CoQ10 and PQQ are two of the commonly supplemented mitochondrial compounds, but advice on whether they help sleep ranges from "life-changing" to "gave me insomnia." The contradiction is not arbitrary — dose, timing, and the form of CoQ10 used determine whether these compounds improve sleep or work against it. Getting the regimen wrong means spending months on a supplement schedule that disrupts the sleep it was meant to restore.
The controlled trial evidence below covers CoQ10 and PQQ sleep outcomes — doses tested, effect sizes measured, who benefits, and the timing window that separates improvement from insomnia. For how mitochondria regulate sleep architecture, see the parent pillar on Metabolic Sleep Disruption. For exercise-based mitochondrial support, see the sibling article on exercise, mitochondrial biogenesis, and sleep.
Mitochondrial energy production is one pathway among several — including blood sugar regulation, cortisol rhythm, and hormonal status — that influence sleep quality. For the full overview, see Metabolic Sleep Disruption.
Does CoQ10 Improve Sleep Quality — Or Can It Cause Insomnia?
Recent and direct evidence comes from Abbasi et al. (2025), a double-blind, placebo-controlled trial of 50 individuals with age-related hearing reduction. The CoQ10 group received 100 mg/day for six weeks. Sleep disturbance was among the assessed outcomes, and the between-group difference was statistically significant: -7.60 in the CoQ10 group versus -1.0 in placebo (p<0.001). This was a low, accessible dose producing rapid, measurable sleep improvement.
A larger trial is Castro-Marrero et al. (2021) — a 12-week RCT enrolling 207 individuals with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). Participants received 200 mg CoQ10 plus 20 mg NADH daily. Sleep duration showed a between-group improvement at week 4 (p=0.018), and habitual sleep efficiency improved within the CoQ10 group at week 8 (p=0.038), measured by the Pittsburgh Sleep Quality Index. Overall between-group PSQI differences did not reach statistical significance. The study also reported improvements in fatigue perception and health-related quality of life over the 12-week period.
Why the insomnia reports? CoQ10’s mechanism explains both outcomes. CoQ10 is a component of the mitochondrial electron transport chain — it shuttles electrons from Complex I and Complex II to Complex III, increasing ATP synthesis. During the day, enhanced ATP production reduces fatigue. At night, the same ATP increase opposes the natural reduction in cellular energy output that facilitates sleep onset.
Dose matters. Tsai et al. (2022) conducted a meta-analysis of 13 RCTs (n=1,126) and found a dose-response coefficient of -0.0017 per mg for fatigue reduction — higher doses reduce fatigue more, which also implies greater arousal potential at night. The overall pooled effect size was Hedges’ g=-0.398 (p=0.001), with CoQ10-only formulations showing a stronger effect of g=-0.552 (p=0.001), and longer supplementation duration amplified effects (coefficient -0.0042 per day). CoQ10-induced insomnia may be more likely at doses above 200 mg or with evening administration.
Safety across all 13 meta-analyzed trials was favorable: only one gastrointestinal adverse event among 602 CoQ10 participants.
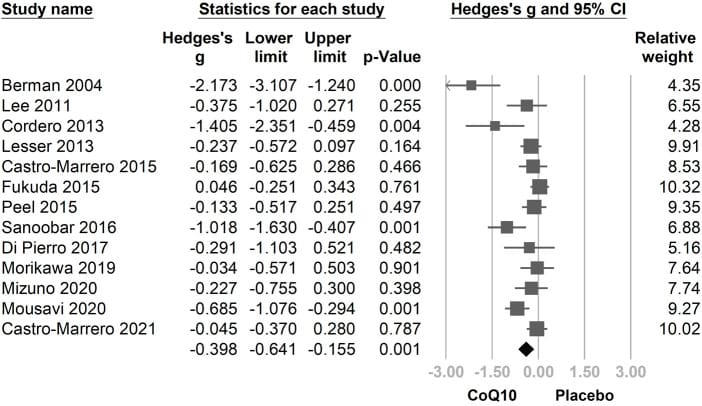
What Does the Controlled Trial Evidence Show for PQQ and Sleep?
