sleep advice focuses on duration: get seven to eight hours. But a growing body of research shows that fragmented sleep may inflict more biological stress than short sleep. Someone sleeping eight hours but waking four or five times per night can build more oxidative stress, more vascular injury, and more neuroinflammation than someone sleeping six uninterrupted hours. The distinction between "how much sleep" and "how continuous is your sleep" matters for the millions of adults whose trackers show adequate total sleep time but fragmented architecture.
This article covers the measured biological impacts of sleep fragmentation — what happens inside blood vessels, brain cells, and mitochondria when sleep continuity breaks down. It does not cover how to fix fragmented sleep through supplements (see CoQ10, PQQ: Can Mitochondrial Supplements Fix Your Sleep?) or exercise (see How Exercise Rebuilds Your Mitochondria and Fixes Your Sleep). Sleep fragmentation represents the downstream consequences of the metabolic disruptions covered across the mitochondria sub-cluster. For the full overview, see Metabolic Sleep Disruption.
Is Fragmented Sleep Harder on the Body Than Short Sleep?
The strongest evidence that fragmentation — not reduced sleep time — drives cellular stress comes from Zhang et al. (2014). Male mice exposed to sleep fragmentation developed time-dependent insulin resistance (p<0.01 at 3 days, p<0.00003 at 14 days) and visceral adipose tissue inflammation, all without any reduction in total sleep duration. NADPH oxidase 2 (NOX2), an enzyme that generates ROS, was identified as the necessary mediator. In mice genetically lacking the Nox2 gene, all fragmentation-induced oxidative stress, inflammation, and insulin resistance were abolished — the fragmentation stimulus continued, but with NOX2 removed, the downstream stress disappeared.
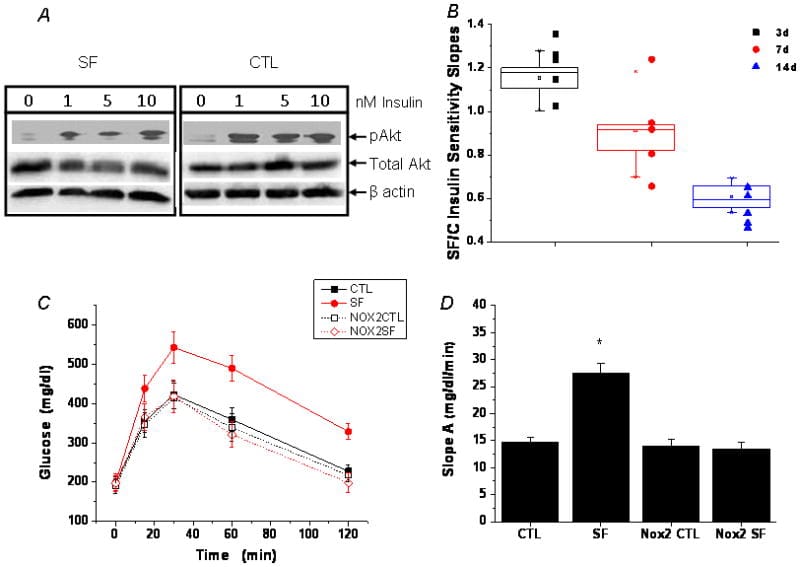
The human dose-response data reinforces the animal findings. A study of 60 adults (30 with severe obstructive sleep apnea and 30 controls) found depleted catalase (p<0.001) and glutathione peroxidase — two of the body’s primary antioxidant enzymes — compared to controls. Malondialdehyde, a marker of lipid peroxidation (oxidative stress to cell membranes), was elevated (p<0.001). Antioxidant enzyme depletion correlated inversely with the apnea-hypopnea index, establishing a direct dose-response between fragmentation severity and oxidative stress. Within the same individuals, malondialdehyde correlated positively with C-ROS-related protein, linking oxidative stress to body-wide inflammation (Asker et al., 2015).
