Overview: Metabolic impairment — including insulin resistance, blood sugar instability, mitochondrial decline, and disrupted fat metabolism — can directly fragment sleep and degrade its deepest and restorative stages. Five mechanisms contribute:
- Insulin resistance suppresses slow-wave sleep — the deepest sleep stage responsible for overnight metabolic restoration — creating a self-reinforcing cycle where less deep sleep worsens insulin resistance further
- Blood sugar instability triggers counterregulatory hormone surges (epinephrine, cortisol) during sleep that wake the brain, often between 2 and 4am
- Mitochondrial decline dysregulates sleep pressure through excessive reactive oxygen species, impairing the brain’s ability to maintain consolidated deep sleep
- NAD+ depletion weakens circadian clock amplitude, blurring the boundaries between deep sleep and wakefulness — particularly in midlife and beyond
- Disrupted fat metabolism prevents the normal overnight transition from glucose burning to fat burning, driving free fatty acid accumulation that feeds back into insulin resistance and sleep fragmentation
These mechanisms compound each other. Insulin resistance suppresses deep sleep, which worsens insulin resistance. Mitochondrial decline accelerates with age, as does NAD+ depletion. Many people have more than one cause contributing to their sleep disruption.
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 25 references cited
What Is Metabolism and What Is Metabolic Dysfunction?
Every cell in the body depends on metabolic processes running correctly. These processes regulate how glucose enters cells, how insulin manages blood sugar after meals, how fat is stored and burned for fuel, and how mitochondria produce ATP — the energy currency that powers every biological function.
When metabolic processes work well, blood sugar stays stable between meals, insulin moves glucose into cells efficiently, mitochondria produce energy with minimal waste, and the body transitions between fuel sources (glucose during activity, fatty acids during rest) on a predictable circadian schedule.
When they break down, the consequences accumulate across every organ.
Insulin resistance — where cells stop responding normally to insulin — is one of the earliest and frequently occurring forms of metabolic impairment. It affects an estimated 40% of US adults aged 18-44, and the prevalence increases with age. Insulin resistance is the precursor to prediabetes and type 2 diabetes, and it often develops years before blood sugar rises enough to meet a formal threshold.
The longevity consequences of metabolic impairment are well documented. It raises the risk of cardiovascular disease — the leading cause of death globally. It drives type 2 diabetes, which affects over 500 million people worldwide and can shorten life expectancy by 6 years or more depending on age at diagnosis. It accelerates cognitive decline and increases Alzheimer’s risk — a 2025 meta-analysis of 91 studies and over 2.2 million participants (Chen et al.) found that insomnia symptoms were associated with more than double the odds of adverse glycaemic control, with measurably higher glucose levels across all analyses — underscoring that sleep and metabolic health decline together. It promotes chronic inflammation, which damages tissues across the body over decades. For anyone focused on longevity and long-term brain health, metabolic function is not optional — it is the base on which everything else rests.
But metabolic impairment does not only undermine longevity over years and decades. It also directly disrupts sleep — night after night — through measurable mechanisms.
How Does Metabolic Impairment Disrupt Sleep?
What Is Insulin Resistance and What Are the Impacts on Sleep and Deep Sleep?
Insulin is a hormone released by the pancreas after eating. It tells cells in muscle, fat, and liver to absorb glucose from the blood. In insulin resistance, those cells respond weakly to the message — so the pancreas produces more insulin to compensate, blood sugar stays elevated longer, and the entire glucose regulation process runs under strain.
Insulin resistance exists on a spectrum. At one end, cells are mildly less responsive — fasting glucose may still appear normal, but postmeal glucose stays elevated longer than it should. At the other end, the pancreas can no longer keep up, and blood sugar rises into the prediabetic and eventually diabetic range.
The relationship between insulin resistance and deep sleep is bidirectional — and this is where the self-reinforcing cycle begins.
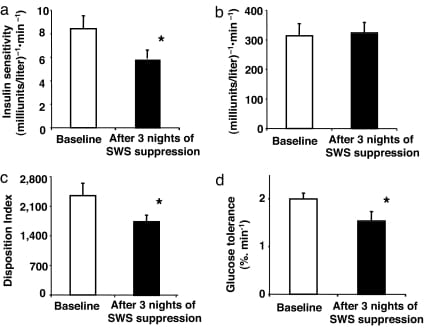
A 2008 study (Tasali et al.) selectively suppressed slow-wave sleep for 3 nights — without reducing total sleep time — and found that insulin sensitivity decreased, without any compensatory increase in insulin release. This is an important distinction: the participants slept the same number of hours, but their deep sleep was specifically disrupted. The result was measurably impaired glucose handling. It was deep sleep quality — not sleep duration — that regulated insulin sensitivity.