The broadest PQQ human trial is Shiojima et al. (2022) — a 12-week, double-blind, placebo-controlled study of 64 healthy elderly volunteers (58 completers, mean age approximately 71 years). At 21.5 mg/day, PQQ produced improvement across all seven cognitive domains measured: composite memory, verbal memory, reaction time, complex attention, cognitive flexibility, executive function, and motor speed. In preclinical research, PQQ has been shown to upregulate PGC-1alpha, driving mitochondrial biogenesis and reducing ROS — mechanisms that overlap with pathways involved in sleep-wake cycling.
Baltic et al. (2024) conducted a 6-week double-blind RCT of 34 elderly individuals with mild cognitive impairment, testing PQQ combined with dihydrogen. Serum BDNF (brain-derived neurotrophic factor) rose in the PQQ group (p=0.01) while the placebo group showed no change. Cerebral oxygenation increased from 48.4% to 52.8% (p=0.005). Brain N-acetyl aspartate — a marker of neuronal mitochondrial integrity — improved at 7 of 13 measured brain locations. In separate research, BDNF elevation is associated with improved slow-wave sleep depth and hippocampal memory consolidation during sleep, and cerebral oxygenation reflects enhanced mitochondrial ATP production in brain tissue. But sleep was not measured in this trial.
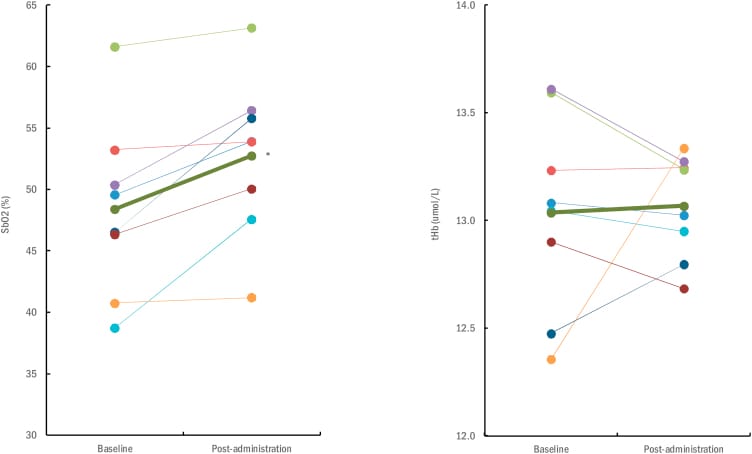
Co-administration is mechanistically logical: PQQ drives mitochondrial biogenesis via PGC-1alpha (creating new mitochondria), while CoQ10 optimizes electron transport chain function (making existing mitochondria produce ATP more efficiently).
The evidence gap needs to be stated plainly: no placebo-controlled human RCT has measured PQQ and sleep as a primary or secondary outcome. The sleep claims circulating online extrapolate from cognitive and BDNF data. The hypothesis is reasonable, but it is not proven.
When Should You Take Mitochondrial Supplements — Morning or Night?
Neither Abbasi et al. (2025) nor Castro-Marrero et al. (2021) specified the time of day for CoQ10 administration in their published study designs. Based on CoQ10’s ATP-enhancing mechanism, morning dosing is a reasonable strategy to avoid potential evening arousal effects.
Rossello Aubach et al. (2025) tested CoQ10 combined with tryptophan and magnesium in an 89-completer crossover trial of fibromyalgia participants. Sleep quality improved during the supplement period and was not replicated during the placebo period — making sleep one of the clearest supplement-attributable findings. The combination formulation may buffer CoQ10’s energizing properties because tryptophan is a precursor for serotonin and melatonin synthesis, and magnesium supports GABA receptor function.
