Most approaches to mast cell insomnia target downstream symptoms — antihistamines block histamine receptors, mast cell stabilizers reduce degranulation pharmacologically, and melatonin supplements attempt to restore circadian signaling. But the vagus nerve sits upstream of all of these targets, physically controlling whether mast cells degranulate in the first place. When vagal tone drops — as occurs in dysautonomia, postural orthostatic tachycardia syndrome, or chronic stress — mast cell activation proceeds without adequate neural restraint. This article covers the vagus nerve’s role as the upstream regulator of mast cell activity: the anatomical connection between vagal fibers and mast cells, the functional suppression evidence from animal models, and the clinical trial data for vagus nerve stimulation as a treatment for insomnia. Mast cell regulation by the vagus nerve is one component of the broader autonomic sleep picture covered in the parent pillar on autonomic sleep disruption.
How Does the Vagus Nerve Physically Control Mast Cells?
The connection between the vagus nerve and mast cells is not merely biochemical signaling across a distance. It is anatomical. Stead and colleagues demonstrated in 2006 that vagal afferent nerve fibers physically penetrate the small intestinal mucosa and form direct contacts with intestinal mucosal mast cells. These nerve endings are in direct apposition to mast cell membranes, establishing a structural basis for bidirectional neuroimmune communication that operates at the level of cellular contact rather than systemic circulation.
The functional significance of this physical connection becomes clear in vagotomy experiments. When researchers surgically severed the vagus nerve in animal models, mast cell density in the intestinal mucosa decreased. This finding establishes that vagal innervation exerts a trophic maintenance effect on local mast cell populations — the nerve does not merely signal to mast cells but actively sustains their presence and population density in the tissue. The vagus nerve is a structural requirement for normal mast cell residence in the gut mucosa, not a passive bystander.
Electrical stimulation experiments confirmed the functional direction of this relationship. When the cervical vagus nerve was electrically stimulated in animal models, researchers measured increases in histamine and serotonin content within intestinal mucosal mast cells. Vagal signaling actively modulates mast cell mediator stores — the nerve can alter how much histamine and serotonin mast cells contain. In the opposite direction, antigen challenge in parasite-infected rats produced dramatic increases in mesenteric afferent nerve firing, demonstrating that mast cell activation generates measurable neural signals traveling back to the brain through vagal pathways. Parasite infection also induced hyperinnervation of the mucosa concurrent with elevated mast cell numbers, indicating bidirectional structural plasticity — when mast cells proliferate during inflammation, the nerve network grows to match them, and vice versa. This anatomy is the foundation for understanding how vagal tone loss leads to unregulated mast cell behavior.
What Happens to Mast Cells When Vagal Tone Drops?
The vagal anatomy from the preceding section implies a clear prediction: if vagal tone drops, mast cell regulation should fail. Nishino and colleagues described this phenomenon in a 2022 review, drawing on experimental data from a chronic mild stress model in mice. Animals subjected to sustained stress developed reduced non-rapid eye movement sleep, fragmented sleep architecture, and impaired glucose tolerance — a phenotype that mirrors the clinical presentation of human chronic insomnia with metabolic comorbidity. When the researchers examined tissue samples from stressed animals, they found elevated mast cell counts in both the brain and adipose tissue compared to unstressed controls. Chronic stress — which reduces vagal tone and shifts autonomic balance toward sympathetic dominance — produced measurable mast cell proliferation in multiple organ systems simultaneously.
The causal role of mast cells in this stress-insomnia phenotype was confirmed through genetic and pharmacological experiments. Mast cell-deficient mice (W/Wv genotype) subjected to the identical chronic stress protocol did not develop glucose intolerance, establishing that the metabolic dysfunction depended specifically on mast cell presence rather than stress alone. Treatment with cromolyn sodium (a mast cell stabilizer) or a histamine H1 receptor antagonist reversed both the insomnia and the metabolic dysfunction in stressed animals that had normal mast cell populations. These interventions worked by blocking the downstream consequences of mast cell degranulation — but they did not address the upstream cause: the loss of vagal regulatory control over mast cell activation.