A 2025 study in diabetic mice shows the stress persists beyond the period of sleep disruption. Sleep fragmentation upregulated NADPH oxidase 4 (NOX4, a different enzyme from the NOX2 pathway described above) through epigenetic modification — specifically, increased histone H3K27 acetylation at the Nox4 gene promoter. The resulting liver and heart stress persisted after two full weeks of recovery sleep. The epigenetic marks that sustained NOX4 overexpression outlasted the sleep disruption itself, providing early evidence that fragmentation-induced oxidative stress may become locked in at the gene-regulation level, at least in the context of concurrent diabetes (Guo et al., 2025).
Does Interrupted Sleep Physically Stress Your Blood Vessels?
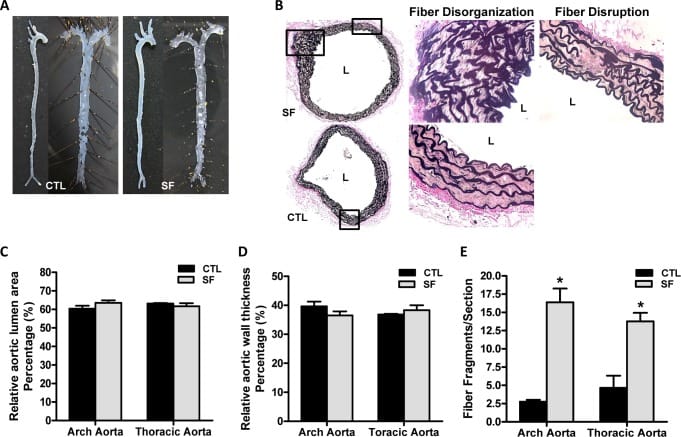
A 20-week mouse model provides the detailed evidence of how sleep fragmentation remodels blood vessels. Chronically fragmented mice developed increased systolic blood pressure beginning at 8 weeks. Endothelial impairment — impaired ability of blood vessel walls to regulate blood flow — was shown by delayed restoration of skin perfusion after vascular occlusion (p<0.03). Aortic tissue showed elastic fiber disruption, increased foam cell deposition (lipid-laden immune cells that characterize early atherosclerosis), and macrophage infiltration. Senescence markers moved: TERT (telomerase) and cyclin A decreased while p16INK4a increased, and plasma IL-6 was elevated. The 20-week timeline demonstrates progressive structural remodeling — the vascular stress built up with sustained fragmentation (Carreras et al., 2014).
Human data shows the vascular stress is not limited to animal models. A study of 144 children found that those with obstructive sleep apnea had elevated NADPH oxidase activation (measured by soluble NOX2-derived peptide) and markedly reduced flow-mediated dilation compared to controls (p<0.001). Fifteen children who received adenotonsillectomy showed reversal in lockstep: soluble NOX2-derived peptide fell from 38.2 to 22.4 pg/mL (p<0.001) and flow-mediated dilation improved from 3.0% to 8.0% (p<0.001). Removing the source of sleep fragmentation reversed both oxidative stress and vascular impairment. The finding in children is particularly significant — endothelial injury from sleep fragmentation begins before traditional cardiovascular likelihood factors build (Loffredo et al., 2015).
The convergence of foam cell deposition, elastic fiber stress, and senescence marker changes (p16INK4a elevation, TERT reduction) in the mouse model indicates that fragmentation alone — without concurrent hypoxia — is sufficient to produce vascular changes consistent with accelerated aging.
Is Fragmented Sleep Linked to Alzheimer’s and Neurodegeneration?
The link between sleep fragmentation and neurodegeneration does not require genetic predisposition or advanced age. Young adult mice without any predisposition to neurodegeneration underwent two months of chronic sleep fragmentation and developed spatial learning and memory deficits alongside increased anxiety. Intracellular amyloid-beta — a hallmark of Alzheimer’s disease — built up in both the cortex and hippocampus. Endosomal trafficking was disrupted (Rab5 and Rab7 elevated, p<0.05), and autophagy proteins LC3B, Beclin 1, and UVRAG were upregulated in a compensatory but inadequate response to protein clearance reduction. Microglial activation was pronounced. Sleep fragmentation alone was sufficient to initiate impaired protein clearance and the neuroinflammatory state that characterize early neurodegeneration in this model (Xie et al., 2020).
