Millions of people describe the same paradox: bone-deep exhaustion that does not resolve with more time in bed. The pattern points beyond willpower or sleep hygiene. Emerging research points to mitochondria — the organelles responsible for producing approximately 90% of cellular energy — as a central driver. When mitochondrial adenosine triphosphate production breaks down, the deficit directly undermines the brain’s ability to generate and maintain deep sleep, with downstream consequences for cognitive function, metabolic health, and biological aging.
This article covers the experience of unrefreshing sleep and persistent fatigue from a mitochondrial perspective — what the research shows, how mitochondrial fatigue differs from ordinary tiredness, and what testing exists. For the full metabolic cause overview, see Metabolic Sleep Disruption. Mitochondrial impairment is one of several interconnected causes of sleep disruption. Not everyone with fatigue has a mitochondrial pattern, but for those who do, the pattern is distinctive and measurable.
Why Doesn’t Rest Fix Mitochondrial Fatigue?
The assumption behind "just get more sleep" is that the energy-production machinery works and needs time to run. A 2009 study by Myhill, Booth, and McLaren-Howard directly challenged that assumption. The researchers recruited 71 individuals with chronic fatigue syndrome and 53 healthy controls, then measured five biochemical factors related to mitochondrial adenosine triphosphate production and transport. The correlation between mitochondrial impairment severity and illness severity (measured by the Bell Ability Scale) reached P<0.001. Only one of 71 CFS participants’ results overlapped with the normal control range — giving the test near-complete discriminatory power in this cohort (Myhill et al., 2009).
That finding establishes the cellular deficit, but why does sleep not correct it? A 2010 study by Dworak and colleagues demonstrated the mechanism: sleep triggers a region-specific adenosine triphosphate surge in wake-active brain areas, including the basal forebrain and frontal cortex. The magnitude of that surge correlates with non-rapid eye movement delta activity — the deepest phase of restorative sleep. Sleep deprivation fully prevents the surge (Dworak et al., 2010). The restorative function of sleep is the adenosine triphosphate recharge. If mitochondria cannot execute oxidative phosphorylation efficiently, the sleep-triggered adenosine triphosphate surge is blunted or absent, meaning subjective rest occurs without the cellular restitution that makes rest feel restorative.
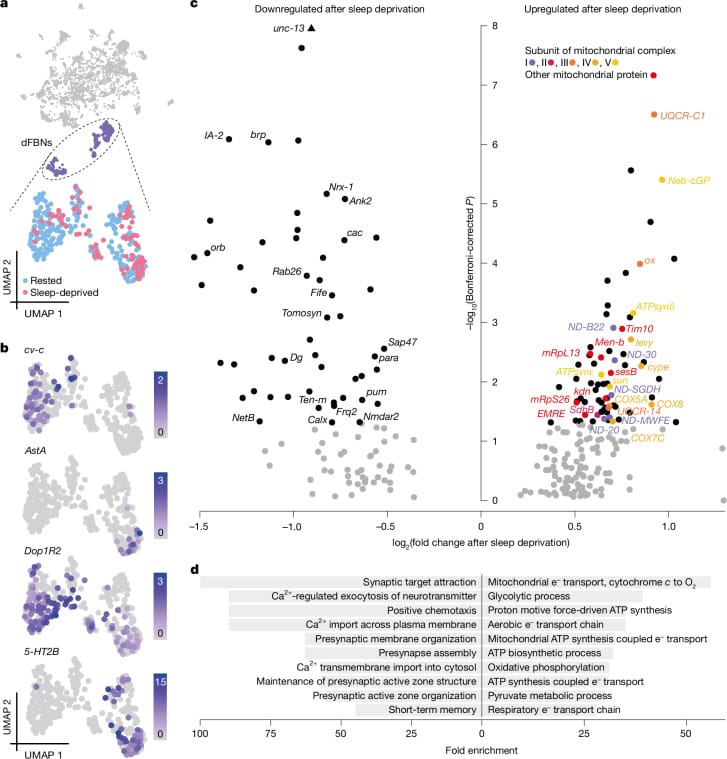
A 2025 Nature study by Sarnataro and colleagues provided evidence that these findings extend to the fundamental physiology of sleep pressure. Using single-cell transcriptomics in Drosophila sleep-control neurons, the researchers found that genes upregulated after sleep deprivation encoded "alexclusively proteins with roles in mitochondrial respiration and ATP synthesis" (Sarnataro et al., 2025). This was a fruit fly study, so direct translation to human sleep regulation requires further showation, but the conserved mitochondrial physiology is notable. Sleep deprivation stressd mitochondrial morphology — reducing size, elongation, and branching — in these neurons, consistent with a self-reinforcing trap documented by Zhang and colleagues in a 2024 review of the bidirectional sleep-mitochondria stress loop (Zhang et al., 2024).
