals at adequate velocity and amplitude. When both the peripheral vagal fibers and their brainstem relay nuclei are inflamed and structurally damaged, this parasympathetic shift is not just chemically suppressed by circulating cytokines — it is physically disabled by the destruction of the hardware that executes it.
What Is Small Fiber Neuropathy and Why Does It Predict Sleep Disruption?
Small fiber neuropathy is a classification for peripheral nerve disease that selectively affects the thinnest nerve fibers in the body. Finsterer and Scorza (2022) classify the affected fibers into two categories: A-delta fibers, which are thinly myelinated and carry pain and temperature signals, and C-fibers, which are unmyelinated and carry both pain signals and autonomic commands. The distinction between large fiber neuropathy and small fiber neuropathy matters clinically because standard nerve conduction studies — the tests most neurologists perform first — only detect damage to large myelinated fibers. Small fiber damage is invisible on these tests.
The diagnostic gold standard for small fiber neuropathy is intraepidermal nerve fiber density measured from a skin biopsy. A small punch biopsy is taken from the distal leg, and the density of nerve fiber endings crossing into the epidermal layer is counted under a microscope. When the fiber count falls below age- and sex-adjusted norms, small fiber neuropathy is confirmed. Finsterer and Scorza classify the causes across metabolic (diabetes, prediabetes), immune-mediated (autoimmune attack on nerve fibers), infectious (post-viral destruction), toxic (chemotherapy, alcohol), and hereditary categories. The immune-mediated causes are specifically relevant because they are potentially treatable with immunosuppression.
The strongest quantitative evidence linking small fiber neuropathy to sleep disruption comes from Xie et al. (2025), who studied 100 Parkinson’s disease patients and performed multiple regression analysis controlling for relevant confounders. Small fiber neuropathy symptom severity — assessed using the 13-item Small Fiber Neuropathy Symptom Inventory Questionnaire (SFN-SIQ) — was independently associated with sleep disturbance with a regression coefficient of B=0.694 (95% confidence interval 0.309-1.080, p=0.001). The association remained significant after adjusting for confounding variables, indicating that small fiber neuropathy symptoms contribute to sleep disturbance independently of other non-motor domains. Whether this association generalizes beyond Parkinson’s populations requires further study, but the biological mechanism — autonomic C-fiber loss disrupting overnight parasympathetic regulation — applies to any condition involving small fiber degeneration.
The same study found an even stronger association between small fiber neuropathy symptom severity and autonomic dysfunction (B=1.118, p<0.001), which was the highest regression coefficient among all non-motor domains tested. This result is consistent with what the fiber anatomy predicts: the unmyelinated C-fibers destroyed in small fiber neuropathy are the same fibers that regulate heart rate variability, blood pressure responses, and the parasympathetic-sympathetic balance that shifts during sleep onset.
The connection between these two findings — small fiber neuropathy predicting both autonomic dysfunction and sleep disturbance — points to a specific mechanism. The autonomic C-fibers that degenerate in small fiber neuropathy are the fibers responsible for overnight parasympathetic regulation. When these fibers are destroyed, the overnight autonomic profile shifts toward sympathetic dominance — elevated heart rate, reduced heart rate variability, suppressed parasympathetic tone. This produces the same sympathetic-dominant overnight pattern seen in other autonomic insomnia subtypes covered elsewhere in this cluster, but the cause is structural fiber loss rather than a functional imbalance that might respond to behavioral interventions.
Does the Vicious Cycle Between Vagal Damage and Inflammation Explain Chronic Insomnia?
The vagus nerve is not only a target of inflammation — it is part of the immune regulatory system that normally keeps inflammation in check. Andersson and Tracey (2024) describe the cholinergic anti-inflammatory pathway, also called the inflammatory reflex: afferent vagal fibers detect peripheral inflammation and relay that information to the brainstem, which then activates efferent vagal fibers that release acetylcholine in peripheral organs. Acetylcholine binds to alpha-7 nicotinic receptors on macrophages and suppresses the production of pro-inflammatory cytokines including TNF-alpha, interleukin-1, and interleukin-6. This reflex functions as a continuous brake on peripheral immune activation.
When inflammation physically damages the vagus nerve — destroying the fibers that carry this anti-inflammatory signal — the cholinergic anti-inflammatory pathway is disabled. Peripheral macrophages continue producing cytokines, but the vagal signal that would normally suppress that production no longer arrives. The inflammation that caused the initial nerve damage is now unregulated because the regulator has been destroyed. This transforms a one-time inflammatory insult into a chronic condition: the resolution mechanism itself is damaged.