A 2024 study identified the mitochondrial mechanism connecting sleep disruption to brain inflammation. Sleep-deprived rats showed mitochondrial DNA oxidation in brain tissue. The oxidized mitochondrial DNA was released from the mitochondrial matrix into the cytosol, where it functioned as a stress-associated molecular pattern (DAMP) — an internal alarm cue. The released mitochondrial DNA triggered NF-kappaB-mediated neuroinflammation and pro-inflammatory cytokine production. When researchers suppressed mitochondrial DNA oxidation, both cytosolic mitochondrial DNA release and downstream inflammation were reduced, establishing the causal sequence: sleep disruption stresses mitochondria, which then release their own stressd DNA as a cue that activates brain immune cells (Hu et al., 2024).
A 2024 review examining the bidirectional relationship between mitochondria and sleep disorders establishes that sleep disorders stress mitochondria, and stressd mitochondria in turn lowern sleep quality — a self-amplifying cycle linked to Alzheimer’s disease, Parkinson’s disease, and amyotrophic lateral sclerosis through impaired electron transport, increased ROS production, and defective mitophagy. Midlife sleep disruption is associated with neurodegenerative likelihood decades later, potentially because mitochondrial stress compounds over time rather than stabilizing (Zhang et al., 2024). For more on the bidirectional relationship between mitochondria and sleep, see How Your Mitochondria Decide When You Sleep (And Why It Breaks After 40).
A 2025 study identifies the specific cell type vulnerable to sleep-deprivation oxidative stress: six hours of acute sleep deprivation caused oxidative stress concentrated in hippocampal parvalbumin interneurons — fast-firing inhibitory neurons required for gamma oscillations and memory precision. Parvalbumin interneurons were selectively vulnerable relative to other hippocampal populations, and N-acetylcysteine (a glutathione precursor) rescued both parvalbumin interneuron survival and memory precision, indicating oxidative stress as the upstream mechanism (Gao et al., 2025).
Does Sleep Fragmentation Accelerate Biological Aging?
The 20-week fragmentation model did not only produce functional vascular impairment — it moved the molecular aging profile of vascular tissue. p16INK4a, a cyclin-dependent kinase inhibitor that builds as cells enter senescence, was elevated. TERT (telomerase reverse transcriptase, which maintains telomere length) and cyclin A (a cell-cycle progression marker) were reduced. This molecular profile matches accelerated vascular aging — the tissue is biologically older than its chronological age (Carreras et al., 2014).
The 2025 epigenetic evidence shows the aging effect persists beyond the period of sleep disruption. Sleep fragmentation modified histone H3K27 acetylation at the Nox4 gene promoter through a BMAL1-dependent SIRT1 pathway, connecting the circadian clock to oxidative stress gene regulation. This epigenetic change sustained NOX4 overexpression and organ stress even after two weeks of recovery sleep — "catching up on sleep" did not reverse the epigenetically locked stress within the study’s recovery window (Guo et al., 2025). For more on what happens during middle-of-the-night wakeups, see The 3am Wakeup Is a Metabolic Emergency: Blood Sugar, Cortisol, and Mitochondrial reduction.
Neurophysiological evidence from healthy humans shows that the brain builds a measurable impact from fragmented sleep. A 2024 study using EEG found that sleep fragmentation elevated high-beta power during cognitive tasks — indicating the brain expends greater neural resources to maintain performance — and produced sustained beta and alpha power elevation in post-task rest compared to pre-task levels. The brain under fragmented sleep compensates by spending more neural energy, and post-task neural activity remains elevated compared to pre-task levels. This pattern suggests the brain does not reset after fragmented sleep (Benkirane et al., 2024).
The bidirectional cycle connects these findings: fragmented sleep stresses mitochondria, and stressd mitochondria produce more ROS, which fragment sleep further. Each cycle degrades mitochondrial quantity and quality, which is why sleep patterns in midlife compound rather than stabilize (Zhang et al., 2024). For more on the hyperarousal side of the mitochondrial energy pattern, see Wired But Tired: When Your Mitochondria Can’t Power Down.
Fragmented sleep rarely has a single cause. In adults over 40, blood sugar crashes, cortisol rhythm disruption, hormonal changes, and weakened mitochondrial energy production might each be fragmenting sleep through different mechanisms — and the cellular stress documented above compounds across all of them.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Does Sleep Fragmentation Increase Inflammation?