What Does Mitochondrial Fatigue Feel Like Compared to Normal Tiredness?
The distinction between ordinary sleep-debt tiredness and mitochondrial energy impairment matters because the two conditions require fundamentally different responses. A 2020 proteomic study by Sweetman and colleagues used SWATH-MS (Sequential Window Acquisition of all Theoretical Mass Spectra) to profile peripheral blood mononuclear cells from 11 ME/CFS participants and 9 age- and sex-matched healthy controls. Nine of eleven ME/CFS participants clustered distinctly from all controls in unsupervised proteomic analysis. Comparative analysis identified 99 differentially expressed proteins, with notable enrichment mapping to mitochondrial energy production and oxidative stress pathways (Sweetman et al., 2020). The proteomic signature of cellular energy impairment is visible in a standard blood draw.
A separate 2020 study added an unexpected dimension. Martucci and colleagues enrolled post-menopausal women classified with either objective insomnia (showed by actigraphy showing sleep efficiency below 85%) or paradoxical insomnia (reported sleep complaints with normal sleep efficiency on actigraphy). Both groups showed the same altered levels of the mitokines FGF21 and Humanin — mitochondrial stress markers — compared to age-matched controls. No difference existed between the objective and paradoxical insomnia groups on any mitochondrial stress marker (Martucci et al., 2020). Even the perception of poor sleep activates mitochondrial stress responses, making "it’s all in your head" biologically inaccurate.
The recognizable pattern of mitochondrial fatigue includes post-exertional malaise (lowerning after modest activity), cognitive impairment disproportionate to sleep reduction, and morning fatigue identical to bedtime fatigue. A 2014 mechanistic review by Morris and Maes explained the connection — pro-inflammatory cytokines including TNF-alpha, combined with oxidative and nitrosative stress, may inhibit electron transport chain activity, reducing adenosine triphosphate output (Morris & Maes, 2014). Inflammation common in chronic conditions drives the energy-production impairment responsible for persistent exhaustion.
Does Unrefreshing Sleep Mean Your Mitochondria Are Underperforming?
The 2025 Nature study by Sarnataro and colleagues represents the strongest evidence to date that mitochondrial function is inseparable from sleep regulation — though importantly, this work was conducted in fruit flies (Drosophila), where sleep-control neurons can be precisely manipulated. Using optogenetics and adenosine triphosphate sensors in sleep-control neurons, the researchers demonstrated that preventing mitochondrial fission increased sleep duration, while inducing mitochondrial fission decreased sleep duration. Installing an artificial electron-overflow mechanism that dissipated excess electron pressure from the respiratory chain relieved sleep pressure without actual sleep — showing that mitochondrial electron transport chain status is itself the sleep-regulatory mechanism in this model. The authors concluded that "sleep may be an inescapable consequence of aerobic metabolism" (Sarnataro et al., 2025). Whether these findings translate directly to mammalian sleep regulation remains to be shown, but the underlying mitochondrial physiology is conserved across species.
