Millions of people now take semaglutide (Ozempic, Wegovy) or tirzepatide (Mounjaro, Zepbound) for weight reduction or type 2 diabetes. A growing number report difficulty falling or staying asleep — a complaint that does not appear on the prescribing label but dominates user discussion forums. The gap between the official side-effect profile and the lived experience calls for a careful look at what the research says.
This article covers three things: the mechanistic case for why GLP-1 drugs might cause insomnia, the user-reporting data that quantifies how common sleep complaints are, and the controlled-trial evidence showing these same drugs improve sleep apnea. It does not cover GLP-1 dosing or weight-management approaches.
GLP-1 drugs alter the same appetite-hormone axis — including leptin and orexin — that drives metabolic sleep disruption more broadly. The parent pillar on metabolic sleep disruption covers that full picture.
Why Would a Weight Loss Drug Affect Sleep?
How Does GLP-1 Directly Activate Orexin Neurons?
The earliest mechanistic evidence comes from a 2004 electrophysiology study by Acuna-Goycolea and van den Pol. Their experiments showed GLP-1 excites orexin neurons through two distinct pathways. The direct pathway: GLP-1 depolarizes orexin neurons and increases their firing rate through a G-protein-dependent, sodium-dependent conductance. The indirect pathway: GLP-1 potentiates glutamate release from the excitatory axons that feed into orexin neurons, amplifying their activation from upstream inputs (Acuna-Goycolea & van den Pol, 2004).
Two additional findings from that study matter. GLP-1 decreased afterhyperpolarization currents in orexin neurons — the electrical “cooldown” that normally limits how long a neuron fires. Reduced afterhyperpolarization means orexin neurons stay active longer per activation event. And GLP-1 had no detectable effect on melanin-concentrating hormone (MCH) neurons — the opposing, sleep-promoting population located in the same brain region. The selectivity is notable: GLP-1 activates the wake side without engaging the sleep side.

Why Does Orexin Matter for Sleep?
A 2009 review by Tsujino and Sakurai established it as the central integrator linking sleep-wake regulation, energy balance, and reward. Orexin deficiency causes narcolepsy in humans, dogs, and rodents, supporting orexin as necessary for consolidated wakefulness. Orexin neurons receive direct input from peripheral metabolic cues including leptin, ghrelin, and glucose, making them responsive to the same metabolic environment that GLP-1 drugs reshape (Tsujino & Sakurai, 2009).
A 2022 review by Pizza et al. traces the pharmaceutical extension of this science: FDA-approved orexin receptor antagonists (suvorexant, lemborexant) now exist to block orexin for insomnia. These drugs reduce arousal by inhibiting the orexin receptors. If GLP-1 excites orexin neurons, as the electrophysiology data shows, then GLP-1 receptor agonists are doing the pharmacological opposite of these insomnia drugs — promoting wakefulness rather than suppressing it (Pizza et al., 2022).
What Other Mechanisms Could GLP-1 Drugs Use to Disrupt Sleep?
Beyond the orexin pathway, at least three additional mechanisms could contribute to sleep disruption on GLP-1 drugs. Gastrointestinal side effects — nausea in particular — tend to worsen in the supine position at night. Reduced evening food intake may lower glycogen stores enough to trigger a cortisol-mediated arousal response in the second half of the night. And for people with diabetes on combination therapy, nocturnal hypoglycemia remains a possibility, though semaglutide monotherapy carries a low hypoglycemia likelihood.
How Common Is Insomnia on Semaglutide?
What Does Social Media Data Show About GLP-1 and Insomnia?
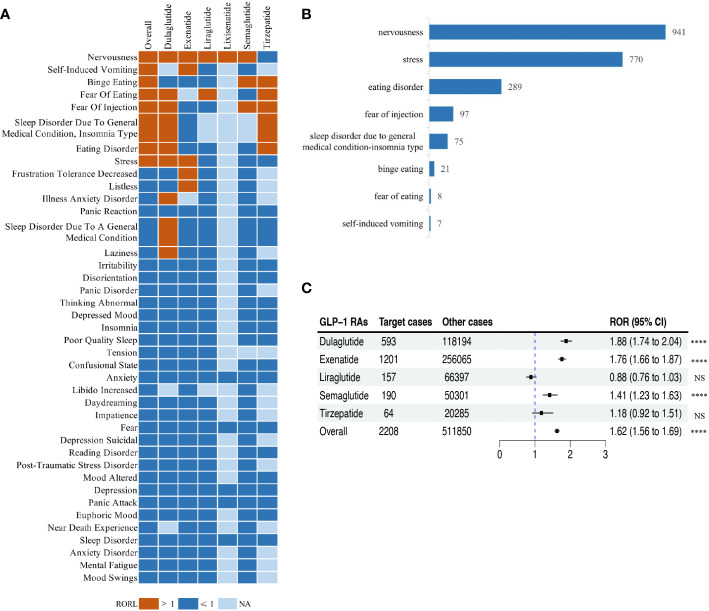
Arillotta et al. (2023) analyzed 12,136 Reddit comments, 14,515 YouTube videos, and 17,059 TikTok videos discussing GLP-1 receptor agonists. Among adverse mental health categories, insomnia topped the list at 620 matched posts — more than anxiety (353 matches) and depression (204 matches). Insomnia led even in a dataset dominated by discussions about weight loss and metabolic outcomes (Arillotta et al., 2023).
