People notice a measurable change in sleep quality somewhere around their 40s — less deep sleep, more waking, morning fatigue that was absent a decade earlier. This reduction maps to measurable changes in mitochondrial function that are now well-documented in human and animal studies.
This article covers the cellular mechanism by which mitochondria generate sleep pressure, how that mechanism degrades with age, what happens to mitochondrial DNA over decades, and the cognitive consequences of lower deep sleep. It does not cover the fatigue-insomnia paradox (see the energy paradox article), cortisol and stress pathways (see the wired-but-tired article), 3am timing (see the 3am metabolic emergency article), supplements (see the CoQ10 and PQQ article), or exercise-based approaches (see the exercise and mitochondria article).
For the broader metabolic context — including blood sugar instability, cortisol patterns, and inflammatory pathways — see the parent overview: Metabolic Sleep Disruption.
Do Your Mitochondria Produce the Molecular Pressure That Makes You Sleepy?
Until recently, sleep pressure was understood mainly through adenosine accumulation — a byproduct of ATP consumption that builds up during wakefulness and promotes drowsiness. The 2025 Nature study by Sarnataro et al. identified a more upstream mechanism: mitochondria themselves undergo structural changes that generate sleep pressure at the cellular level.
The study used single-cell RNA sequencing on Drosophila sleep-control neurons (237 rested, 86 sleep-deprived). In sleep-deprived neurons, genes encoding mitochondrial respiration and ATP synthesis proteins were selectively upregulated (Bonferroni-corrected p<0.05). The structural consequences were measurable: mitochondrial fragmentation increased (p<0.0001), mitophagy — the selective degradation of stressd mitochondria — was enhanced (p=0.0101), and mitochondria-endoplasmic reticulum contact sites expanded (p<0.0001). All of these changes reversed after recovery sleep.
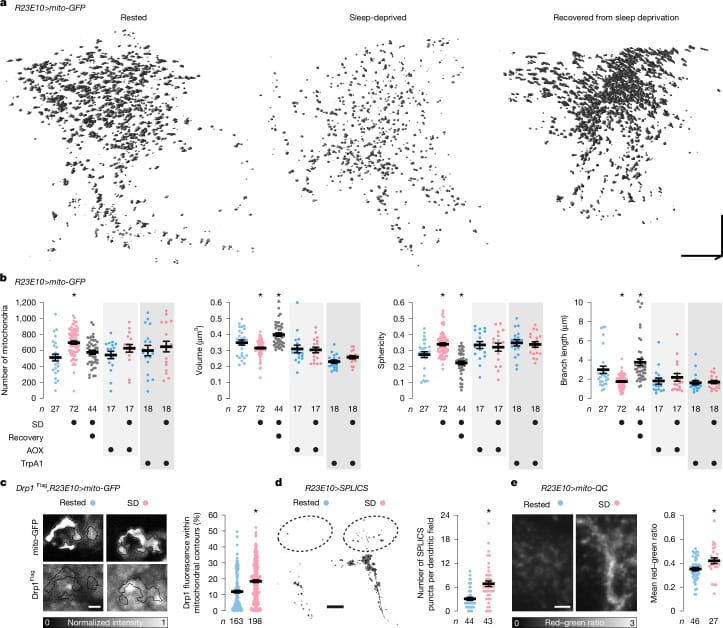
The causal chain runs in one direction: extended wakefulness produces ATP accumulation (p=0.0003), which causes mitochondrial fragmentation, which decreases neuronal excitability, which initiates sleep. The bidirectional proof was the study’s central finding: artificially inducing mitochondrial fusion increased both neuronal excitability and sleep duration, while promoting fission decreased both (p<0.0332). Uncoupling electron flux from ATP synthesis chemically relieved sleep pressure. This study was conducted in Drosophila. The authors discuss potential parallels in mammals, though the mechanism has been directly demonstrated only in Drosophila sleep-control neurons.