Earlier work showed the same direction. Spiegel et al. (1999) restricted sleep to 4 hours per night for 6 nights and measured a 40% reduction in glucose clearance and a 30% reduction in the acute insulin response. Broussard et al. (2012) found that 4 nights of short sleep reduced adipose tissue insulin responsiveness by 30% and whole-body insulin sensitivity by 16%.
A 2025 meta-analysis of 91 studies and 2,217,521 participants (Chen et al., Journal of Global Health) found that insomnia symptoms were associated with 2.28-fold higher odds of adverse glycaemic control. People with insomnia had measurably higher glucose levels across all analyses (p < 0.001). The association held across all 10 separate meta-analyses in the study — the direction of effect was consistent regardless of how sleep was measured.
The sex-specific dimension is emerging. A 2026 study (Oishi and Suzuki, Life Sciences) found that in a mouse model of chronic sleep disruption, only males exhibited a reduction in slow-wave sleep, and only males developed insulin resistance and impaired insulin secretion. Females showed oral glucose intolerance but did not develop the insulin resistance or slow-wave sleep loss seen in males. This pairing of slow-wave sleep loss with insulin resistance in males — but not females — is consistent with the observation that men may be more metabolically vulnerable to deep sleep disruption.
In a study of 6,966 participants with obstructive sleep apnea or primary snoring (Huang et al., 2021), those with slow-wave sleep in the lowest category of total sleep time had approximately 35% higher odds of insulin resistance compared to those with adequate slow-wave sleep — but this effect was present in the sleep apnea group, not in primary snorers. The finding suggests that the combination of reduced deep sleep and another sleep-disrupting condition amplifies the metabolic impact.
The cycle then reinforces itself. Insulin resistance degrades slow-wave sleep quality. Degraded slow-wave sleep worsens insulin resistance. When slow-wave sleep is suppressed, the brain spends more time in lighter sleep stages that are more vulnerable to arousal — creating conditions for repeated wakeups in the early morning hours, when the circadian drive for sleep is already weakening.
What Is Blood Sugar Instability and How Does It Contribute to Poor Sleep and 3am Wakeups?
In a healthy metabolism, blood sugar declines gradually overnight as the body transitions to burning fat for fuel. The decline is smooth, the brain stays asleep, and the fasting state is maintained until morning.
In someone with insulin resistance or prediabetes, this process can go wrong in two ways.
First, blood sugar may drop too fast or too low — a condition called nocturnal hypoglycemia. When blood glucose falls below approximately 70 mg/dL during sleep, the body mounts a counterregulatory response: epinephrine, glucagon, cortisol, and growth hormone surge to raise blood sugar. This is a survival mechanism — the brain requires a continuous glucose supply to function. But the epinephrine and cortisol that rescue blood sugar also activate the sympathetic nervous division and the brain, producing the experience of waking abruptly at 2-4am with a racing heart or the feeling of being suddenly, fully alert.
Second, in the early morning hours (approximately 3-5am), the liver releases stored glucose in preparation for waking — a process called the dawn phenomenon. In a person with adequate insulin sensitivity, the pancreas releases enough insulin to manage this glucose release smoothly. In an insulin-resistant person, the insulin response is too weak to match the liver’s glucose output, producing a glucose spike that can fragment sleep.
Banarer and Cryer (2003, Diabetes) found that the autonomic defense against nocturnal hypoglycemia is impaired during sleep. In their study, the epinephrine response during hypoglycemia was markedly blunted in type 1 diabetic subjects during sleep compared to wakefulness (85 ± 47 pg/ml asleep versus 240 ± 86 pg/ml awake). Sleep efficiency during hypoglycemia was 77% in diabetic subjects versus only 26% in controls — meaning the diabetic participants remained asleep through glucose drops that woke healthy sleepers. The result is that diabetic individuals are more likely to remain asleep through dangerous glucose drops that would wake a healthy sleeper.
This is more common than many people realize. Wang et al. (2022, Frontiers in Endocrinology) found that 24.3% of type 2 diabetic outpatients experienced asymptomatic nocturnal hypoglycemia — glucose drops during sleep that produced no conscious awareness but were detected by continuous glucose monitoring. People with higher glycemic variability (larger swings in glucose) were more likely to experience these episodes.