Timing sensitivity follows from CoQ10’s mechanism. CoQ10 enhances Complex I and Complex II electron transport, increasing ATP synthesis. In the morning, this increased ATP production contributes to daytime energy and, in principle, supports adenosine accumulation — adenosine is the molecule that builds sleep pressure throughout the day. In the evening, the same ATP increase opposes the natural reduction in cellular energy production that facilitates sleep onset.
The time-to-effect data from the meta-analysis (Tsai et al., 2022) shows that longer supplementation duration amplified CoQ10’s effects (coefficient -0.0042 per day). Castro-Marrero et al. (2021) showed between-group improvement in sleep duration at week 4, with broader fatigue and quality-of-life improvements across 12 weeks. Abbasi et al. (2025) found significance at 6 weeks. A minimum 6-8 week commitment is needed before assessing sleep response.
Ubiquinol versus ubiquinone: Ubiquinol is the reduced, active form of CoQ10 with higher bioavailability — the body absorbs more per milligram. Standard CoQ10 (ubiquinone) must be converted to ubiquinol before cells can use it. No head-to-head trial has compared the two forms for sleep outcomes. The form difference: ubiquinol may reach the therapeutic window at lower doses, potentially staying below the arousal threshold. Sanoobar et al. (2016) achieved large effect sizes for fatigue (eta-squared=0.56) at 500 mg/day CoQ10, while Abbasi et al. (2025) showed significance at 100 mg/day — suggesting the effective dose range is wide, and higher-bioavailability forms may allow the lower end of that range to be sufficient.
Can You Stack CoQ10, PQQ, and Magnesium Safely?
Rossello Aubach et al. (2025) provide the strongest stacking evidence: a double-blind crossover trial of 89 fibromyalgia completers receiving CoQ10 + tryptophan + magnesium for 3 months, separated by a 1-month washout. Sleep quality improved during the supplement period and was not replicated during placebo. The combination addresses multiple sleep-relevant pathways — CoQ10 for mitochondrial energy production, tryptophan for serotonin and melatonin synthesis, magnesium for GABA receptor function and muscle relaxation.
Castro-Marrero et al. (2022) tested 400 mg CoQ10 + 200 micrograms selenium for 8 weeks in 27 ME/CFS participants in an open-label pilot study. Total antioxidant capacity increased, lipid peroxidation decreased (p<0.0001), and inflammatory cytokines fell (p<0.01). Sleep disturbance did not reach significance at 8 weeks (p=0.480). The oxidative stress improvements are relevant to sleep because elevated ROS and pro-inflammatory cytokines (IL-1beta and TNF-alpha) drive sleep fragmentation and reduce slow-wave sleep — but sleep improvement may require a longer supplementation window than 8 weeks at this dose.
PQQ and CoQ10 address different parts of the mitochondrial network: PQQ drives mitochondrial biogenesis via PGC-1alpha (creating new mitochondria), while CoQ10 optimizes electron transport chain function in existing mitochondria.
Dose ranges from evidence: CoQ10 100-200 mg/day for sleep (Abbasi et al., 2025 at 100 mg; Castro-Marrero et al., 2021 at 200 mg). PQQ 20 mg/day (Shiojima et al., 2022 at 21.5 mg). These doses are well below thresholds associated with adverse events across all reviewed trials. Two recent meta-analyses of CoQ10 and depression (Magalhaes et al., 2026; Akwan et al., 2025) found antidepressant effects at 100-200 mg/day — a dose range overlapping with the sleep-improvement window. Because depression and insomnia are bidirectionally linked, the mood benefits of CoQ10 at these doses may contribute to sleep improvement through a separate pathway.
Mitochondrial supplements address one piece of a larger metabolic picture. In adults over 40, blood sugar instability, cortisol rhythm disruption, hormonal changes, and inflammation can compound alongside weakened mitochondrial energy production — and each pathway responds to different approaches. Identifying which causes might be active is a useful step before committing to a supplement approach.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
How Much CoQ10 Should I Take for Sleep?