Sarnataro’s 2025 review in The Journal of Physiology proposed that mitochondrial fission and fusion dynamics may represent a conserved, cell-autonomous mechanism for regulating neuronal excitability, though he noted that Drosophila sleep-control neurons are currently the only identified neurons where this coupling has been directly demonstrated (Sarnataro, 2025). Separately, a 2024 Stanford review by Terzi et al. established that the ROS-sleep loop — where ROS drive sleep pressure and sleep clears ROS — is evolutionarily conserved from invertebrates to mammals. Cavefish populations that independently evolved reduced sleep show lower ROS levels and enhanced antioxidant activities, which may contribute to their reduced sleep need (Terzi et al., 2024).
A 2023 framework paper by Richardson and Mailloux described this as a dual-phase cycle: wakefulness is "nucleorestorative" — peak oxidative stress, nuclear DNA repair, protein synthesis — while sleep is "mitorestorative" — mitochondrial fusion, redox restoration, immune facilitation (Richardson & Mailloux, 2023). As Sarnataro et al. stated in the Nature study, sleep may be "an inescapable consequence of aerobic metabolism."
How Much Deep Sleep Do You Lose Per Decade After 40?
The Richardson and Mailloux (2023) framework explains why deep sleep amplitude and mitochondrial function reduction together. Their tripartite model — redox balance, bioenergetics, and temperature regulation — describes how all three converge during slow-wave sleep to restore mitochondrial architecture. Reversible cysteine modifications (S-glutathionylation, S-nitrosylation) act as molecular switches controlling mitochondrial respiration across the sleep-wake cycle. As circadian redox cycling weakens with age, slow-wave sleep quality may degrade because the molecular switches that coordinate mitochondrial restoration lose their timing precision.
The circadian coupling between mitochondria and the rest-activity cycle depends on clock genes. Mezhnina et al. (2022) established that the transcriptional activators CLOCK and BMAL1 drive circadian expression of mitochondrial genes, while mitochondrial sirtuins (SIRT3, SIRT4, SIRT5) regulate the antioxidant response with time-of-day precision. NRF2 — nuclear factor erythroid 2-related factor 2 — links CLOCK/BMAL1 activity to glutathione synthesis and superoxide dismutase induction. BMAL1 expression is known to reduction with age. When BMAL1 reductions, mitochondrial physiology may decouple from the circadian cycle, potentially generating chronic mitochondrial oxidative stress.
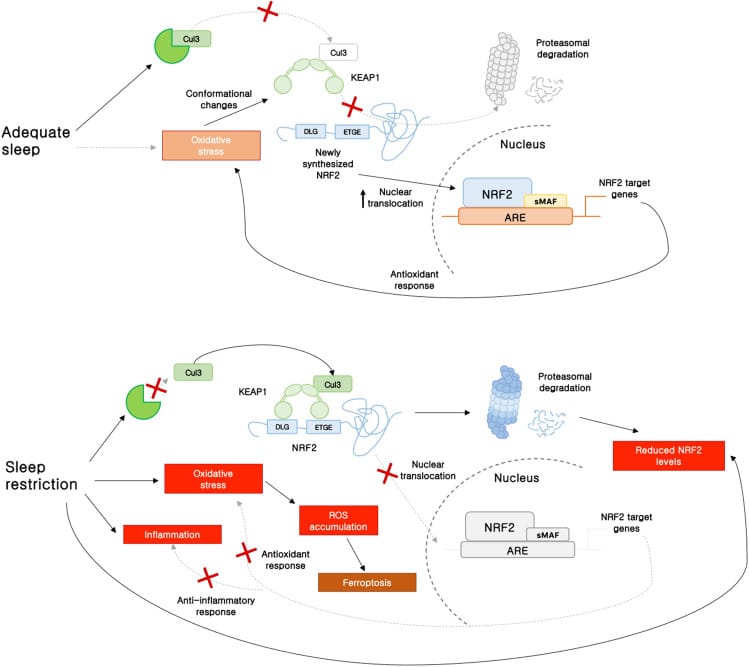
Davinelli et al. (2024) identified NRF2 as the central transcriptional hub coupling sleep homeostasis to the cellular antioxidant response. NRF2 activity is known to reduction with age. When NRF2 activity drops, the antioxidant genes it regulates — including those governing glutathione production and mitochondrial ROS buffering — are less responsive to the oxidative stress that builds during wakefulness. The result is a compounding vulnerability: less NRF2 activity produces weaker antioxidant recovery during sleep, which may produce more oxidative stress, which may further impair the mitochondrial machinery that generates deep sleep.