Standard definitions may undercount the problem. Martine-Edith et al. (2024, Diabetes Technology & Therapeutics) studied 574 participants and found that the conventional “nocturnal” window of midnight to 6am consistently underestimates true sleep-time hypoglycemia. Participants’ actual sleep periods extended well beyond that window, and sensor-detected hypoglycemia rates during actual sleep were higher than clock-based rates (1.7 versus 1.4 episodes per week in type 1 diabetes, p < 0.001).
What Is Mitochondrial Dysfunction and How Does It Fragment Sleep and Contribute to 3am Wakeups?
Mitochondria take in oxygen and nutrients and produce ATP — the energy currency that powers every cellular process. As a byproduct of energy production, mitochondria generate reactive oxygen species (ROS), which are chemically reactive molecules that can damage DNA, proteins, and the mitochondria themselves. In healthy cells, antioxidant defenses neutralize ROS. But when mitochondrial function declines — due to aging, metabolic impairment, or chronic oxidative stress — ROS production outpaces the cleanup, and damage accumulates.
Recent research has identified a direct mechanism connecting mitochondrial ROS to sleep drive.
A 2026 review in Free Radical Biology & Medicine (Tian, Liu, and Liu) established that ROS — particularly hydrogen peroxide (H2O2) — function as molecules that encode sleep drive, not as harmful byproducts alone. In both fruit flies and mice, H2O2 levels in sleep-promoting brain circuits track the duration of wakefulness and gate sleep initiation. The relationship follows an inverted-U curve: moderate ROS levels stabilize sleep, but excessive ROS — from mitochondrial impairment — fragment it. The authors proposed circuit-specific redox tuning as a therapeutic approach, noting that the goal is restoring moderate ROS levels rather than eliminating ROS.
Sleep itself is the primary window for mitochondrial quality control. Mauri et al. (2022, Frontiers in Cell and Developmental Biology) proposed that mitophagy — the selective recycling of damaged mitochondria — is regulated by the circadian clock and may occur predominantly during sleep. A significant percentage of mitochondrial mass is replaced daily through this process. When sleep is disrupted, mitochondria cannot be properly maintained, which generates more ROS, which fragments sleep further — another self-reinforcing cycle.
Wu et al. (2025, Tissue & Cell) showed a downstream consequence: sleep deprivation in mice caused measurable cardiac damage with elevated oxidative stress markers (MDA rose to 7.16 nmol/mg protein, GSH dropped to 33.88 μg/mL, SOD fell to 56.12 U/mL). Transmission electron microscopy revealed mitochondrial structural damage in heart tissue. The sleep deprivation activated a ferroptosis pathway — a form of iron-dependent cell death — through BACH1, linking mitochondrial ROS accumulation to tissue damage beyond the brain.
Why this worsens after 40: The glutathione redox buffer (GSH:GSSG ratio) — the body’s primary antioxidant defense inside cells — shifts toward a more pro-oxidant state with advancing age, and antioxidant enzyme expression declines in parallel (reviewed in Mir, Lark, and Nehs, 2025). The resulting mismatch between ROS production and antioxidant capacity means that the metabolic swing between wakefulness and sleep becomes more extreme even as the buffer capacity weakens. This makes mitochondrial sleep fragmentation progressively worse in midlife and beyond — directly relevant to longevity.
As ROS accumulate beyond the moderate range, the brain’s sleep-promoting circuits lose the stable input that keeps deep sleep consolidated. Deep sleep bouts become shorter, transitions to lighter stages become more frequent, and the probability of full arousal increases — particularly after 2am, when the circadian drive for sleep is waning.
What Is NAD+, How Does NAD+ Change with Age, and How Does NAD+ Decline Weaken the Sleep-Wake Cycle and Contribute to 3am Wakeups?
NAD+ is a coenzyme that plays two roles relevant to sleep.
1. First, it is an electron carrier in the mitochondrial electron transport chain — essential for converting food into energy.
2. Second, it activates SIRT1, an enzyme that regulates gene expression, DNA repair, inflammation, and — critically — the circadian clock.
Without adequate NAD+, both energy production and cellular maintenance decline. This makes NAD+ depletion a convergence point for metabolic and sleep impairment.
NAD+ levels can decline by half or more between ages 40 and 60 in human tissue studies.