The dose-response coefficient from Tsai et al. (2022) is -0.0017 per mg, meaning each additional milligram of CoQ10 produces slightly more fatigue reduction. Sanoobar et al. (2016) used 500 mg/day and achieved large fatigue effect sizes (eta-squared=0.56) but did not measure sleep directly — at that dose, morning administration becomes especially relevant to avoid evening arousal. Start at 100 mg/day with morning dosing and assess sleep response over 6-8 weeks before adjusting.
What Is the Difference Between CoQ10 and Ubiquinol for Sleep?
No head-to-head trial has compared ubiquinone versus ubiquinol for sleep outcomes. The trials reviewed here used both forms at different doses. The form consideration: if CoQ10 at a given dose causes evening alertness, switching to ubiquinol at a lower milligram dose may deliver equivalent mitochondrial support with less stimulation. The meta-analysis (Tsai et al., 2022) did not differentiate between forms in its pooled analysis, so the dose-response data applies across both.
How Long Does It Take for CoQ10 to Work for Sleep?
CoQ10 is not a fast-acting sleep aid. It works by restoring mitochondrial electron transport chain function, which gradually improves cellular energy production in ways that support healthy sleep architecture. The meta-analysis found longer supplementation duration amplified effects (coefficient -0.0042 per day, p=0.007). Benefits continue to build past the initial improvement window. Minimum commitment: 6-8 weeks of daily dosing before assessing whether the supplementation is producing a sleep effect.
Can Mitochondrial Supplements Replace Exercise for Sleep?
CoQ10 makes existing mitochondria produce ATP more efficiently. Exercise generates new mitochondria through PGC-1alpha activation — the same pathway PQQ acts on, but exercise is a stronger and more sustained activator. Both matter, and they are complementary rather than interchangeable. The Castro-Marrero et al. (2022) oxidative stress data showed that CoQ10+selenium reduced inflammatory cytokines and lipid peroxidation, but the cellular-level repair from exercise-driven biogenesis addresses mitochondrial quantity in a way that oral supplementation does not. For the full evidence on exercise and mitochondrial sleep, see the sibling article on exercise, mitochondrial biogenesis, and sleep.
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep — the pillar article for metabolic sleep disruption, covering glucose regulation, cortisol rhythm, mitochondrial stress, NAD+ reduction, and fat metabolism
- Why Are You Exhausted But Can’t Sleep? Is It a Mitochondrial Energy Paradox? — mitochondrial fatigue, unrefreshing sleep, ATP production, and why rest may not restore cellular energy
- Do Your Mitochondria Control When You Sleep? and Why Does It Break Down After 40? — ROS sleep pressure, deep sleep reduction after 40, mitochondrial DNA aging, and sleep timing
- Is Your 3am Wakeup a Metabolic Emergency? Blood Sugar, Cortisol, and Mitochondrial reduction? — the 2-4am vulnerability window, glucose drops, cortisol rescue, and mitochondrial energy buffering
- Why Are You Wired but Tired, and What Role Do Mitochondria Play? — hyperarousal, cortisol rhythm changes, stress physiology, and mitochondrial power-down reduction
- How Does Exercise Rebuild Your Mitochondria and Fix Your Sleep? — exercise type, timing, mitochondrial biogenesis, PGC-1?, and sleep architecture
- What Is the Measured Cellular Impact of Sleep Fragmentation? — fragmented sleep, oxidative stress, vascular injury, inflammation, and cellular aging impact
References
