The experience of crashing through the day but surging to alertness at bedtime carries consequences for brain health, cognitive function, and long-term longevity. This is not about sleep hygiene or screen habits. The wired-but-tired state is a condition where your stress-response architecture and your cellular energy production are working against each other.
This article covers hyperarousal physiology, the cortisol-mitochondria connection, the adrenal fatigue versus glucocorticoid resistance debate, and the hormonal changes that amplify the wired-but-tired pattern. It does not cover the fatigue-insomnia paradox (The Mitochondrial Energy Paradox), 3am timing mechanisms (The 3am Metabolic Emergency), supplements (CoQ10, PQQ, and Mitochondrial Supplements), or exercise approaches (How Exercise Rebuilds Your Mitochondria). For the full metabolic overview, see Metabolic Sleep Disruption.
Hyperarousal from cortisol-mitochondrial impairment is one of several metabolic causes of disrupted sleep; the pillar page maps the broader picture.
What Is Hyperarousal and Why Does It Keep Your Brain Alert at Night?
The assumption that insomnia is a sleep-time pattern does not match the evidence. Bonnet and Arand’s review defined chronic insomnia as a disorder of 24-hour central nervous activation hyperarousal, synthesizing convergent findings from four independent measurement domains: electroencephalography, hormonal profiling, metabolic imaging, and autonomic monitoring (Bonnet & Arand, 2010).
PET and SPECT brain imaging studies have found elevated regional and whole-body metabolic activation in people with insomnia compared to good sleepers. The brain does not disengage from its daytime energy expenditure. During sleep, people with insomnia show increased high-frequency EEG activation — the neurophysiological marker of a cortically aroused brain that has not transitioned into the low-frequency oscillations required for restorative sleep.
Abnormal cortisol and adrenocorticotropic hormone secretion patterns persist through both waking and sleeping hours in people with insomnia. Elevated resting heart rate and heightened sympathetic nervous activity tone continue during sleep, compounding the neuroendocrine and cardiovascular burden. The core finding is that in chronic insomnia, sleep pathways and arousal pathways run simultaneously — the brain is generating sleep-promoting activity while the stress-response architecture remains fully engaged. This dual activation is what produces the wired-but-tired experience.
A 15-day longitudinal study tracking 95 adults with 2,345 cortisol samples and EEG-measured sleep showed the within-person association between cortisol and sleep quality: higher pre-sleep cortisol predicted shorter total sleep time (p<0.001) and lower sleep efficiency (p<0.001) on the same night (Yap et al., 2024). Elevated evening cortisol is not a background phenomenon — it disrupts that sleep period.
Is "Wired but Tired" the Same as Adrenal Fatigue — Or Something Else?
The distinction between acute and chronic stress activation is central to understanding the wired-but-tired trajectory. A 2025 review detailed this divergence at the receptor level: under acute stress, cortisol appropriately restrains pro-inflammatory cytokines (interleukin-1 beta, interleukin-6, tumor necrosis factor-alpha) through glucocorticoid receptor pathways (Nunez et al., 2025). Under chronic stress, glucocorticoid receptor resistance develops — target tissues no longer respond to cortisol’s suppressive action despite continued or elevated cortisol secretion.
Glucocorticoid receptor resistance produces a paradoxical cellular outcome: circulating cortisol remains elevated or dysrhythmic while cortisol’s tissue-level anti-inflammatory effects are absent. The result is a chronic pro-inflammatory cytokine environment dominated by interleukin-6, tumor necrosis factor-alpha, and interleukin-17, with suppressed regulatory interleukin-10.