The breadth of the dataset — drawing from three platforms with different user demographics and content formats — gives this finding more weight than a single-platform observation would.
What Does FDA Adverse Event Data Show?
Chen et al. (2024) took a different approach, analyzing 181,238 GLP-1 receptor agonist adverse event reports from the FDA’s FAERS database spanning 2004 to 2023. Of these, 8,240 involved psychiatric complications. Sleep disturbances met disproportionality criteria — a statistical threshold indicating the reporting rate exceeds what would be expected by chance — alongside nervousness and stress. The median time to onset for psychiatric adverse events was 31 days after starting a GLP-1 drug (Chen et al., 2024).
The study population skewed female (65.89%) with a median age of 56 — reflecting the observed demographics of GLP-1 drug users. Fear of eating showed one of the strongest pharmacovigilance associations (reporting odds ratio 3.35), but sleep disturbances were among the eight distinct psychiatric categories that met the statistical threshold.
What Can and Cannot Be Concluded From This Data?
Social media data captures user experience at scale but cannot establish causation — people who have insomnia are more likely to post about it than people who sleep fine. Pharmacovigilance data quantifies reporting patterns but suffers from underreporting and confounding by comorbid conditions. Neither dataset proves GLP-1 drugs cause insomnia. Both datasets establish that sleep disruption is a prominent user-perceived effect.
The FDA label gap matters: insomnia does not appear as a listed adverse reaction in semaglutide prescribing information. This means it did not reach statistical significance in the original registration trials that defined the label. It does not mean it does not occur — it means the trials were not designed or powered to detect it.
Do GLP-1 Drugs Improve Sleep Apnea?
What Do Randomized Trial Numbers Show for Sleep Apnea?
Kow et al. (2025) pooled three randomized, placebo-controlled trials in non-diabetic participants and found GLP-1 receptor agonists produced a statistically significant reduction in the apnea-hypopnea index (AHI) — the standard measure of sleep apnea severity. The weighted mean difference was -16.6 events per hour (95% CI: -27.9 to -5.3). For context, moderate sleep apnea is defined as 15-30 events per hour, so a 16.6-event reduction can move a person from moderate to mild or from mild to normal range (Kow et al., 2025).
The same meta-analysis flagged a safety consideration: GLP-1 receptor agonists were associated with higher overall adverse event frequency compared to placebo (odds ratio 1.62), encompassing gastrointestinal and other tolerability issues. The sleep apnea benefit is documented, but so is the broader side-effect burden.
How Do GLP-1 Drugs Reduce Sleep Apnea Severity?
El-Solh et al. (2025) reviewed the mechanisms by which liraglutide, semaglutide, and tirzepatide improve sleep apnea. The primary pathway: body-fat reduction lowers fat deposition around the upper airway — the tissue that collapses during sleep and blocks breathing. Secondary mechanisms include reduced body-wide inflammation and possibly improved central respiratory drive through GLP-1 receptors in brainstem respiratory centers. Tirzepatide showed the largest absolute AHI reductions in the SURMOUNT-OSA trial (El-Solh et al., 2025).
Why Did Tirzepatide Receive FDA Approval for Sleep Apnea?
Mifsud et al. (2025) reviewed tirzepatide’s FDA approval for obstructive sleep apnea — the first time any drug has received FDA approval for this condition. The approval reframes sleep apnea from a condition managed by mechanical devices (CPAP) toward one that can also be addressed through metabolic means. The central unresolved question: are the benefits purely weight-mediated, or do direct GLP-1 receptor mechanisms in the brain and respiratory centers contribute independently? The answer matters because people with limited weight loss may or may not still benefit (Mifsud et al., 2025).
How Do Tirzepatide, Semaglutide, and Liraglutide Compare on Sleep Apnea?