What Happens to Your Mitochondrial DNA as You Age?
The twin study by Wrede et al. (2015) provides among the strongest available human evidence that the relationship between sleep and mitochondrial DNA is not confounded by genetics. The study enrolled 15 monozygotic twin pairs (mean age 42.1 years) discordant for habitual sleep duration, with a mean within-pair difference of 94.3 minutes measured by actigraphy. The twin design controls for genetics, shared environment, and early-life exposures. Within twin pairs, shorter sleep duration was associated with reduced mitochondrial DNA copy number: each additional minute of sleep predicted a beta of 0.06 increase in mitochondrial DNA copy number (95% CI 0.004-0.12, p<0.05). Sleep efficiency showed an even stronger association — each 1% decrease in sleep efficiency predicted a 0.51 decrease in mitochondrial DNA copy number (95% CI 0.06-0.95, p<0.05).
A 2024 cross-sectional study by Han et al. found consistent results in a larger sample. Among 238 healthy middle-aged Korean adults, global Pittsburgh Sleep Quality Index (PSQI) score was inversely correlated with mitochondrial DNA copy number (r = -0.15, p = 0.025). The component that drove the association was sleep latency — how long it takes to fall asleep — not sleep duration. Longer sleep latency independently predicted reduced mitochondrial DNA copy number (beta = -0.16, p = 0.011), suggesting that autonomic arousal during sleep onset may be more relevant to mitochondrial DNA integrity than shorter total sleep time (Han et al., 2024). As a cross-sectional study, this finding shows an association, not a causal direction.
Circadian timing of sleep matters independently of sleep quantity. Bhatti et al. (2016) studied 440 workers and found that overnight workers excreted 77% less 8-hydroxydeoxyguanosine (8-OH-dG) — a biomarker of oxidative DNA repair — during daytime sleep compared to their own circadian-aligned sleep (p=0.03). Higher melatonin metabolite levels correlated with elevated 8-OH-dG excretion, consistent with melatonin’s role in driving nocturnal oxidative DNA repair. As melatonin production falls with age, this repair capacity falls in parallel.
A 2024 study by Hu et al. identified one downstream consequence of mitochondrial DNA stress in the brain. In sleep-deprived rats, oxidized mitochondrial DNA in microglia — the brain’s immune cells — escaped into the cytoplasm, where it activated inflammatory pathways — including TLR9, cGAS-STING, and NLRP3 — associated with NF-kB-mediated neuroinflammatory cascades. This cascade — from sleep deprivation to mitochondrial DNA oxidation to neuroinflammation — was reversed by blocking the mitochondrial DNA-release step with hydroxytyrosol butyrate (Hu et al., 2024). Whether this same cascade works identically in humans has not been shown.
Does Losing Deep Sleep Accelerate Brain Aging?
The Vaccaro et al. (2020) study in Cell established that sleep deprivation causes death through gut-specific ROS accumulation — not through direct neurological stress. In Drosophila and mice, lethality was reversed by oral antioxidants or genetically engineered gut-targeted antioxidant enzymes. ROS was the causal killing agent. This means sleep pressure may be encoded at least in part by peripheral ROS accumulation, not solely by central adenosine accumulation. The implication for aging: as antioxidant defenses reduction with age, the same amount of wakefulness produces more oxidative stress, and the same amount of sleep clears less of it.