Morris and colleagues (2017) identified the mechanistic bridge between inflammation and adrenal suppression. Elevated tumor necrosis factor-alpha inhibits hypothalamic corticotropin-releasing hormone secretion, dampening the upstream hormonal drive to the adrenal cortex. At the same time, heightened nitric oxide production impairs steroidogenesis by inhibiting cytochrome P450 enzymes in adrenal mitochondria — a direct link between cellular oxidative stress and reduced cortisol-producing capacity. The wired-to-tired trajectory follows a predictable path: early hypothalamic-pituitary-adrenal hyperactivation (high cortisol, hyperarousal) gives way to late hypothalamic-pituitary-adrenal hypofunction (low cortisol, exhaustion) as oxidative mitochondrial stress builds.
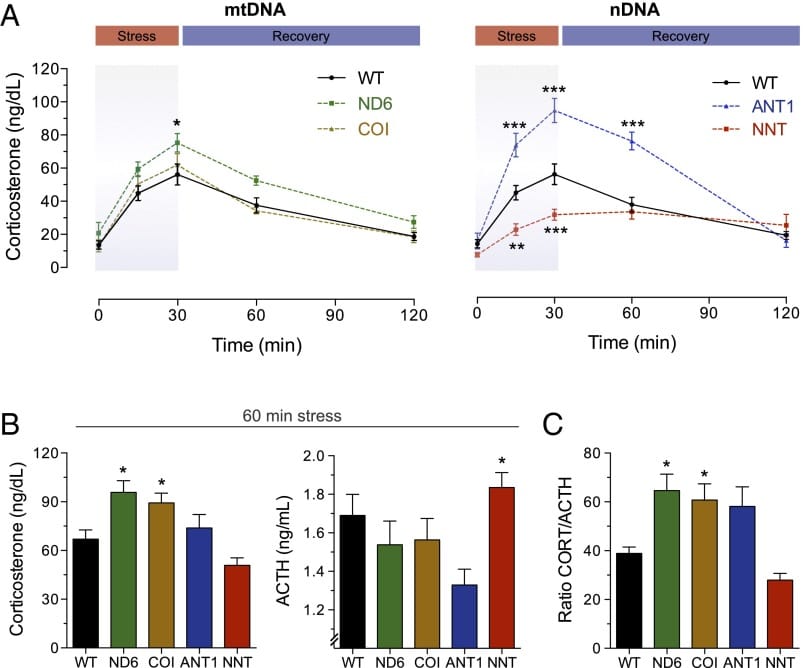
A PNAS study provided the direct evidence — in a mouse model — that mitochondrial function determines stress-hormone output. Using four mouse models with distinct mitochondrial defects, Picard and colleagues (2015) demonstrated that each mitochondrial defect produced a qualitatively distinct corticosterone response profile — hypothalamic-pituitary-adrenal axis output is tuned by cellular energetic status, not just psychological stress load. Mice deficient in nicotinamide nucleotide transhydrogenase (NNT) — an enzyme critical for maintaining the mitochondrial redox balance — showed the lowest corticosterone levels alongside paradoxically elevated adrenocorticotropic hormone (p<0.001 to p<0.02). Central ACTH was elevated while adrenal corticosterone output was suppressed — the adrenal glands were not converting the upstream hormonal cue into corticosterone production. Whether these animal findings translate proportionally to humans has not yet been established.
Can Chronic Stress Durably Alter Your Cortisol Rhythm?