This model illuminates the wired-but-tired presentation common in mast cell activation syndrome patients with comorbid dysautonomia. The Ehlers-Danlos syndrome, postural orthostatic tachycardia syndrome, and mast cell activation syndrome triad is well-documented clinically. Postural orthostatic tachycardia syndrome specifically involves sympathetic dominance and reduced vagal tone. Low vagal tone removes the parasympathetic input that suppresses mast cell degranulation. Unchecked degranulation produces multi-mediator sleep disruption through histamine (which promotes wakefulness), prostaglandins (which drive neuroinflammation), and cytokines (which disrupt hypothalamic sleep regulation). The released mediators further impair autonomic function, creating a reinforcing cycle: low vagal tone leads to unchecked mast cell activation, which leads to mediator release, which leads to further autonomic disruption and lower vagal tone.
Can Stimulating the Vagus Nerve Reduce Mast Cell Degranulation?
Wang and colleagues published in 2024 direct evidence that vagus nerve stimulation suppresses mast cell degranulation in living animals. Using a rat ischemic stroke model — which produces systemic neuroinflammation and mast cell activation — the researchers demonstrated that vagus nerve stimulation-treated animals showed significantly reduced mast cell activation compared to untreated controls. The key quantifiable readout was chymase secretion. Chymase is a serine protease released exclusively by mast cells during degranulation, making it a specific biomarker for mast cell activation rather than general inflammation. Reduced chymase in vagus nerve stimulation-treated animals confirmed that the intervention suppressed mast cell degranulation specifically, not merely downstream inflammatory signaling.
The mechanistic pathway for this suppression operated through the microbiota-gut-brain axis rather than through direct local nerve effects alone. Gut microbiota composition, which was severely disrupted by the initial neuroinflammatory injury, normalized in vagus nerve stimulation-treated animals. Gastrointestinal motility and barrier function were both restored. This implicates a systems-level mechanism: vagal stimulation restores gut microbiota composition, which in turn modulates mast cell behavior systemically through microbial metabolites and immune signaling. The suppression is not limited to mast cells physically contacted by vagal fibers — it extends throughout the body via the gut-brain axis.
This finding connects directly to the anatomy established by Stead in 2006. The same vagal nerve fibers that physically contact mast cells in the intestinal mucosa can be externally stimulated to suppress mast cell degranulation. The mechanism proceeds through a defined sequence: stimulate the vagus nerve, activate gut-brain axis signaling, normalize microbiota composition, suppress mast cell degranulation, reduce mediator release. For mast cell-driven insomnia specifically, this sequence means that restoring vagal input may address the problem at its regulatory origin rather than blocking individual mediators after release.
Does Transcutaneous Auricular Vagus Nerve Stimulation Improve Sleep?
Zhang and colleagues published in JAMA Network Open in 2024 the largest sham-controlled randomized trial of transcutaneous auricular vagus nerve stimulation for chronic insomnia. The trial randomized 72 participants to active stimulation or sham over 8 weeks, with 68 completing the intervention. Conducted between October 2021 and December 2022 in Beijing, the study used the auricular branch of the vagus nerve — accessible through a clip placed on the ear — as the stimulation target. This auricular branch projects to brainstem nuclei involved in autonomic and arousal regulation.
The active transcutaneous auricular vagus nerve stimulation group achieved a mean Pittsburgh Sleep Quality Index reduction of 8.2 points (95% confidence interval: -9.3 to -7.0), compared to 3.9 points (95% confidence interval: -5.1 to -2.7) in the sham group. The between-group difference of 4.2 points reached statistical significance (P < .001) with a Cohen d effect size of 1.2 -- classified as a large effect. The minimal clinically important difference threshold for the Pittsburgh Sleep Quality Index is 2.5 points; the observed 4.2-point between-group difference exceeded this threshold by 68%, confirming clinical meaningfulness beyond statistical significance alone. Secondary outcomes including mental health scores, anxiety measures, and fatigue all improved in parallel with sleep quality. Benefits were fully sustained through a 20-week follow-up assessment, indicating durable autonomic remodeling rather than a transient stimulation effect that disappears when treatment stops.