Two forces drive this decline:
- increased consumption of NAD+ by DNA repair enzymes (CD38 and PARP respond to accumulating DNA damage by using NAD+ as a substrate) and
- decreased production through the NAD+ salvage pathway, where the enzyme NAMPT is the rate-limiting step.
The result is a progressively depleted supply of the molecule that powers both energy metabolism and clock regulation.
Two landmark 2009 studies in Science established how NAD+ and the circadian clock are coupled.
Ramsey et al. found that NAMPT and NAD+ levels oscillate across the 24-hour cycle, and that this oscillation is directly regulated by the core clock machinery. Nakahata et al. showed that the CLOCK-SIRT1 complex controls the NAD+ salvage pathway by regulating NAMPT transcription — creating a feedback loop where the clock regulates its own metabolic fuel supply. Together, these two papers established the core loop: CLOCK/BMAL1 (core clock proteins) drive NAMPT transcription → NAMPT produces NAD+ → NAD+ activates SIRT1 → SIRT1 feeds back into clock gene regulation, ensuring high-amplitude circadian gene expression.
When NAD+ levels decline, SIRT1 activity drops and clock amplitude weakens.
The consequence for sleep is this: weaker circadian clock amplitude means the boundary between deep sleep and light sleep becomes less distinct. Deep sleep bouts become shorter and more easily interrupted. The first half of the night — normally dominated by consolidated deep sleep — becomes more fragmented.
By 2-4am, when the circadian drive for sleep is already waning naturally, a weakened clock provides even less protection against arousal. This is the window where 3am wakeups occur.
Poljsak (2018, Rejuvenation Research) proposed that NAMPT-mediated NAD+ decline is not only a 24-hour circadian timer but also a longer-term biological aging clock. The amplitude and duration of NAD+-mediated clock oscillations may determine the rate of organismal aging — connecting the nightly experience of fragmented sleep to the longer-term trajectory of longevity.
What Is Disrupted Fat Metabolism and How Does It Contribute to Poor Sleep and 3am Wakeups?
The body burns different fuels at different times of day.
During waking hours, glucose is the primary fuel. During sleep, metabolism moves toward fatty acid oxidation — the respiratory quotient drops toward 0.7, indicating fat is the dominant fuel source.
This overnight transition is governed by two mechanisms: the circadian clock, which coordinates the timing of metabolic enzymes, and the glucose-fatty acid cycle (also called the Randle cycle), which prevents the body from burning glucose and fat simultaneously. When metabolic impairment disrupts this switch — through insulin resistance, circadian misalignment, or excess circulating fatty acids — the overnight metabolic program breaks down.
The downstream effect is direct. When the transition to fat oxidation does not occur, non-esterified fatty acids (NEFAs) accumulate from adipose tissue. These excess fatty acids inhibit glucose uptake in muscle through Randle cycle competition, worsening insulin resistance. They also promote ceramide accumulation in muscle and liver tissue — Sergi et al. (2025, Nutrition Research Reviews) identified ceramide as the molecular link between saturated fatty acid exposure and insulin resistance, showing that ceramides directly impair insulin receptor pathways in skeletal muscle and liver.
Approximately 13% of plasma lipids show 24-hour rhythmic oscillations (Patel et al., 2016, reviewed in Potter et al., 2016), and sleep disruption flattens these oscillations. Liu (2025, Health and Metabolism) showed that key enzymes in fatty acid metabolism — COX, LOX, and cytochrome P450 enzymes — exhibit circadian expression patterns. Circadian disruption can impair the timed production of lipid-derived molecules that these enzymes produce.
The metabolic timing dimension is important. Leproult et al. (2014) demonstrated that circadian misalignment reduced insulin sensitivity by 58% — nearly double the 32% reduction from sleep restriction alone. This means that disrupted metabolic timing is a more potent driver of metabolic impairment than reduced sleep hours. Ferrell and Chiang (2015, Cellular and Molecular Gastroenterology and Hepatology) showed this in an animal model: short-term circadian disruption impaired hepatic bile acid and lipid homeostasis even without changes in sleep duration.
When the overnight fat-burning switch breaks down, the body cannot complete its normal overnight metabolic program. This metabolic instability contributes to the same glucose variability described in the blood sugar section above, and the inflammatory lipid byproducts from improperly metabolized fatty acids contribute to low-grade inflammation that fragments sleep. The result compounds: disrupted fat metabolism worsens insulin resistance, insulin resistance suppresses deep sleep, and lighter sleep further disrupts metabolic timing — feeding the cycle forward into the 2-4am waking window.