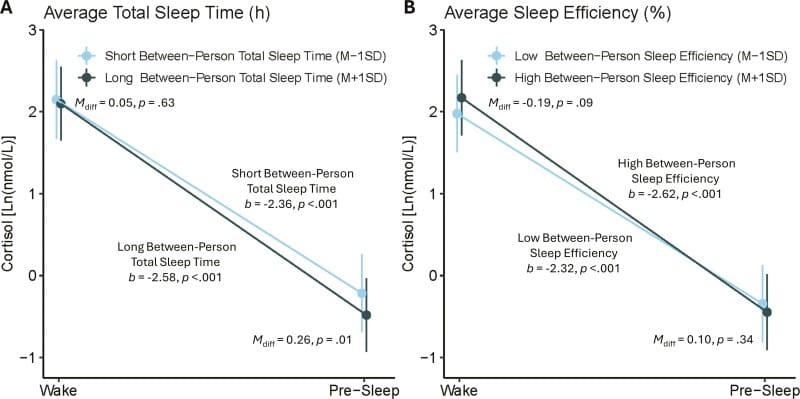
The 15-day intensive longitudinal study by Yap and colleagues (2024) examined cortisol-sleep associations across 2,345 cortisol samples at both the within-person and between-person levels. Higher pre-sleep cortisol predicted shorter sleep on the same night (p<0.001). Individuals who averaged shorter sleep across the study period had higher pre-sleep cortisol levels (p=0.01) — a between-person association suggesting that chronic short sleep co-occurs with cortisol dysregulation. Individuals who averaged lower sleep efficiency displayed flatter diurnal cortisol slopes (p<0.001), reflecting blunted morning-to-evening cortisol amplitude — a hallmark of chronic hypothalamic-pituitary-adrenal dysregulation.
Together, the findings converge: elevated pre-sleep cortisol impairs that sleep period, and people with chronically shorter sleep show both higher evening cortisol and flatter diurnal cortisol slopes. While the within-person data show that cortisol disrupts sleep, the longer-term association between habitual short sleep and cortisol dysregulation suggests these factors compound each other over time.
Sleep onset normally inhibits cortisol secretion, while each arousal and awakening stimulates cortisol release (Balbo et al., 2010). Fragmented sleep therefore generates repeated micro-activations of the hypothalamic-pituitary-adrenal axis across the night. Experimental reductions in sleep duration from eight to four hours produced a dose-response elevation in evening cortisol levels, showing that sleep quantity governs the degree of hypothalamic-pituitary-adrenal reactivation during the late-day window.
Velazquez Sanchez and Dalley reframed understanding of the cortisol awakening response in a 2025 review analyzing a microdialysis study of 201 healthy volunteers. The post-awakening cortisol rise is circadian in origin — not triggered by the act of waking up (Velazquez Sanchez & Dalley, 2025). Disrupted sleep timing therefore dysregulates the entire daily cortisol architecture, not merely the morning response. Salivary cortisol awakening response approaches, widely used as markers of hypothalamic-pituitary-adrenal reactivity, may be measuring circadian continuation rather than a discrete stress event. Full diurnal cortisol profiling is needed to characterize hypothalamic-pituitary-adrenal function in people with the wired-but-tired pattern.
Does Perimenopause Amplify the Wired-But-Tired Pattern?
Estrogen normally buffers cortisol reactivity. In a randomized trial in postmenopausal women found that estradiol therapy blunted cortisol responses to an acute stressor compared to placebo (p = 0.017), possibly through increased corticosteroid-binding globulin that may reduce bioavailable cortisol (Herrera et al., 2017). During perimenopause, lower estrogen removes this dampening effect, increasing the cortisol response to stressors that previously would have been contained. This elevated cortisol reactivity overlays onto already-building mitochondrial oxidative stress — tumor necrosis factor-alpha and oxidative stress progressively suppress adrenal function over time (Morris et al., 2017).
The "second wind" phenomenon maps to cortisol physiology. Sleep onset normally inhibits cortisol secretion (Balbo et al., 2010). When cortisol remains elevated at night — due to chronic hypothalamic-pituitary-adrenal dysregulation associated with estrogen withdrawal — the normal inhibitory transition does not occur. The circadian cortisol nadir (typically between 11pm and 2am) does not reach its required low point, generating a paradoxical alertness surge at the hour when the body should be entering its deepest sleep.
Sarnataro and colleagues identified a cellular mechanism underlying sleep pressure itself in a 2025 Nature study: in Drosophila, mitochondrial dynamics in sleep-control neurons regulate the buildup of sleep need — hyperfused mitochondria increase sleep pressure, while fragmented mitochondria decrease it (Sarnataro et al., 2025). The mechanism involves an electron surplus during waking that generates ROS, cueing the need for sleep. If this mechanism is conserved in mammals, mitochondria that are already impaired from chronic stress, aging, and hormonal changes could disrupt normal sleep-pressure pathway activity at the organelle level. This connection remains to be shown in humans.