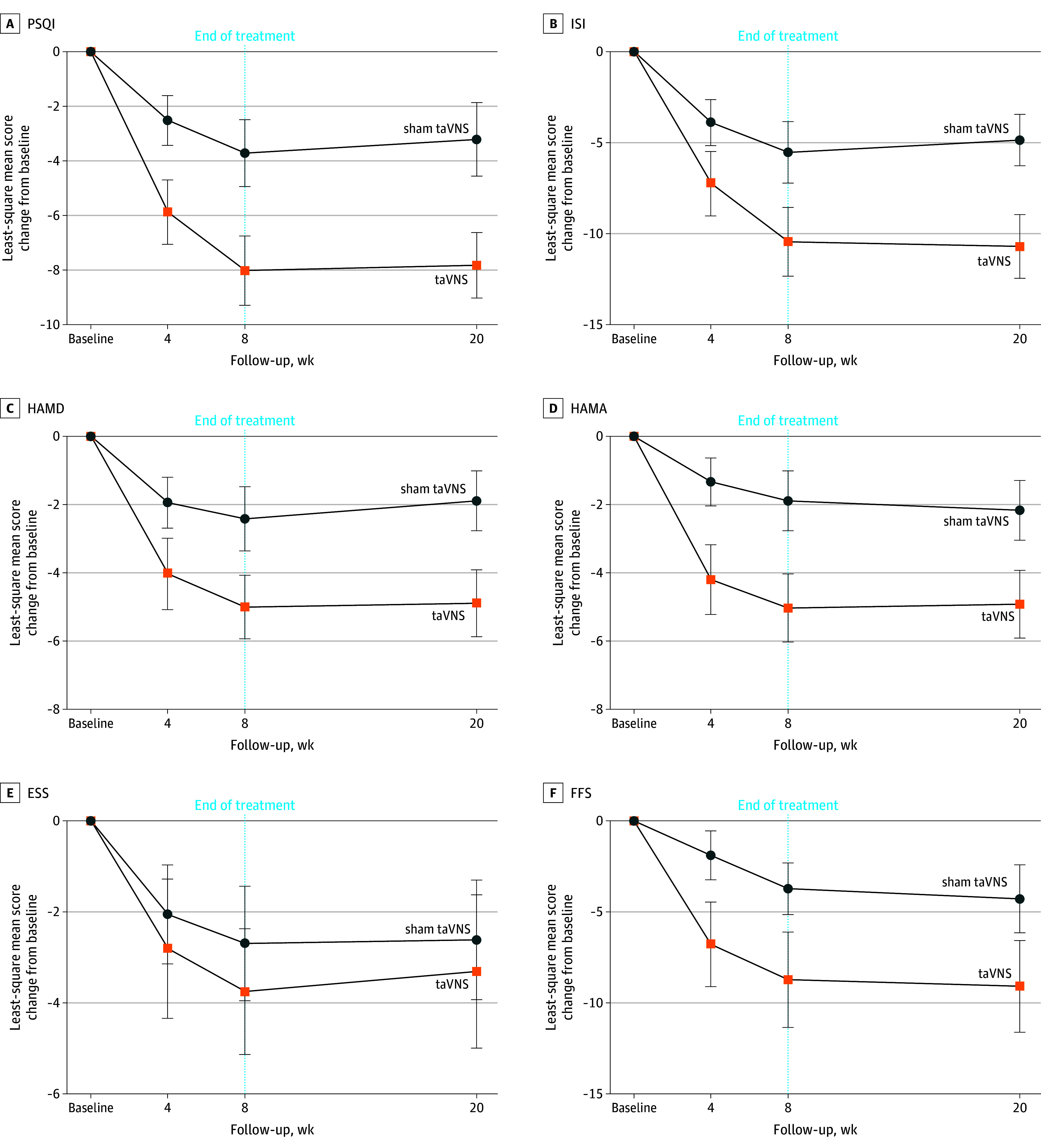
Yeom and colleagues independently replicated these findings in 2025. Their double-blind, sham-controlled trial enrolled 40 participants with chronic insomnia disorder in a 6-week transcutaneous auricular vagus nerve stimulation protocol. Pittsburgh Sleep Quality Index scores decreased by approximately 4.5 points in the active group compared to 1.9 points in the sham group. The between-group difference of approximately 2.6 points exceeded the 2.5-point minimal clinically important difference threshold. Insomnia Severity Index scores also improved substantially. Total sleep duration increased in the active group. The intervention was well-tolerated with no significant adverse events.