In rats, Hu et al. (2024) identified the cascade connecting mitochondrial stress to cognitive impairment: sleep deprivation oxidizes mitochondrial DNA in microglia, oxidized mitochondrial DNA escapes into the cytoplasm, cytoplasmic mitochondrial DNA activates inflammatory pathways — including TLR9, cGAS-STING, and NLRP3 — which are associated with NF-kB activation and pro-inflammatory cytokine production. The resulting neuroinflammation impaired performance on Morris water maze testing — a measure of spatial learning and memory in rodents. Blocking the mitochondrial DNA-release step interrupted the cascade and reversed the cognitive deficits. Whether this microglial cascade is a primary driver of age-related cognitive reduction in humans remains to be established.
The aging convergence involves multiple lower pathways compounding at the same time. NRF2 activity is known to reduction with age. BMAL1 expression is known to reduction with age. Mitochondrial DNA copy number reductions with reduced sleep quantity and quality (Wrede et al., 2015; Han et al., 2024). Each of these reductions weakens a different component of the same repair infrastructure — the mitochondrial restoration that occurs during deep sleep. The Richardson and Mailloux (2023) framework positions this convergence as a mechanistic explanation for why deep sleep reduction and neurodegeneration track together across aging populations.
The mechanisms are identified. The reduction is measurable. Actionable targets exist — including NRF2-activating compounds (covered in the CoQ10 and PQQ article), exercise-induced mitochondrial biogenesis (covered in the exercise and mitochondria article), and addressing sleep fragmentation directly (covered in the sleep fragmentation article).
Age-related mitochondrial reduction might not be the only factor degrading your sleep. Many people over 40 have overlapping metabolic, hormonal, or inflammatory processes that compound the mitochondrial changes described here. When multiple causes are active, the reduction in deep sleep and the difficulty staying asleep might reflect more than one mechanism breaking down at the same time.
Find out which causes might be driving your 3am wakeups –>
Do Older People Need Less Sleep or Just Get Less Deep Sleep?
The distinction between sleep need and sleep capacity is central. Richardson and Mailloux (2023) established that sleep is the phase during which mitochondria undergo fusion, redox balance is restored, and immune responses are facilitated. The need for these functions does not decrease as a person ages — if anything, aging increases the need for mitochondrial repair because oxidative stress builds faster when antioxidant defenses weaken.
What degrades is the machinery that generates deep sleep. BMAL1 reduction decouples mitochondrial physiology from the circadian cycle. NRF2 reduction weakens the antioxidant gene expression that responds to sleep-wake redox cycling. Accepting reduced deep sleep as a normal part of aging may accelerate the mitochondrial stress that drives further sleep reduction — creating a compounding vulnerability where the repair phase shortens as the stress it needs to address increases.
Can You Measure Your Mitochondrial Health Through Sleep Data?
Han et al. (2024) found that among individual sleep components, sleep latency was the independent predictor of mitochondrial DNA copy number (beta = -0.16, p = 0.011). Sleep duration and sleep efficiency did not independently reach significance in that study. The selectivity of sleep latency suggests that autonomic arousal at sleep onset — the physiological activation that delays falling asleep — may be more biologically relevant to mitochondrial DNA integrity than total hours in bed.
Wrede et al. (2015) found that sleep efficiency — the percentage of time in bed spent asleep — predicted mitochondrial DNA copy number more strongly than sleep duration (beta = 0.51 per 1% decrease). Consumer wearable devices (Oura, Whoop, Apple Watch) track sleep latency, sleep efficiency, and deep sleep duration. While these devices do not directly measure mitochondrial function, lower trends in deep sleep percentage, rising sleep latency, and falling sleep efficiency over months or years may reflect underlying changes in mitochondrial capacity.
Is the Age-Related Reduction in Sleep Quality Reversible?