What Triggers or Worsens Metabolic Sleep Disruption?
Does Metabolic Sleep Disruption Get Worse with Age?
The age-related intensification of metabolic sleep disruption is not a single process — it is the convergence of several independent declines happening in the same decades.
Insulin sensitivity decreases with age, even in adults who maintain a healthy weight. NAD+ levels can decline by half or more between ages 40 and 60, weakening both mitochondrial energy production and circadian clock amplitude. The glutathione redox buffer — the primary antioxidant defense inside cells — shifts toward a more pro-oxidant state with age, reducing the body’s capacity to manage the oxidative stress that accumulates during wakefulness. Mitochondrial efficiency decreases, producing more ROS per unit of energy generated.
Each of these changes independently fragments sleep. Together, they explain why metabolic sleep disruption tends to worsen in midlife — and why addressing metabolic health is directly relevant to longevity and long-term brain protection.
How Does Late-Night Eating Affect Overnight Metabolism?
The body expects to process food during waking hours and move to fasting metabolism during sleep. When eating extends into the late evening or night, insulin secretion continues into the sleep period. This delays the transition from glucose burning to fat burning, prevents the normal decline in blood sugar that supports deep sleep, and can produce the glucose variability that triggers counterregulatory hormone surges.
Gut microbiome circadian rhythms — bacteria perform different metabolic functions during active versus rest phases — are also disrupted by misaligned eating. The gut processes nutrients during the active phase and moves to detoxification during the rest phase. Late-night eating scrambles this timing.
Leproult et al. (2014) showed that circadian misalignment produced nearly double the metabolic damage of sleep restriction alone — suggesting that WHEN metabolic processes occur matters as much as whether they occur.
Can Metabolic Impairment Itself Disrupt Sleep?

When multiple components are present — a cluster sometimes called metabolic syndrome — the sleep disruption compounds. Visceral fat promotes chronic inflammation. Elevated triglycerides reflect disrupted lipid metabolism. Elevated fasting glucose indicates insulin resistance. Each of these feeds back into insulin resistance, deep sleep suppression, and blood sugar instability.
A 2025 prospective cohort study from the MIDUS (Midlife in the US) study, with approximately 12 years of follow-up, found that both short sleep (under 6 hours) and long sleep (over 9 hours) predicted incident metabolic syndrome, obesity, and type 2 diabetes — a U-shaped curve showing that sleep duration and metabolic health are linked in both directions.
For anyone focused on longevity, metabolic impairment is not a background condition that affects sleep as a side effect. It is a direct, measurable disruptor of the sleep architecture that the brain depends on for repair, waste clearance, and cognitive maintenance.
How Do You Know If Metabolic Impairment Is Disrupting Your Sleep?
Some experiences associated with metabolic sleep disruption:
- Waking between 2 and 4am — this is the window when overnight blood sugar reaches its lowest point, the counterregulatory hormone response is likeliest to trigger, and the circadian drive for sleep is already weakening
- Feeling alert or “wired” upon waking in the middle of the night — the epinephrine and cortisol surge that rescues low blood sugar also activates the brain, producing the paradox of being exhausted but wide awake
- Daytime fatigue despite sleeping 7-8 hours — when insulin resistance suppresses slow-wave sleep, total sleep hours may be adequate but deep sleep is insufficient. The brain sleeps long enough but not deeply enough.
- Carbohydrate cravings, especially late at night — disrupted appetite hormone regulation from metabolic impairment can manifest as late-night hunger, particularly for high-carbohydrate foods
- Sleep quality that worsens alongside weight gain or dietary changes — if sleep deterioration tracks with metabolic changes, the two may be linked through the mechanisms described above
Biomarkers that can indicate metabolic contribution to sleep disruption include:
- Fasting glucose and HbA1c — the prediabetic range (fasting glucose 100-125 mg/dL, HbA1c 5.7-6.4%) is where metabolic sleep disruption often begins
- Fasting insulin and HOMA-IR — direct measures of insulin resistance
- Triglyceride-to-HDL ratio — a marker of lipid metabolism health
- hsCRP — a marker of metabolic inflammation
- Continuous glucose monitoring data showing overnight glucose variability — the direct measurement of the mechanisms described above
Many people have 2-3 causes contributing to their sleep disruption, not just one. Metabolic disruption frequently overlaps with autonomic and inflammatory causes — and the mechanisms interact. Insulin resistance can worsen inflammation, inflammation can worsen insulin resistance, and both can amplify the autonomic stress response.