Two independent randomized controlled trials both showing significant sleep improvements over sham constitute reliable evidence for transcutaneous auricular vagus nerve stimulation as an effective insomnia intervention. One important transparency note: both trials enrolled general chronic insomnia populations, not mast cell activation syndrome cohorts specifically. No trial has yet tested transcutaneous auricular vagus nerve stimulation in mast cell activation syndrome patients. However, the mechanistic chain from the preceding sections — vagal fibers physically contact mast cells (Stead 2006), vagus nerve stimulation suppresses mast cell degranulation through the gut-brain axis (Wang 2024) — provides a rationale for why this approach may be particularly relevant when mast cell activation contributes to sleep disruption. Restoring vagal tone through transcutaneous auricular vagus nerve stimulation addresses the upstream regulatory failure that allows mast cells to degranulate unchecked.
Mast cell-driven sleep disruption controlled by vagal tone may overlap with other autonomic causes of insomnia — sympathetic overdrive, heart rate variability-indexed inflammatory load, or vagal damage from chronic inflammation. Identifying which autonomic mechanisms might be active helps guide the right approach.
Find out which causes might be driving your 3am wakeups –>
Frequently Asked Questions
Is Transcutaneous Auricular Vagus Nerve Stimulation the Same as an Implanted Vagus Nerve Stimulator?
How Long Does Transcutaneous Auricular Vagus Nerve Stimulation Take to Improve Sleep?
Does Vagus Nerve Stimulation Work for Mast Cell Activation Syndrome Specifically?
Can Mast Cell Stabilizers and Transcutaneous Auricular Vagus Nerve Stimulation Work Together?
Why Might Mast Cell Activation Syndrome Patients Have Low Vagal Tone?
Related Reading
- How Does Autonomic Nervous System Dysfunction Disrupt Sleep?
- Why Does Inflammation Cause Insomnia?
- How Does Your Vagus Nerve Actively Suppress Inflammation?
- Can Inflammation Damage Your Vagus Nerve?
- What Does Heart Rate Variability Reveal About Inflammation and Vagal Tone During Sleep?
References
1. Stead, R. H., Colley, E. C., Wang, B., Partosoedarso, E., Lin, J., Stanisz, A., & Hillsley, K. (2006). Vagal influences over mast cells. Autonomic Neuroscience: Basic & Clinical, 125(1-2), 53-61. https://pubmed.ncbi.nlm.nih.gov/16500155/
2. Nishino, S., Sakai, N., Nishino, N., & Ono, T. (2022). Brain Mast Cells in Sleep and Behavioral Regulation. Current Topics in Behavioral Neurosciences, 59, 427-446. https://pubmed.ncbi.nlm.nih.gov/35711025/
3. Wang, Y., Tan, Q., Pan, M., Yu, J., Wu, S., Tu, W., Li, M., & Jiang, S. (2024). Minimally invasive vagus nerve stimulation modulates mast cell degranulation via the microbiota-gut-brain axis to ameliorate blood-brain barrier and intestinal barrier damage following ischemic stroke. International Immunopharmacology, 132, 112030. https://pubmed.ncbi.nlm.nih.gov/38603861/
4. Zhang, S., Zhao, Y., Qin, Z., Han, Y., He, J., Zhao, B., Wang, L., Duan, Y., Huo, J., Wang, T., Wang, Y., & Rong, P. (2024). Transcutaneous Auricular Vagus Nerve Stimulation for Chronic Insomnia Disorder: A Randomized Clinical Trial. JAMA Network Open, 7(12), e2451217. https://pubmed.ncbi.nlm.nih.gov/39680406/
5. Yeom, J. W., Kim, H., Park, S., Yoon, Y., Seo, J. Y., Cho, C. H., & Lee, H. J. (2025). Transcutaneous auricular vagus nerve stimulation (taVNS) improves sleep quality in chronic insomnia disorder: A double-blind, randomized, sham-controlled trial. Sleep Medicine, 133, 106579. https://pubmed.ncbi.nlm.nih.gov/40398066/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 5 references cited