Sleep deprivation compounds the perimenopausal inflammatory state through a mitochondrial pathway identified in animal research. A 2024 study in rats demonstrated that sleep deprivation oxidizes mitochondrial DNA in microglia — the brain’s immune cells — triggering cytoplasmic mitochondrial DNA release (Hu et al., 2024). This released mitochondrial DNA activates NF-kB transcription, producing pro-inflammatory cytokines and establishing a neuroinflammatory cascade. Reducing the oxidative stress that triggers mitochondrial DNA release attenuated the inflammatory cascade and improved cognitive performance in the same study — supporting mitochondrial oxidative stress as a key driver in this model.
Hyperarousal, cortisol dysregulation, and mitochondrial impairment might not be the only factors disrupting your sleep. Many people with the wired-but-tired pattern also have overlapping metabolic, hormonal, or inflammatory processes that compound one another. When multiple causes are active, the experience of exhaustion paired with bedtime alertness might reflect more than one mechanism at the same time.
Find out which causes might be driving your 3am wakeups →
What Is Hypothalamic-Pituitary-Adrenal Axis Dysregulation and How Do You Know If You Have It?
The hypothalamic-pituitary-adrenal axis is a three-node feedback loop. The hypothalamus releases corticotropin-releasing hormone, which prompts the pituitary to release adrenocorticotropic hormone, which stimulates the adrenal cortex to produce cortisol. Cortisol then feeds back to suppress both hypothalamic and pituitary output, completing the negative feedback loop.
Under chronic stress, the feedback breaks at the glucocorticoid receptor level. Target tissues develop resistance to cortisol’s suppressive action, and the normal restraining influence of cortisol on hypothalamic and pituitary output weakens (Nunez et al., 2025). The result is a feedback loop that continues producing cortisol but has reduced the ability to self-regulate — the loop runs open.
Traditional testing often misses this dysregulation. A single morning cortisol draw captures one point on a curve that should span 24 hours. The 2025 review established that the post-awakening cortisol rise reflects circadian rhythm continuation rather than a discrete waking event (Velazquez Sanchez & Dalley, 2025), meaning morning cortisol values alone cannot distinguish between normal circadian function and dysregulated hypothalamic-pituitary-adrenal output.
Why Does Your Brain Get a Second Wind Right Before Bed?
Normal cortisol physiology follows a predictable pattern: cortisol peaks within the first hour after waking, reductions through the day, and reaches its 24-hour low point in the late evening and early night hours. Sleep onset reinforces cortisol suppression — the transition from wakefulness to sleep actively inhibits adrenal cortisol secretion (Balbo et al., 2010).
In people with chronic hypothalamic-pituitary-adrenal dysregulation, this sleep-onset cortisol inhibition does not occur. Cortisol remains elevated through the evening window, and the brain registers this as a cue to maintain alertness. The dose-response relationship is documented: experimental reduction from eight hours of sleep to four hours elevated evening cortisol levels in the subsequent days, meaning each night of shortened sleep raises the cortisol floor for the following evening.
The second wind is distinct from circadian phase delay, which is a clock-timing pattern. In circadian phase delay, the entire sleep-wake cycle moves later, but the internal cortisol architecture remains intact relative to the adjusted schedule. In hypothalamic-pituitary-adrenal dysregulation, the cortisol curve itself loses its amplitude and does not reduction appropriately, regardless of when the person goes to bed.
Can Sleep Deprivation Cause Brain Inflammation Through Mitochondrial Stress?