Find out which causes are driving your 3am wakeups →
Frequently Asked Questions
Is Metabolic Sleep Disruption Reversible?
Samy et al. (2024, QJM) demonstrated this in an animal model: after 4 weeks of induced insomnia produced elevated fasting blood glucose, insulin resistance, dyslipidemia, and inflammation in aged female rats, 2 weeks of sleep recovery normalized fasting blood glucose, total cholesterol, LDL-C, and body adiposity. Insulin resistance and triglycerides were reduced, though they remained elevated compared to controls — suggesting that full metabolic recovery may require longer recovery periods.
A 2025 study (Cheng et al., Frontiers in Nutrition) tested a 6-week dietary approach in 158 individuals with metabolic impairment and poor sleep (PSQI scores above 7). Sleep quality improved, and the improvement was mediated through three measurable pathways: reduced insulin resistance, reduced inflammatory markers (TNF-a and hsCRP, p < 0.001), and reduced oxidative stress. Insulin resistance was an independent predictor of sleep quality even after controlling for other variables.
Can You Have Metabolic Sleep Disruption Without Being Diabetic?
Insulin resistance develops gradually. Fasting glucose may appear normal while postmeal glucose responses are already impaired. The slow-wave sleep suppression described by Tasali et al. (2008) occurred in response to experimental sleep manipulation in healthy adults — not in diabetics. The metabolic-sleep cycle does not wait for a formal threshold.
An estimated 62% of people with prediabetic glucose levels report poor sleep — compared to 46% of those with normal glucose. The sleep disruption can begin on the insulin resistance spectrum while fasting glucose still appears normal.
Does Poor Sleep Cause Metabolic Problems, or Do Metabolic Problems Cause Poor Sleep?
The 2025 meta-analysis by Chen et al. — analyzing 91 studies and over 2.2 million participants — found that the association between insomnia symptoms and adverse glycaemic control was consistent across all analysis methods, supporting a bidirectional relationship. Tasali et al. (2008) demonstrated the sleep-to-metabolism direction experimentally (suppressing deep sleep impaired glucose handling). The insulin-resistance-to-sleep direction is supported by the observation that metabolically impaired individuals have measurably less deep sleep than metabolically healthy individuals at the same age.
Can Blood Sugar Affect Sleep Even If Fasting Glucose Is Normal?
The dawn phenomenon (the liver’s early-morning glucose release) and nocturnal glucose dips occur in people with insulin resistance who have not yet developed elevated fasting glucose. Continuous glucose monitoring has revealed that overnight glucose variability is more common than fasting glucose levels alone would suggest — Wang et al. (2022) found that nearly 1 in 4 type 2 diabetic outpatients had asymptomatic nocturnal hypoglycemia detected only by CGM.
A 2025 review in Diabetic Medicine (Pham et al.) found that 71% of studies measuring sleep quality in people with diabetes identified a measurable association with glycemic variability — the more unstable the overnight glucose, the worse the sleep.
Why Does Metabolic Sleep Disruption Get Worse in Midlife?
The midlife convergence of these declines explains why sleep quality often deteriorates between ages 40 and 60 even in people who maintain otherwise healthy habits. NAD+ depletion weakens circadian amplitude. Mitochondrial decline increases ROS beyond the threshold that stabilizes sleep. Glutathione depletion reduces the buffer against oxidative damage. Insulin resistance impairs deep sleep.
Each decade of metabolic decline adds another dimension of sleep fragmentation. This is one of the reasons that sleep quality and metabolic health are both central to any longevity-focused health strategy — they are not separate concerns, but two expressions of the same underlying metabolic trajectory.
Related Reading
- Can a Blood Sugar Drop Wake You Up at 3am? — The highest-intent gateway for blood sugar instability and 3am waking.
- Which Nicotinamide Adenine Dinucleotide Precursor Improves Sleep — Nicotinamide Mononucleotide or Nicotinamide Riboside? — How NAD+ biology overlaps with sleep-wake regulation.
- How Do Blood Sugar, Cortisol, and Leptin Control Whether You Sleep? — How leptin fits into overnight metabolic control.
- Why Are You Exhausted But Can’t Sleep? Is It a Mitochondrial Energy Paradox? — How mitochondrial energy problems can produce wired exhaustion.
- Why Does Ozempic Wake You Up at 3am? — How GLP-1 medications can interact with overnight blood sugar and sleep.
- Autonomic Sleep Disruption — How cortisol and sympathetic activation overlap with metabolic night waking.