Abbasi, R., Emami, F., Atighechi, S., & Sadeghi, Z. (2025). The effect of coenzyme Q10 on tinnitus severity and sleep quality in individuals with presbycusis. Iranian Journal of Otorhinolaryngology, 37(1), 33-39. https://pubmed.ncbi.nlm.nih.gov/39850225/
Akwan, R., Elsharkawy, M. M., Zrineh, A., Amleh, A., Douden, B., Alhouseini, M., Alsaeed, L., & Eldesouki, M. (2025). The effect of coenzyme Q10 supplementation on depressive indicators and anxiety: A review and meta-analysis of randomized controlled trials. European Journal of Clinical Pharmacology, 81(11), 1555-1568. https://pubmed.ncbi.nlm.nih.gov/40833470/
Baltic, S., Nedeljkovic, D., Todorovic, N., Ranisavljev, M., Korovljev, D., Cvejic, J., Ostojic, J., LeBaron, T. W., Timmcke, J., Stajer, V., & Ostojic, S. M. (2024). The impact of six-week dihydrogen-pyrroloquinoline quinone supplementation on mitochondrial biomarkers, brain metabolism, and cognition in elderly individuals with mild cognitive impairment: A randomized controlled trial. The Journal of Nutrition, Health & Aging, 28(8), 100287. https://pubmed.ncbi.nlm.nih.gov/38908296/
Castro-Marrero, J., Segundo, M. J., Lacasa, M., Martinez-Martinez, A., Sanmartín Sentañes, R., & Alegre-Martin, J. (2021). Effect of dietary coenzyme Q10 plus NADH supplementation on fatigue perception and health-related quality of life in individuals with myalgic encephalomyelitis/chronic fatigue syndrome: A prospective, randomized, double-blind, placebo-controlled trial. Nutrients, 13(8), 2658. https://pubmed.ncbi.nlm.nih.gov/34444817/
Castro-Marrero, J., Domingo, J. C., Cordobilla, B., Ferrer, R., Giralt, M., Sanmartín-Sentañes, R., & Alegre-Martín, J. (2022). Does coenzyme Q10 plus selenium supplementation ameliorate clinical outcomes by modulating oxidative stress and inflammation in individuals with myalgic encephalomyelitis/chronic fatigue syndrome? Antioxidants & Redox Signaling, 36(10-12), 729-739. https://pubmed.ncbi.nlm.nih.gov/35229657/
Magalhaes, P. L. M., da Silva, A. M. P., Maximiano, M. L. B., Fernandes, J. V. A., Amaral, D. C., Fortes, A. A. T., Filho, H. N. F., Silva, L. O., Teixeira, B. D. D. S., Franco, E. S., & Maia, M. B. S. (2026). Effects of coenzyme Q10 supplementation on depressive indicators and fatigue: A review and meta-analysis of randomized controlled trials. Journal of Clinical Psychopharmacology, 46(1), 93-100. https://pubmed.ncbi.nlm.nih.gov/41294251/
Rosselló Aubach, L., Fornós Roca, X., & Fernández Álvarez, M. E. (2025). Effects of coenzyme Q10, tryptophan, and magnesium supplementation on fatigue in individuals with fibromyalgia – A randomized trial. Journal of Dietary Supplements, 22(3), 433-444. https://pubmed.ncbi.nlm.nih.gov/40151031/
Sanoobar, M., Dehghan, P., Khalili, M., Azimi, A., & Seifar, F. (2016). Coenzyme Q10 as a approach for fatigue and depression in multiple sclerosis individuals: A double-blind randomized clinical trial. Nutritional Neuroscience, 19(3), 138-143. https://pubmed.ncbi.nlm.nih.gov/25603363/
Shiojima, Y., Takahashi, M., Takahashi, R., Moriyama, H., Bagchi, D., Bagchi, M., & Akanuma, M. (2022). Effect of dietary pyrroloquinoline quinone disodium salt on cognitive function in healthy volunteers: A randomized, double-blind, placebo-controlled, parallel-group study. Journal of the American Nutrition Association, 41(8), 796-809. https://pubmed.ncbi.nlm.nih.gov/34415830/
Tsai, I. C., Hsu, C. W., Chang, C. H., Tseng, P. T., & Chang, K. V. (2022). Effectiveness of coenzyme Q10 supplementation for reducing fatigue: A review and meta-analysis of randomized controlled trials. Frontiers in Pharmacology, 13, 883251. https://pubmed.ncbi.nlm.nih.gov/36091835/
Written by Kat Fu, M.S., M.S.? Last reviewed: May 2026? 10 references cited