In the rat model, the inflammatory cascade proceeds in a defined sequence: sleep deprivation induces mitochondrial oxidative stress in microglia, oxidative stress stresses mitochondrial DNA within microglia, stressd mitochondrial DNA is released into the cytoplasm, cytoplasmic mitochondrial DNA triggers NF-kB transcriptional activity, and NF-kB produces pro-inflammatory cytokines that establish neuroinflammation across surrounding brain tissue (Hu et al., 2024).
This cascade is self-amplifying: neuroinflammation itself impairs sleep quality, which drives further mitochondrial oxidative stress, which releases more mitochondrial DNA, which sustains the inflammatory state. Animals subjected to sleep deprivation in the same study showed cognitive impairment on behavioral testing, consistent with mitochondrial-stress-driven neuroinflammation disrupting hippocampal memory consolidation.
For the wired-but-tired experience, the connection is plausible but not yet shown in humans. Neuroinflammation from mitochondrial stress may sustain arousal by activating immune-alert pathways in the brain — the same pathways that evolved to keep organisms awake during infection. The brain would interpret the inflammatory input as a reason to remain vigilant, even as the body registers exhaustion from the energy impact of sustained immune activation. For more on the measured cellular consequences of interrupted sleep, see Sleep Fragmentation and Cellular Stress.
How Do You Lower Cortisol at Night to Sleep?
Yap and colleagues documented a cortisol-sleep association — where elevated cortisol impairs sleep, and chronic short sleep co-occurs with cortisol dysregulation — helps explain why single-step approaches to pre-sleep cortisol often do not work. Reducing cortisol on one evening does not break the cortisol-sleep pattern if the underlying drivers — hypothalamic-pituitary-adrenal dysregulation, glucocorticoid receptor resistance, mitochondrial oxidative stress — remain active.
Consistent sleep timing preserves the circadian cortisol architecture identified in the 2025 review (Velazquez Sanchez & Dalley, 2025). Because the post-awakening cortisol rise is circadian rather than waking-triggered, irregular sleep timing disrupts the entire daily cortisol curve, not just the morning response. Maintaining consistent bed and wake times stabilizes the circadian framework within which cortisol is regulated.
Stress-response work that targets autonomic tone — rather than cognitive relaxation alone — addresses the 24-hour hyperarousal state identified by Bonnet and Arand (2010). Hyperarousal involves sympathetic nervous activity activation, not just racing thoughts; approaches that reduce sympathetic tone may be more effective than thought-based relaxation techniques for this pattern.
For evidence on mitochondrial biogenesis through exercise, see How Exercise Rebuilds Your Mitochondria and Fixes Your Sleep. For evidence on mitochondrial support compounds, see CoQ10, PQQ, and Mitochondrial Supplements.
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep — the pillar article for metabolic sleep disruption, covering glucose regulation, cortisol rhythm, mitochondrial stress, NAD+ reduction, and fat metabolism
- Why Are You Exhausted But Can’t Sleep? Is It a Mitochondrial Energy Paradox? — mitochondrial fatigue, unrefreshing sleep, ATP production, and why rest may not restore cellular energy
- Do Your Mitochondria Control When You Sleep? and Why Does It Break Down After 40? — ROS sleep pressure, deep sleep reduction after 40, mitochondrial DNA aging, and sleep timing