A 2025 NIH review by Syed and colleagues identified a molecular driver of the energy impairment in humans: WASF3 (Wiskott-Aldrich syndrome protein family member 3). Under endoplasmic reticulum stress, WASF3 is overproduced and localizes to mitochondria, where it physically disrupts the assembly of respiratory supercomplexes III and IV, directly blocking oxidative phosphorylation. Simultaneously, WASF3 promotes actin polymerization, which further suppresses mitochondrial respiration while upregulating glycolysis — a metabolic change that may support short-term immune activation but leads to chronic energy deficiency when sustained. The resulting impairment of oxidative phosphorylation reduces cellular adenosine triphosphate generation (Syed et al., 2025).
The vicious cycle involves multiple reinforcing mechanisms. Sleep deprivation fragments mitochondria. Fragmented mitochondria impair cellular energy production and may disrupt melatonin synthesis — pineal gland melatonin production depends on mitochondrial energy supply and is sensitive to oxidative stress (Zhang et al., 2024). Each disrupted night compounds the cellular stress from the previous one. A 2024 study by Davinelli and colleagues identified an additional arm of the cycle: sleep restriction increases cellular availability of Cul3, a component of the NRF2 ubiquitination complex. Increased Cul3 sequesters NRF2 — the master regulator of antioxidant gene expression — and prevents NRF2 from activating antioxidant genes including HO-1, NQO1, and glutathione-synthesizing enzymes (Davinelli et al., 2024). With NRF2 suppressed, the cellular defense against oxidative stress from malfunctioning mitochondria is removed, accelerating the deterioration.
Can Mitochondrial Impairment Be Measured or Tested?
The adenosine triphosphate profile test developed in the Myhill 2009 study measures five biochemical parameters from a blood sample: adenosine triphosphate concentration, the ratio of adenosine triphosphate available with endogenous magnesium, oxidative phosphorylation recycling efficiency, and two measures of adenosine triphosphate transport across the mitochondrial membrane. In the study cohort, near-complete separation between chronic fatigue syndrome participants and controls made the test a reliable discriminator of mitochondrial energy impairment (Myhill et al., 2009). Specialized laboratories offer this testing.
For direct tissue-level evidence, a 2024 cross-sectional study by Bizjak and colleagues compared skeletal muscle mitochondria across three groups: 15 post-COVID syndrome participants, 15 chronic fatigue syndrome participants, and 13 healthy controls, using both electron microscopy and high-resolution respirometry. The findings revealed mechanistically distinct conditions: chronic fatigue syndrome participants showed more severe morphological degradation — degraded cristae and structural disorganization consistent with long-term cellular stress — while post-COVID syndrome participants showed reduced Complex I oxidative phosphorylation capacity with better-preserved mitochondrial structure (Bizjak et al., 2024). The distinction matters because these conditions converge on fatigue as a shared outcome but may require different measurement and measurement approaches.
A review by Filler and colleagues examined 25 studies and identified coenzyme Q10 as the consistently dysregulated mitochondrial marker across fatigue conditions studied, while carnitine abnormalities were frequently reported though with variable patterns. Low coenzyme Q10 correlated with fatigue severity whenever measured, and carnitine abnormalities were reported in every study that investigated carnitine, though the specific impairments varied across studies (Filler et al., 2014). Both are commercially available blood tests that provide indirect but accessible evidence of mitochondrial status.
What is available now includes organic acids testing, coenzyme Q10 and carnitine levels, and specialized adenosine triphosphate profiles through select laboratories. Blood-based proteomic profiling — where 9 of 11 ME/CFS participants clustered distinctly from all controls (Sweetman et al., 2020) — and high-resolution respirometry remain primarily research tools, but adoption is expanding.
Mitochondrial impairment rarely acts alone. Blood sugar instability, cortisol rhythm disruption, hormonal changes, and inflammation each compound the energy deficit — and many people dealing with unrefreshing sleep have more than one of these causes active simultaneously. The pattern you are experiencing might be primarily mitochondrial, primarily metabolic, or a combination that requires a different approach than targeting any single cause.