Camici et al. (2024) extend this single-axis failure into a triple-axis model. Their review proposes that long-term inflammatory conditions produce simultaneous dysfunction of three interconnected regulatory systems: the vagal cholinergic anti-inflammatory pathway, the hypothalamic-pituitary-adrenal cortisol axis, and mitochondrial redox homeostasis. The authors identify acetylcholine and cortisol as the two “fundamental players” — both function as immune suppressors, and both are regulated through pathways that traverse or depend on the vagus nerve.
When vagal cholinergic signaling is lost due to structural nerve damage, the acetylcholine-mediated suppression of peripheral cytokines fails. When the hypothalamic-pituitary-adrenal axis is simultaneously disrupted — because vagal afferent signals to the hypothalamus are also impaired — cortisol regulation becomes dysfunctional. The result is double immune brake failure: neither the neural pathway (acetylcholine) nor the hormonal pathway (cortisol) adequately suppresses peripheral inflammation.
Both vagal tone and cortisol are critical regulators of sleep. Vagal tone determines overnight parasympathetic dominance and heart rate variability patterns. Cortisol follows a circadian rhythm that reaches its nadir in the first half of the night, permitting deep sleep, then rises toward morning. When both regulatory systems are damaged by the same inflammatory process they would normally control, sleep disruption becomes embedded in the pathology — a direct consequence of the structural damage driving the immune dysregulation.
This vicious cycle explains why post-inflammatory insomnia persists long after the original trigger has resolved. Vagal inflammatory neuropathy disables the system responsible for resolving inflammation. The inflammation does not resolve because the resolution mechanism is damaged, and unresolved inflammation continues damaging the nerve.
Can Vagus Nerve Damage Be Reversed?
Whether vagal small fiber damage can be reversed depends on what is causing the ongoing fiber destruction. Finsterer and Scorza (2022) present a three-tier treatment framework that distinguishes between addressing the cause, modifying the injury mechanism, and managing symptoms.
The first tier — causal therapy — targets the underlying disease driving fiber loss. For metabolic causes, causal therapy means optimizing glucose control or repleting nutritional deficiencies. For toxic causes, it means removing the offending agent. For immune-mediated small fiber neuropathy, causal therapy means immunosuppression: corticosteroids, intravenous immunoglobulin, or other immunosuppressants that halt the autoimmune attack on nerve fibers. This distinction matters because immune-mediated fiber destruction is driven by an ongoing process that can be stopped, unlike fiber loss from completed toxic damage where the agent has already been removed.
The second tier — pathophysiologic therapy — modifies the mechanisms of nerve injury independently of the underlying cause. This includes interventions that reduce oxidative stress, improve nerve blood flow, or protect remaining fibers from further damage during the period before causal therapy takes full effect.
The third tier — symptomatic therapy — manages pain and autonomic dysfunction while causal and pathophysiologic treatments work to halt and reverse the underlying fiber loss. Yaukey and Kaur (2026) present the most current pharmacological guidelines: first-line options include amitriptyline, nortriptyline, gabapentin, and pregabalin. Second-line options include duloxetine and venlafaxine (serotonin-norepinephrine reuptake inhibitors). Topical treatments include lidocaine patches and capsaicin for localized neuropathic pain.
Yaukey and Kaur emphasize that identifying and treating the root cause is critical for symptom improvement — not just symptom management. The diagnostic workup they recommend begins with needle electromyography and nerve conduction studies to exclude large fiber involvement, followed by skin biopsy to confirm small fiber neuropathy, then systematic evaluation for metabolic, autoimmune, infectious, nutritional, and genetic etiologies. When a treatable cause is identified — particularly an autoimmune or metabolic one — directed therapy can halt fiber destruction and permit regeneration.
Small unmyelinated C-fibers can regenerate when the cause of their destruction is removed, with regrowth potentially occurring within months in immune-mediated cases. This means that immune-mediated vagal small fiber neuropathy treated with appropriate immunotherapy has a biological basis for recovery — the autonomic C-fibers that regulate overnight parasympathetic function can regenerate, potentially restoring the parasympathetic shift that sleep onset requires.
Recovery can be tracked objectively. Heart rate variability monitoring provides a non-invasive measure of vagal function over time — as vagal C-fibers regenerate, overnight heart rate variability metrics should increase, reflecting restored parasympathetic signaling capacity. For a detailed guide to interpreting overnight heart rate variability as a vagal function marker, see What Your Overnight Heart Rate Variability Is Telling You About Your Sleep: The Vagal Tone Connection. Vagus nerve stimulation devices may also serve as an adjunct during the recovery period by providing external stimulation to the nerve while endogenous fiber regeneration proceeds; see Can Vagus Nerve Stimulation Devices Improve Insomnia? for available device options and evidence.