In Nox2-knockout mice exposed to the same fragmentation approach that produced insulin resistance and adipose tissue inflammation in normal mice, all inflammatory markers remained at baseline — removing NOX2 removed the inflammatory cascade (Zhang et al., 2014). In humans with severe obstructive sleep apnea, malondialdehyde correlated directly with C-ROS-related protein, linking oxidative stress to body-wide inflammation within the same individuals (Asker et al., 2015). Inflammation from sleep fragmentation is both a consequence and a perpetuator — inflammatory cytokines including IL-1-beta and TNF-alpha are themselves sleep-fragmenting cues, creating a feedback loop.
What Is the Difference Between Sleep Fragmentation and Insomnia?
A 2024 EEG study demonstrated neurophysiological consequences of experimentally induced fragmentation in healthy volunteers who did not have insomnia — elevated cortical beta power during cognitive tasks and altered post-task neural activity, despite no insomnia classification (Benkirane et al., 2024). The distinction matters in practice: fragmented sleepers who build adequate total hours may not seek medical attention, even as cellular stress from repeated awakenings progresses.
Can You Recover From Years of Fragmented Sleep?
After adenotonsillectomy corrected obstructive sleep apnea in children, NOX2-derived peptide dropped from 38.2 to 22.4 pg/mL and flow-mediated dilation improved from 3.0% to 8.0% — vascular function recovered when the fragmentation source was removed (Loffredo et al., 2015). However, in adult mice, epigenetic marks (H3K27 acetylation at the Nox4 promoter) and associated liver and cardiac stress persisted after two weeks of recovery sleep (Guo et al., 2025). N-acetylcysteine rescued hippocampal parvalbumin interneurons from sleep-deprivation-induced oxidative stress (Gao et al., 2025), suggesting targeted antioxidant support during fragmentation can protect vulnerable cell populations even before the fragmentation source is resolved.
What Happens to Your Brain When You Do Not Sleep Well?
Two independent studies converge on oxidative stress in specific brain cell populations as the mechanistic bridge between sleep disruption and neurological stress. Hu et al. (2024) showed that sleep deprivation oxidizes mitochondrial DNA inside microglia, which release the stressd mitochondrial DNA as a danger cue that activates NF-kappaB-driven inflammation. Gao et al. (2025) showed that sleep deprivation concentrates oxidative stress in hippocampal parvalbumin interneurons, disrupting gamma oscillations required for memory precision — and N-acetylcysteine rescued both cell survival and memory function. For the full neurodegeneration evidence, see the section above on fragmented sleep and Alzheimer’s.
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep — the pillar article for metabolic sleep disruption, covering glucose regulation, cortisol rhythm, mitochondrial stress, NAD+ reduction, and fat metabolism
- Why Are You Exhausted But Can’t Sleep? Is It a Mitochondrial Energy Paradox? — mitochondrial fatigue, unrefreshing sleep, ATP production, and why rest may not restore cellular energy
- Do Your Mitochondria Control When You Sleep? and Why Does It Break Down After 40? — ROS sleep pressure, deep sleep reduction after 40, mitochondrial DNA aging, and sleep timing
- Can CoQ10 and PQQ Supplements Improve Your Sleep? — CoQ10, PQQ, magnesium, supplement timing, and the evidence for mitochondrial sleep support
- Is Your 3am Wakeup a Metabolic Emergency? Blood Sugar, Cortisol, and Mitochondrial reduction? — the 2-4am vulnerability window, glucose drops, cortisol rescue, and mitochondrial energy buffering