Find out which causes might be driving your 3am wakeups →
Why Am I So Tired But My Body Will Not Let Me Sleep?
During normal waking hours, adenosine builds as a byproduct of adenosine triphosphate consumption and drives sleep pressure — the increasing urge to sleep that builds throughout the day. This process depends on functional mitochondrial adenosine triphosphate cycling. When mitochondria are stressd, adenosine-mediated sleep drive may be blunted even as subjective exhaustion intensifies (Sarnataro et al., 2025 — shown in Drosophila; human showation pending). The sensation of being "wired but tired" — explored in detail in Wired But Tired: When Your Mitochondria Can’t Power Down — reflects this decoupling: the body’s energy-sensing pathways register depletion, but the sleep-initiating circuitry does not receive the mitochondrial-driven processes required to consolidate restorative sleep.
Can Chronic Fatigue Syndrome Be Caused by Mitochondrial Patterns?
Whether mitochondrial impairment is the initiating cause of ME/CFS or a downstream consequence of immune activation remains an active research question, but the energy deficit is measurable and consistent across study cohorts (Syed et al., 2025). Chronic fatigue syndrome and post-COVID fatigue show distinct mitochondrial pathology at the tissue level. In the 2024 biopsy study by Bizjak and colleagues, chronic fatigue syndrome participants showed more severe structural degradation of mitochondrial cristae, while post-COVID participants showed more functional impairment of Complex I oxidative phosphorylation with better-preserved morphology (Bizjak et al., 2024). The distinction suggests these conditions may require different approaches despite sharing fatigue as their defining experience.
Can Poor Sleep Create a Cycle That Makes Fatigue Progressively Lower?
The NRF2 suppression mechanism identified by Davinelli and colleagues adds a third arm to the cycle beyond the direct sleep-mitochondria-sleep loop. Sleep restriction blocks the master antioxidant regulator NRF2 through increased Cul3-mediated ubiquitination, removing cellular protection against oxidative stress from malfunctioning mitochondria (Davinelli et al., 2024). Oxidative stress further degrades mitochondrial DNA and membrane integrity, which further impairs sleep architecture — a compounding deterioration that does not self-correct without targeted support. For more on the measurable consequences of fragmented sleep on cellular health, see What Is the Measured Cellular Impact of Sleep Fragmentation? (Zhang et al., 2024).
What Is Unrefreshing Sleep and What Causes It?
The Dworak 2010 finding that sleep’s restorative function depends on an adenosine triphosphate surge in wake-active brain regions during non-rapid eye movement sleep provides the mechanistic explanation: if the mitochondrial machinery is impaired, the adenosine triphosphate surge is blunted (Dworak et al., 2010). The electroencephalogram may look normal while cellular restoration does not occur. The disconnect between sleep duration and sleep quality in mitochondrial impairment explains why standard sleep guidance — consistent bedtime, dark room, limited screens — does not resolve the fatigue. Those steps optimize sleep conditions but do not repair the energy-production machinery that makes sleep restorative (Martucci et al., 2020).