- Can CoQ10 and PQQ Supplements Improve Your Sleep? — CoQ10, PQQ, magnesium, supplement timing, and the evidence for mitochondrial sleep support
- Is Your 3am Wakeup a Metabolic Emergency? Blood Sugar, Cortisol, and Mitochondrial reduction? — the 2-4am vulnerability window, glucose drops, cortisol rescue, and mitochondrial energy buffering
- How Does Exercise Rebuild Your Mitochondria and Fix Your Sleep? — exercise type, timing, mitochondrial biogenesis, PGC-1?, and sleep architecture
- What Is the Measured Cellular Impact of Sleep Fragmentation? — fragmented sleep, oxidative stress, vascular injury, inflammation, and cellular aging impact
References
Balbo, M., Leproult, R., & Van Cauter, E. (2010). Impact of sleep and its disturbances on hypothalamo-pituitary-adrenal axis activity. International Journal of Endocrinology, 2010, 759234. https://pubmed.ncbi.nlm.nih.gov/20628523/
Bonnet, M. H., & Arand, D. L. (2010). Hyperarousal and insomnia: State of the science. Sleep Medicine Reviews, 14(1), 9–15. https://pubmed.ncbi.nlm.nih.gov/19640748/
Dressle, R. J., Feige, B., Spiegelhalder, K., Schmucker, C., Benz, F., Mey, N. C., & Riemann, D. (2022). HPA axis activity in individuals with chronic insomnia: A review and meta-analysis of case-control studies. Sleep Medicine Reviews, 62, 101588. https://pubmed.ncbi.nlm.nih.gov/35091194/
Herrera, A. Y., Hodis, H. N., Mack, W. J., & Mather, M. (2017). Estradiol therapy after menopause mitigates effects of stress on cortisol and working memory. The Journal of Clinical Endocrinology & Metabolism, 102(12), 4457–4466. https://pubmed.ncbi.nlm.nih.gov/29106594/
Hu, Y., Wang, Y., Wang, Y., Zhang, Y., Wang, Z., Xu, X., Zhang, T., Zhang, T., Zhang, S., Hu, R., Shi, L., Wang, X., Li, J., Shen, H., Liu, J., Noda, M., Peng, Y., & Long, J. (2024). Sleep deprivation triggers mitochondrial DNA release in microglia to induce neural inflammation: Preventative effect of hydroxytyrosol butyrate. Antioxidants, 13(7), 833. https://pubmed.ncbi.nlm.nih.gov/39061901/
Morris, G., Anderson, G., & Maes, M. (2017). Hypothalamic-pituitary-adrenal hypofunction in myalgic encephalomyelitis (ME)/chronic fatigue syndrome (CFS) as a consequence of activated immune-inflammatory and oxidative and nitrosative pathways. Molecular Neurobiology, 54(9), 6806–6819. https://pubmed.ncbi.nlm.nih.gov/27766535/
Nunez, S. G., Rabelo, S. P., Subotic, N., Caruso, J. W., & Knezevic, N. N. (2025). Chronic stress and autoimmunity: The role of HPA axis and cortisol dysregulation. International Journal of Molecular Sciences, 26(20), 9994. https://pubmed.ncbi.nlm.nih.gov/41155288/
Picard, M., McManus, M. J., Gray, J. D., Nasca, C., Moffat, C., Kopinski, P. K., Seifert, E. L., McEwen, B. S., & Wallace, D. C. (2015). Mitochondrial functions modulate neuroendocrine, metabolic, inflammatory, and transcriptional responses to acute psychological stress. Proceedings of the National Academy of Sciences, 112(48), E6614–E6623. https://pubmed.ncbi.nlm.nih.gov/26627253/
Sarnataro, R., Velasco, C. D., Monaco, N., Kempf, A., & Miesenböck, G. (2025). Mitochondrial origins of the pressure to sleep. Nature, 645(8081), 722–728. https://pubmed.ncbi.nlm.nih.gov/40670797/
Velazquez Sanchez, C., & Dalley, J. W. (2025). The cortisol awakening response: Fact or fiction? Brain and Neuroscience Advances, 9, 23982128251327712. https://pubmed.ncbi.nlm.nih.gov/40297522/
Yap, Y., Tung, N. Y. C., Shen, L., Bei, B., Phillips, A., & Wiley, J. F. (2024). Daily associations between salivary cortisol and electroencephalographic-assessed sleep: A 15-day intensive longitudinal study. Sleep, 47(9), zsae087. https://pubmed.ncbi.nlm.nih.gov/38587464/
Written by Kat Fu, M.S., M.S.? Last reviewed: May 2026? 11 references cited