- Inflammatory Sleep Disruption — How inflammatory signaling can worsen insulin resistance and sleep quality.
- Circadian Sleep Disruption — How clock timing regulates glucose, insulin, temperature, and cortisol.
- Hormonal Sleep Disruption in Men — How testosterone, cortisol, visceral fat, and sleep interact in men.
- Hormonal Sleep Disruption in Women — How ovarian hormone changes can interact with metabolic sleep disruption.
References
Insulin Resistance and Deep Sleep
1. Spiegel, K., Leproult, R., & Van Cauter, E. (1999). Impact of sleep debt on metabolic and endocrine function. The Lancet, 354(9188), 1435-1439. https://pubmed.ncbi.nlm.nih.gov/10543671/
2. Tasali, E., Leproult, R., Ehrmann, D. A., & Van Cauter, E. (2008). Slow-wave sleep and the risk of type 2 diabetes in humans. Proceedings of the National Academy of Sciences, 105(3), 1044-1049. https://pubmed.ncbi.nlm.nih.gov/18172212/
3. Broussard, J. L., Ehrmann, D. A., Van Cauter, E., Tasali, E., & Brady, M. J. (2012). Impaired insulin signaling in human adipocytes after experimental sleep restriction: A randomized, crossover study. Annals of Internal Medicine, 157(8), 549-557. https://pubmed.ncbi.nlm.nih.gov/23070488/
4. Chen, Y., Tong, E., Rao, Y., Yu, E. Y. W., Zeegers, M., & Wesselius, A. (2025). The association between insomnia (related symptoms) and glycaemic control: A systematic review and meta-analysis. Journal of Global Health, 15, 04016. https://pmc.ncbi.nlm.nih.gov/articles/PMC11803432/
5. Oishi, K., & Suzuki, N. (2026). Sex-specific differences in psychophysiological stress-induced circadian sleep disruption and its impact on glucose metabolism in mice. Life Sciences, 393, 124319. https://pubmed.ncbi.nlm.nih.gov/41819221/
6. Huang, W., Liu, Y., Wang, X., Li, X., Liu, Y., Zou, J., Xu, H., Zhu, H., Yi, H., Guan, J., & Yin, S. (2021). Effect of interaction between slow wave sleep and obstructive sleep apnea on insulin resistance: A large-scale study. Nature and Science of Sleep, 13, 739-749. https://pubmed.ncbi.nlm.nih.gov/34113201/
Blood Sugar Instability and Night Waking
7. Banarer, S., & Cryer, P. E. (2003). Sleep-related hypoglycemia-associated autonomic failure in type 1 diabetes: Reduced awakening from sleep during hypoglycemia. Diabetes, 52(5), 1195-1203. https://pubmed.ncbi.nlm.nih.gov/12716752/
8. Martine-Edith, G., Divilly, P., Zaremba, N., Soholm, U., Broadley, M., Baumann, P. M., … & Choudhary, P. (2024). A comparison of the rates of clock-based nocturnal hypoglycemia and hypoglycemia while asleep among people living with diabetes: Findings from the Hypo-METRICS study. Diabetes Technology & Therapeutics, 26(7), 433-441. https://pubmed.ncbi.nlm.nih.gov/38386436/
9. Wang, S., Tan, Z., Wu, T., Shen, Q., Huang, P., Wang, L., Liu, W., Song, H., Lin, M., Shi, X., & Li, X. (2022). Largest amplitude of glycemic excursion calculating from self-monitoring blood glucose predicted the episodes of nocturnal asymptomatic hypoglycemia detecting by continuous glucose monitoring in outpatients with type 2 diabetes. Frontiers in Endocrinology, 13, 858912. https://pubmed.ncbi.nlm.nih.gov/35498416/
Mitochondrial Function and Sleep
10. Tian, Y., Liu, X., & Liu, D. (2026). Physiological roles of reactive oxygen species in sleep homeostasis. Free Radical Biology & Medicine, 242, 220-236. https://pubmed.ncbi.nlm.nih.gov/41135839/
11. Mauri, S., Favaro, M., Bernardo, G., Mazzotta, G. M., & Ziviani, E. (2022). Mitochondrial autophagy in the sleeping brain. Frontiers in Cell and Developmental Biology, 10, 956394. https://pubmed.ncbi.nlm.nih.gov/36092697/