Vagus nerve damage from inflammation may not be the only factor affecting your sleep. Autonomic hyperarousal, GABA receptor changes, metabolic disruptions, or hormonal imbalances may also be contributing. When multiple causes overlap, identifying which ones are active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Does Long COVID Damage the Vagus Nerve?
How Common Is Small Fiber Neuropathy in Postural Orthostatic Tachycardia Syndrome?
Is All Dysautonomia Caused by Nerve Damage?
How Is Small Fiber Neuropathy Diagnosed?
Can Young People Develop Vagal Small Fiber Neuropathy?
Related Reading
- How Does Autonomic Nervous System Dysfunction Disrupt Sleep?
- Why Does Inflammation Cause Insomnia?
- Can Vagus Nerve Stimulation Stop Mast Cell Insomnia?
- How Does Your Vagus Nerve Actively Suppress Inflammation?
- What Does Heart Rate Variability Reveal About Inflammation and Vagal Tone During Sleep?
References
- Woo, M. S., Shafiq, M., Fitzek, A., Dottermusch, M., Altmeppen, H., Mohammadi, B., Mayer, C., Bal, L. C., Raich, L., Matschke, J., Krasemann, S., Pfefferle, S., Brehm, T. T., Lütgehetmann, M., Schädler, J., Addo, M. M., Schulze Zur Wiesch, J., Ondruschka, B., Friese, M. A., & Glatzel, M. (2023). Vagus nerve inflammation contributes to dysautonomia in COVID-19. Acta neuropathologica, 146(3), 387-394. https://pubmed.ncbi.nlm.nih.gov/37452829/
- Andersson, U., & Tracey, K. J. (2024). Vagus nerve SARS-CoV-2 infection and inflammatory reflex dysfunction: Is there a causal relationship? Journal of internal medicine, 295(1), 91-102. https://pubmed.ncbi.nlm.nih.gov/38018736/
- Finsterer, J., & Scorza, F. A. (2022). Small fiber neuropathy. Acta neurologica Scandinavica, 145(5), 493-503. https://pubmed.ncbi.nlm.nih.gov/35130356/
- Xie, C., Chen, J., Gan, R., Gao, Y., Wang, L., Ma, G., Zhang, Y., Nie, K., & Wang, L. (2025). Patients with small fiber neuropathy exhibit more pronounced non-motor symptoms in Parkinson’s disease. Journal of Parkinson’s disease, 15(8), 1442-1451. https://pubmed.ncbi.nlm.nih.gov/40891687/
- Camici, M., Del Duca, G., Brita, A. C., & Antinori, A. (2024). Connecting dots of long COVID-19 pathogenesis: a vagus nerve-hypothalamic-pituitary-adrenal-mitochondrial axis dysfunction. Frontiers in cellular and infection microbiology, 14, 1501949. https://pubmed.ncbi.nlm.nih.gov/39735263/
- Yaukey, J., & Kaur, D. (2026). Management of Small Fiber Neuropathy: A Clinical Perspective. Muscle & nerve, 73(3), 380-384. https://pubmed.ncbi.nlm.nih.gov/41367223/
- Azcue, N., Del Pino, R., Acera, M., Fernández-Valle, T., Ayo-Mentxakatorre, N., Pérez-Concha, T., Murueta-Goyena, A., Lafuente, J. V., Prada, A., López de Munain, A., Ruiz Irastorza, G., Martín-Iglesias, D., Ribacoba, L., Gabilondo, I., Gómez-Esteban, J. C., & Tijero-Merino, B. (2023). Dysautonomia and small fiber neuropathy in post-COVID condition and Chronic Fatigue Syndrome. Journal of translational medicine, 21(1), 814. https://pubmed.ncbi.nlm.nih.gov/37968647/
- Moak, J. P., Ramwell, C. B., Gordish-Dressman, H., Sule, S. D., & Bettini, E. (2024). Small fiber neuropathy in children, adolescents, and young adults with chronic orthostatic intolerance and postural orthostatic tachycardia syndrome: A retrospective study. Autonomic neuroscience: basic & clinical, 253, 103163. https://pubmed.ncbi.nlm.nih.gov/38537312/
- Bay-Smidt, C. N., Bruun, K. D., Gaist, L. M., Krøigård, A. B., Sindrup, S. H., & Krøigård, T. (2025). Structured Symptom Assessment to Identify Patients With Small Fiber or Autonomic Neuropathy in Fibromyalgia. European journal of neurology, 32(9), e70349. https://pubmed.ncbi.nlm.nih.gov/40913368/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 9 references cited