Henney et al. (2025) compared tirzepatide, semaglutide, and liraglutide in an observational cohort of over 7,000 people with both sleep apnea and type 2 diabetes. Tirzepatide reduced incident sleep apnea cases compared to liraglutide. No statistically significant difference in sleep apnea incidence was detected between tirzepatide and semaglutide, though tirzepatide showed greater reduction in major cardiovascular events (Henney et al., 2025).
GLP-1 drugs interact with the same metabolic pathways — orexin, leptin, cortisol — that might be driving 3am wakeups for reasons unrelated to medication. These pathways may compound with hormonal, autonomic, or circadian factors. Whether or not you take semaglutide, identifying which causes might be active is a useful next step.
Find out which causes might be driving your 3am wakeups →
What Connects GLP-1 Drugs, Leptin, and Nighttime Hunger?
How Does the Leptin-Orexin Link Work During GLP-1 Weight Loss?
Orexin neurons do not fire in isolation — they integrate metabolic inputs from the body. One of the inputs with the largest effect is leptin. As Tsujino and Sakurai (2009) documented, leptin normally suppresses orexin neuron firing. When adipose tissue shrinks — as it does during GLP-1-mediated weight loss — circulating leptin drops. Reduced leptin removes the inhibition on orexin neurons, potentially amplifying the direct GLP-1 excitation that Acuna-Goycolea and van den Pol (2004) demonstrated in their electrophysiology experiments.
This creates a compounding effect: the drug itself excites orexin neurons through its receptor, and the body-fat reduction the drug produces lowers the hormone that would normally restrain those same neurons.
Does Sleep Apnea Itself Alter the Body’s Own GLP-1 Production?
Reutrakul et al. (2017) measured endogenous GLP-1 responses and sleep quality in 71 prediabetic adults using 7-day actigraphy and overnight home sleep monitoring. Increasing apnea-hypopnea index was independently and inversely associated with GLP-1 area-under-the-curve (B = -0.242, P = 0.045). After adjusting for sex, HbA1c, and BMI, higher AHI was associated with lower probability of being in the highest GLP-1 response quartile (odds ratio 0.581, P = 0.028). In other words, sleep apnea suppresses the body’s own GLP-1 production (Reutrakul et al., 2017).
This means people with untreated sleep apnea have lower baseline GLP-1 levels — a pre-existing deficit that exogenous GLP-1 drugs are overriding. The drug is not supplementing a normal level; it is replacing a suppressed one and adding more on top.
Why Do GLP-1 Drugs Improve Sleep in Some People and Worsen It in Others?
GLP-1 drugs both activate wake-promoting orexin neurons (a mechanism that could increase insomnia) and resolve the airway obstruction that fragments sleep (a mechanism that improves sleep continuity). Whether someone’s sleep improves or changes may depend on which pathway dominates at baseline. A person with moderate-to-severe sleep apnea and no pre-existing insomnia may sleep measurably better. A person without sleep apnea but with orexin-sensitive arousal patterns may have more fragmented sleep. Both outcomes are consistent with the pharmacology.
Frequently Asked Questions
Does Ozempic Cause Insomnia?
The evidence falls into three categories, none of which is individually conclusive. Social media analysis found 620 insomnia-matched posts across 43,710 GLP-1-related posts on Reddit, YouTube, and TikTok — the highest adverse mental health category (Arillotta et al., 2023). FDA pharmacovigilance data from 181,238 adverse event reports showed sleep disturbances meeting statistical disproportionality thresholds. And the mechanistic data shows GLP-1 excites orexin neurons, the brain’s wake-maintenance population. A prospective, controlled study designed to measure insomnia as a primary outcome in people taking semaglutide has not been conducted. The absence of insomnia on the FDA label means it was not statistically different in the registration trials — not that it does not occur.
Does Ozempic Improve Sleep Apnea?
The distinction between tirzepatide and semaglutide matters here. Tirzepatide has received FDA approval for obstructive sleep apnea; semaglutide has not, though evidence of benefit exists in the pooled meta-analysis data. The degree of improvement tracks with the magnitude of weight loss — higher weight loss correlates with larger AHI reductions (Mifsud et al., 2025).
Can Semaglutide Affect Sleep Quality?
This is a net-effect question without a single answer. Someone with moderate-to-severe obstructive sleep apnea may find their sleep quality improves as body-fat reduction opens the airway. Someone without sleep apnea but with arousal-sensitive sleep patterns may find sleep quality worsens as GLP-1-driven orexin activation increases nighttime wakefulness. Both outcomes are pharmacologically consistent. The same drug, through different mechanisms, can push sleep quality in opposite directions depending on the individual’s baseline (El-Solh et al., 2025).
Do GLP-1 Drugs Affect Hunger Hormones at Night?