Sarnataro et al. (2025) demonstrated in Drosophila that mitochondrial fragmentation, enhanced mitophagy, and expanded mitochondria-ER contact sites — all caused by sleep deprivation — were reversed after recovery sleep. The morphometric parameters returned to baseline or overcorrected. This establishes that the mitochondrial changes driving sleep pressure are dynamic and responsive to recovery, not permanently fixed. Whether the same degree of reversibility applies in aged human neurons is not yet known.
Davinelli et al. (2024) identified NRF2 modulators as promising candidates for sleep-related oxidative impairment. Restoring NRF2 activity restores antioxidant gene expression, which may help address the oxidative vulnerability that builds with age. Exercise-induced mitochondrial biogenesis (covered in detail in the exercise and mitochondria article) represents a well-studied approach for rebuilding mitochondrial capacity. Mitochondrial-targeted supplements are reviewed in the CoQ10 and PQQ article.
Reversibility is partial and depends on sustained action. Recovery sleep restores acute mitochondrial stress. Rebuilding the long-term reduction in mitochondrial capacity, NRF2 expression, and circadian clock gene activity requires sustained changes in behavior, metabolic inputs, or both.
Does Poor Sleep Timing Matter as Much as Sleep Duration for Mitochondrial Health?
Bhatti et al. (2016) measured 8-hydroxydeoxyguanosine (8-OH-dG) excretion — a validated biomarker of oxidative DNA repair — during sleep periods among 217 daytime and 223 overnight workers. The 77% reduction in 8-OH-dG excretion during circadian-misaligned daytime sleep (p=0.03) suggests that circadian phase is an important factor in the magnitude of oxidative DNA repair.
Higher melatonin metabolite levels correlated with higher DNA repair activity, positioning age-related melatonin reduction as a compounding factor. As melatonin production falls with age, even circadian-aligned sleep produces less oxidative DNA repair than it did in earlier decades. Consistent sleep timing — going to bed and waking at the same circadian phase each day — may be as important as sleep duration for maintaining mitochondrial DNA integrity after 40.
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep — the pillar article for metabolic sleep disruption, covering glucose regulation, cortisol rhythm, mitochondrial stress, NAD+ reduction, and fat metabolism
- Why Are You Exhausted But Can’t Sleep? Is It a Mitochondrial Energy Paradox? — mitochondrial fatigue, unrefreshing sleep, ATP production, and why rest may not restore cellular energy
- Can CoQ10 and PQQ Supplements Improve Your Sleep? — CoQ10, PQQ, magnesium, supplement timing, and the evidence for mitochondrial sleep support
- Is Your 3am Wakeup a Metabolic Emergency? Blood Sugar, Cortisol, and Mitochondrial reduction? — the 2-4am vulnerability window, glucose drops, cortisol rescue, and mitochondrial energy buffering
- Why Are You Wired but Tired, and What Role Do Mitochondria Play? — hyperarousal, cortisol rhythm changes, stress physiology, and mitochondrial power-down reduction
- How Does Exercise Rebuild Your Mitochondria and Fix Your Sleep? — exercise type, timing, mitochondrial biogenesis, PGC-1?, and sleep architecture
- What Is the Measured Cellular Impact of Sleep Fragmentation? — fragmented sleep, oxidative stress, vascular injury, inflammation, and cellular aging impact
References