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep — the pillar article for metabolic sleep disruption, covering glucose regulation, cortisol rhythm, mitochondrial stress, NAD+ reduction, and fat metabolism
- Do Your Mitochondria Control When You Sleep? and Why Does It Break Down After 40? — ROS sleep pressure, deep sleep reduction after 40, mitochondrial DNA aging, and sleep timing
- Can CoQ10 and PQQ Supplements Improve Your Sleep? — CoQ10, PQQ, magnesium, supplement timing, and the evidence for mitochondrial sleep support
- Is Your 3am Wakeup a Metabolic Emergency? Blood Sugar, Cortisol, and Mitochondrial reduction? — the 2-4am vulnerability window, glucose drops, cortisol rescue, and mitochondrial energy buffering
- Why Are You Wired but Tired, and What Role Do Mitochondria Play? — hyperarousal, cortisol rhythm changes, stress physiology, and mitochondrial power-down reduction
- How Does Exercise Rebuild Your Mitochondria and Fix Your Sleep? — exercise type, timing, mitochondrial biogenesis, PGC-1?, and sleep architecture
- What Is the Measured Cellular Impact of Sleep Fragmentation? — fragmented sleep, oxidative stress, vascular injury, inflammation, and cellular aging impact
References
Bizjak, D. A., Ohmayer, B., Buhl, J. L., Schneider, E. M., Walther, P., Calzia, E., Jerg, A., Matits, L., & Steinacker, J. M. (2024). Functional and Morphological Differences of Muscle Mitochondria in Chronic Fatigue Syndrome and Post-COVID Syndrome. International Journal of Molecular Sciences, 25(3), 1675. https://pubmed.ncbi.nlm.nih.gov/38338957/
Davinelli, S., Medoro, A., Savino, R., & Scapagnini, G. (2024). Sleep and Oxidative Stress: Current Perspectives on the Role of NRF2. Cellular and Molecular Neurobiology, 44(1), 52. https://pubmed.ncbi.nlm.nih.gov/38916679/
Dworak, M., McCarley, R. W., Kim, T., Kalinchuk, A. V., & Basheer, R. (2010). Sleep and brain energy levels: ATP changes during sleep. The Journal of Neuroscience, 30(26), 9007-9016. https://pubmed.ncbi.nlm.nih.gov/20592221/
Filler, K., Lyon, D., Bennett, J., McCain, N., Elswick, R., Lukkahatai, N., & Saligan, L. N. (2014). Association of Mitochondrial impairment and Fatigue: A Review of the Literature. BBA Clinical, 1, 12-23. https://pubmed.ncbi.nlm.nih.gov/25147756/
Martucci, M., Conte, M., Ostan, R., Chiariello, A., Miele, F., Franceschi, C., Salvioli, S., Santoro, A., & Provini, F. (2020). Both objective and paradoxical insomnia elicit a stress response involving mitokine production. Aging, 12(11), 10497-10505. https://pubmed.ncbi.nlm.nih.gov/32420904/
Morris, G., & Maes, M. (2014). Mitochondrial impairments in myalgic encephalomyelitis/chronic fatigue syndrome explained by activated immuno-inflammatory, oxidative and nitrosative stress pathways. Metabolic Brain Disease, 29(1), 19-36. https://pubmed.ncbi.nlm.nih.gov/24557875/
Myhill, S., Booth, N. E., & McLaren-Howard, J. (2009). Chronic fatigue syndrome and mitochondrial impairment. International Journal of Clinical and Experimental Medicine, 2(1), 1-16. https://pubmed.ncbi.nlm.nih.gov/19436827/
Sarnataro, R., Velasco, C. D., Monaco, N., Kempf, A., & Miesenböck, G. (2025). Mitochondrial origins of the pressure to sleep. Nature, 645(8081), 722-728. https://pubmed.ncbi.nlm.nih.gov/40670797/
Sweetman, E., Kleffmann, T., Edgar, C., de Lange, M., Vallings, R., & Tate, W. P. (2020). A SWATH-MS analysis of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome peripheral blood mononuclear cell proteomes reveals mitochondrial impairment. Journal of Translational Medicine, 18(1), 365. https://pubmed.ncbi.nlm.nih.gov/32972442/
Syed, A. M., Karius, A. K., Ma, J., Wang, P. Y., & Hwang, P. M. (2025). Mitochondrial impairment in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Physiology (Bethesda, Md.), 40(4), 0. https://pubmed.ncbi.nlm.nih.gov/39960432/
Zhang, W., Liu, D., Yuan, M., & Zhu, L. Q. (2024). The mechanisms of mitochondrial abnormalities that contribute to sleep disorders and related neurodegenerative diseases. Ageing Research Reviews, 97, 102307. https://pubmed.ncbi.nlm.nih.gov/38614368/
Written by Kat Fu, M.S., M.S.? Last reviewed: May 2026? 11 references cited