- Why Are You Wired but Tired, and What Role Do Mitochondria Play? — hyperarousal, cortisol rhythm changes, stress physiology, and mitochondrial power-down reduction
- How Does Exercise Rebuild Your Mitochondria and Fix Your Sleep? — exercise type, timing, mitochondrial biogenesis, PGC-1?, and sleep architecture
References
Asker, S., Asker, M., Sarikaya, E., Sunnetcioglu, A., Aslan, M., & Demir, H. (2015). Oxidative stress parameters and their correlation with clinical, metabolic and polysomnographic parameters in severe obstructive sleep apnea syndrome. International Journal of Clinical and Experimental Medicine, 8(7), 11449-11455. https://pubmed.ncbi.nlm.nih.gov/26379962/
Benkirane, O., Simor, P., Mairesse, O., & Peigneux, P. (2024). Sleep fragmentation modulates the neurophysiological correlates of cognitive fatigue. Clocks & Sleep, 6(4), 602-618. https://pubmed.ncbi.nlm.nih.gov/39449315/
Carreras, A., Zhang, S. X., Peris, E., Qiao, Z., Gileles-Hillel, A., Li, R. C., Wang, Y., & Gozal, D. (2014). Chronic sleep fragmentation induces endothelial impairment and structural vascular changes in mice. Sleep, 37(11), 1817-1824. https://pubmed.ncbi.nlm.nih.gov/25364077/
Gao, Y. Z., Liu, K., Wu, X. M., Shi, C. N., He, Q. L., Wu, H. P., Yang, J. J., Yao, H., & Ji, M. H. (2025). Oxidative stress-mediated reduction of hippocampal parvalbumin interneurons contributes to memory precision reduction after acute sleep deprivation. Molecular Neurobiology, 62(5), 5377-5394. https://pubmed.ncbi.nlm.nih.gov/39546120/
Guo, Y., Wang, J., Zhang, D., Tang, Y., Cheng, Q., Li, J., Gao, T., Zhang, X., Lu, G., Liu, M., Guan, X., Tang, X., & Gu, J. (2025). Diabetes-associated sleep fragmentation impairs liver and heart function via SIRT1-dependent epigenetic modulation of NADPH oxidase 4. Acta Pharmaceutica Sinica B, 15(3), 1480-1496. https://pubmed.ncbi.nlm.nih.gov/40370565/
Hu, Y., Wang, Y., Wang, Y., Zhang, Y., Wang, Z., Xu, X., Zhang, T., Zhang, T., Zhang, S., Hu, R., Shi, L., Wang, X., Li, J., Shen, H., Liu, J., Noda, M., Peng, Y., & Long, J. (2024). Sleep deprivation triggers mitochondrial DNA release in microglia to induce neural inflammation: Preventative effect of hydroxytyrosol butyrate. Antioxidants, 13(7), 833. https://pubmed.ncbi.nlm.nih.gov/39061901/
Loffredo, L., Zicari, A. M., Occasi, F., Perri, L., Carnevale, R., Angelico, F., Del Ben, M., Martino, F., Nocella, C., Savastano, V., Cesoni Marcelli, A., Duse, M., & Violi, F. (2015). Endothelial impairment and oxidative stress in children with sleep disordered breathing: Role of NADPH oxidase. Atherosclerosis, 240(1), 222-227. https://pubmed.ncbi.nlm.nih.gov/25805361/
Xie, Y., Ba, L., Wang, M., Deng, S. Y., Chen, S. M., Huang, L. F., Zhang, M., Wang, W., & Ding, F. F. (2020). Chronic sleep fragmentation shares similar pathogenesis with neurodegenerative diseases: Endosome-autophagosome-lysosome pathway impairment and microglia-mediated neuroinflammation. CNS Neuroscience & Therapeutics, 26(2), 215-227. https://pubmed.ncbi.nlm.nih.gov/31549780/
Zhang, S. X., Khalyfa, A., Wang, Y., Carreras, A., Hakim, F., Neel, B. A., Brady, M. J., Qiao, Z., Hirotsu, C., & Gozal, D. (2014). Sleep fragmentation promotes NADPH oxidase 2-mediated adipose tissue inflammation leading to insulin resistance in mice. International Journal of Obesity, 38(4), 619-624. https://pubmed.ncbi.nlm.nih.gov/23897221/
Zhang, W., Liu, D., Yuan, M., & Zhu, L. Q. (2024). The mechanisms of mitochondrial abnormalities that contribute to sleep disorders and related neurodegenerative diseases. Ageing Research Reviews, 97, 102307. https://pubmed.ncbi.nlm.nih.gov/38614368/
Written by Kat Fu, M.S., M.S.? Last reviewed: May 2026? 10 references cited