12. Wu, Z., Huang, X., Lu, X., & Cao, Y. (2025). The interplay of sleep deprivation, ferroptosis, and BACH1 in cardiovascular disease pathogenesis. Tissue & Cell, 95, 102848. https://pubmed.ncbi.nlm.nih.gov/40081254/
NAD+ and Circadian Clock
13. Ramsey, K. M., Yoshino, J., Brace, C. S., Abrassart, D., Kobayashi, Y., Marcheva, B., Hong, H.-K., Chong, J. L., Buhr, E. D., Lee, C., Takahashi, J. S., Imai, S.-I., & Bass, J. (2009). Circadian clock feedback cycle through NAMPT-mediated NAD+ biosynthesis. Science, 324(5927), 651-654. https://pubmed.ncbi.nlm.nih.gov/19299583/
14. Nakahata, Y., Sahar, S., Astarita, G., Kaluzova, M., & Sassone-Corsi, P. (2009). Circadian control of the NAD+ salvage pathway by CLOCK-SIRT1. Science, 324(5927), 654-657. https://pubmed.ncbi.nlm.nih.gov/19286518/
15. Poljsak, B. (2018). NAMPT-mediated NAD biosynthesis as the internal timing mechanism: In NAD+ world, time is running in its own way. Rejuvenation Research, 21(3), 210-224. https://pubmed.ncbi.nlm.nih.gov/28756747/
Fat Metabolism and Sleep
16. Leproult, R., Holmback, U., & Van Cauter, E. (2014). Circadian misalignment augments markers of insulin resistance and inflammation, independently of sleep loss. Diabetes, 63(6), 1860-1869. https://pubmed.ncbi.nlm.nih.gov/24458353/
17. Ferrell, J. M., & Chiang, J. Y. (2015). Short-term circadian disruption impairs bile acid and lipid homeostasis in mice. Cellular and Molecular Gastroenterology and Hepatology, 1(6), 664-677. https://pubmed.ncbi.nlm.nih.gov/26645046/
18. Liu, J. (2025). Physiological and pathological insights into the circadian rhythm of polyunsaturated fatty acids metabolism. Health and Metabolism, 2(3), 100017. https://doi.org/10.53941/hm.2025.100017
19. Sergi, D., Zauli, E., Celeghini, C., Previati, M., & Zauli, G. (2025). Ceramides as the molecular link between impaired lipid metabolism, saturated fatty acid intake and insulin resistance: Are all saturated fatty acids to be blamed for ceramide-mediated lipotoxicity? Nutrition Research Reviews, 38(1), 256-266. https://pubmed.ncbi.nlm.nih.gov/39263838/
Reversibility
20. Samy, A., et al. (2024). Retrieval of normal sleep for two weeks normalized adiposity and minimized insulin resistance induced by 4-week insomnia in aged female rats. QJM, 117(Suppl 1), hcae175.865. https://doi.org/10.1093/qjmed/hcae175.865
Additional References
21. Cheng, J., Liu, Q., Qiu, Z., Pan, M., Song, J., Zhao, Y., Dong, N., Li, S., & Han, S. (2025). Dietary management improves sleep quality in patients with metabolic syndrome: The mediating roles of metabolic, inflammatory, and oxidative stress changes. Frontiers in Nutrition, 12, 1712215. https://www.frontiersin.org/journals/nutrition/articles/10.3389/fnut.2025.1712215/full
22. Pham, C. T., Ali, A., Churilov, L., Baqar, S., Hendrieckx, C., O’Neal, D. N., Howard, M. E., & Ekinci, E. I. (2025). The association between glycaemic variability and sleep quality and quantity in adults with type 1 and type 2 diabetes: A systematic review. Diabetic Medicine, 42(4), e15485. https://pubmed.ncbi.nlm.nih.gov/39663626/
23. Associations of sleep duration and quality with risk of incident obesity, diabetes, and metabolic syndrome: Results from the Midlife in the United States study (2025). Chronobiology International, 42(11), 1528-1535. https://www.tandfonline.com/doi/full/10.1080/07420528.2025.2556840
24. Huang, T. (2025). Sleep irregularity, circadian disruption, and cardiometabolic disease risk. Circulation Research, 137(5), 709-726. https://pubmed.ncbi.nlm.nih.gov/40811504/
25. Mir, F. A., Lark, A. R. S., & Nehs, C. J. (2025). Unraveling the interplay between sleep, redox metabolism, and aging: Implications for brain health and longevity. Frontiers in Aging, 6, 1605070. https://pubmed.ncbi.nlm.nih.gov/40469623/