Vaccaro, A., Kaplan Dor, Y., Nambara, K., Pollina, E. A., Lin, C., Greenberg, M. E., & Rogulja, D. (2020). Sleep reduction can cause death through accumulation of ROS in the gut. Cell, 181(6), 1307-1328.e15. https://pubmed.ncbi.nlm.nih.gov/32502393/
Richardson, R. B., & Mailloux, R. J. (2023). Mitochondria need their sleep: Redox, bioenergetics, and temperature regulation of circadian rhythms and the role of cysteine-mediated redox pathway activity, uncoupling proteins, and substrate cycles. Antioxidants, 12(3), 674. https://pubmed.ncbi.nlm.nih.gov/36978924/
Mezhnina, V., Ebeigbe, O. P., Poe, A., & Kondratov, R. V. (2022). Circadian control of mitochondria in ROS homeostasis. Antioxidants & Redox Signaling, 37(10-12), 647-663. https://pubmed.ncbi.nlm.nih.gov/35072523/
Wrede, J. E., Mengel-From, J., Buchwald, D., Vitiello, M. V., Bamshad, M., Noonan, C., Christiansen, L., Christensen, K., & Watson, N. F. (2015). Mitochondrial DNA copy number in sleep duration discordant monozygotic twins. Sleep, 38(10), 1655-1658. https://pubmed.ncbi.nlm.nih.gov/26039967/
Bhatti, P., Mirick, D. K., Randolph, T. W., Gong, J., Buchanan, D. T., Zhang, J. J., & Davis, S. (2016). Oxidative DNA stress during sleep periods among overnight workers. Occupational and Environmental Medicine, 73(8), 537-544. https://pubmed.ncbi.nlm.nih.gov/27307003/
Sarnataro, R., Velasco, C. D., Monaco, N., Kempf, A., & Miesenboeck, G. (2025). Mitochondrial origins of the pressure to sleep. Nature, 645(8081), 722-728. https://pubmed.ncbi.nlm.nih.gov/40670797/
Sarnataro, R. (2025). Neurophysiology of mitochondrial dynamics in sleep. The Journal of Physiology, 603(22), 6747-6762. https://pubmed.ncbi.nlm.nih.gov/40846498/
Davinelli, S., Medoro, A., Savino, R., & Scapagnini, G. (2024). Sleep and oxidative stress: Current perspectives on the role of NRF2. Cellular and Molecular Neurobiology, 44(1), 52. https://pubmed.ncbi.nlm.nih.gov/38916679/
Han, S., Kim, D. K., Jun, S. E., & Kim, N. (2024). Association of sleep quality and mitochondrial DNA copy number in healthy middle-aged adults. Sleep Medicine, 113, 19-24. https://pubmed.ncbi.nlm.nih.gov/37979503/
Terzi, A., Ngo, K. J., & Mourrain, P. (2024). Phylogenetic conservation of the interdependent homeostatic relationship of sleep regulation and redox metabolism. Journal of Comparative Physiology B, 194(3), 241-252. https://pubmed.ncbi.nlm.nih.gov/38324048/
Hu, Y., Wang, Y., Wang, Y., Zhang, Y., Wang, Z., Xu, X., Zhang, T., Zhang, T., Zhang, S., Hu, R., Shi, L., Wang, X., Li, J., Shen, H., Liu, J., Noda, M., Peng, Y., & Long, J. (2024). Sleep deprivation triggers mitochondrial DNA release in microglia to induce neural inflammation: Preventative effect of hydroxytyrosol butyrate. Antioxidants, 13(7), 833. https://pubmed.ncbi.nlm.nih.gov/39061901/
Reference Metadata Changes
| # | Field | Draft Value | PubMed Value | Action |
|—|——-|————-|————–|——–|
| 1 | Title case | "Sleep Reduction Can Cause Death…" (title case) | "Sleep reduction can cause death…" (sentence case) | Corrected to sentence case |
| 2 | Title case | "Mitochondria Need Their Sleep…" (title case) | "Mitochondria need their sleep…" (sentence case) | Corrected to sentence case |
| 4 | Pages | 1655-8 | 1655-1658 | Corrected to full page range |
| 5 | Pages | 537-44 | 537-544 | Corrected to full page range |
| 6 | Volume/issue/pages | Missing | 645(8081), 722-728 | Added from PubMed |
| 6 | Author name | Miesenboeck | Miesenboeck (PubMed renders without umlaut) | Acceptable as-is |
| All | Author names | All verified | All match PubMed | No fabricated authors |
Written by Kat Fu, M.S., M.S.? Last reviewed: May 2026? 11 references cited