The appetite-suppression effect of GLP-1 drugs is direct — the drug binds GLP-1 receptors in the hypothalamus and reduces hunger. The leptin effect is indirect but consequential. As fat mass decreases, leptin production falls. Leptin normally inhibits orexin neurons; with less leptin, orexin faces less opposition. This indirect pathway unfolds on a different timescale than the direct GLP-1 effect — it emerges over weeks to months as body composition changes, not on the first dose. The relationship between sleep apnea and endogenous GLP-1 production further complicates the picture: Reutrakul et al. (2017) showed that increasing AHI independently suppresses GLP-1 secretion, meaning many people starting these drugs already have a disrupted baseline.
Related Reading
- Metabolic Sleep Disruption
- Why Do You Wake Up Hungry at 3am?
- Does Leptin Resistance Cause Insomnia?
- Why Does Menopause Cause Insomnia?
- How Do Blood Sugar, Cortisol, and Leptin Control Whether You Sleep?
References
- Acuna-Goycolea, C., & van den Pol, A. (2004). Glucagon-like peptide 1 excites hypocretin/orexin neurons by direct and indirect mechanisms: implications for viscera-mediated arousal. The Journal of Neuroscience, 24(37), 8141–8152. https://pubmed.ncbi.nlm.nih.gov/15371515/
- Tsujino, N., & Sakurai, T. (2009). Orexin/hypocretin: a neuropeptide at the interface of sleep, energy homeostasis, and reward system. Pharmacological Reviews, 61(2), 162–176. https://pubmed.ncbi.nlm.nih.gov/19549926/
- Reutrakul, S., Sumritsopak, R., Saetung, S., Chanprasertyothin, S., & Anothaisintawee, T. (2017). The relationship between sleep and glucagon-like peptide 1 in patients with abnormal glucose tolerance. Journal of Sleep Research, 26(6), 756–763. https://pubmed.ncbi.nlm.nih.gov/28560837/
- Pizza, F., Barateau, L., Dauvilliers, Y., & Plazzi, G. (2022). The orexin story, sleep and sleep disturbances. Journal of Sleep Research, 31(4), e13665. https://pubmed.ncbi.nlm.nih.gov/35698789/
- Arillotta, D., Floresta, G., Guirguis, A., Corkery, J. M., Catalani, V., Martinotti, G., Sensi, S. L., & Schifano, F. (2023). GLP-1 Receptor Agonists and Related Mental Health Issues; Insights from a Range of Social Media Platforms Using a Mixed-Methods Approach. Brain Sciences, 13(11), 1503. https://pubmed.ncbi.nlm.nih.gov/38002464/
- Chen, W., Cai, P., Zou, W., & Fu, Z. (2024). Psychiatric adverse events associated with GLP-1 receptor agonists: a real-world pharmacovigilance study based on the FDA Adverse Event Reporting System database. Frontiers in Endocrinology, 15, 1330936. https://pubmed.ncbi.nlm.nih.gov/38390214/
- El-Solh, A. A., Gould, E., Aibangbee, K., Jimerson, T., & Hartling, R. (2025). Current perspectives on the use of GLP-1 receptor agonists in obesity-related obstructive sleep apnea: a narrative review. Expert Opinion on Pharmacotherapy, 26(1), 51–62. https://pubmed.ncbi.nlm.nih.gov/39621418/
- Kow, C. S., Sangarran Ramachandram, D. S., Hasan, S. S., & Thiruchelvam, K. (2025). Efficacy and safety of GLP-1 receptor agonists in the management of obstructive sleep apnea in individuals without diabetes: A systematic review and meta-analysis of randomized, placebo-controlled trials. Sleep Medicine, 129, 40–44. https://pubmed.ncbi.nlm.nih.gov/39978242/
- Henney, A. E., Riley, D. R., Anson, M., Heague, M., Hernandez, G., Alam, U., Craig, S., & Cuthbertson, D. J. (2025). Comparative Efficacy of Tirzepatide, Liraglutide, and Semaglutide in Reduction of Risk of Major Adverse Cardiovascular Events in Patients with Obstructive Sleep Apnea and Type 2 Diabetes: Real-World Evidence. Annals of the American Thoracic Society, 22(7), 1042–1052. https://pubmed.ncbi.nlm.nih.gov/40590655/
- Mifsud, C. S., Kolla, B. P., Rushlow, D. R., & Mansukhani, M. P. (2025). The impact of GLP-1 agonists on sleep disorders: spotlight on sleep apnea. Expert Opinion on Pharmacotherapy, 26(14–15), 1529–1538. https://pubmed.ncbi.nlm.nih.gov/41114602/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 10 references cited
