Section 1: Why an AI that “reads” your sleep matters after 40
You already know sleep is more than just feeling rested.
How you sleep is tied to memory, mood, blood pressure, blood sugar, and whether you can show up for the people and activities that matter to you. For years, though, the tools we’ve had to measure sleep and long-term health risk have been fairly blunt: “how many hours,” questionnaires, or a diagnosis like sleep apnea.
Now something new is arriving: large AI models that look at every second of your sleep study and try to map that pattern to future health. One of the latest examples is the 2026 project, SleepFM—developed by a multi-institution collaboration including researchers at Stanford & Harvard.
SleepFM is a “foundation model” trained on more than half a million hours of clinical polysomnography (full overnight sleep studies with EEG, breathing, heart rhythm, and more).
From one overnight sleep study, it can estimate risk for conditions ranging from dementia to heart failure and all-cause mortality.
At the same time, other human studies are sharpening the picture of which features of sleep matter most in later life: how much deep slow-wave sleep you get, how stable your emotional brain feels after sleep, and how “old” or “young” your brain looks.
In this article, we’ll cover:
- How the 2026 SleepFM study uses one overnight sleep study to predict risk for about 130 conditions, including dementia and cardiovascular disease.
- What new work in older adults shows about deep non-REM slow-wave sleep and anxiety, and why that matters for brain aging..
- How slow-wave sleep loss over years relates to your chance of developing dementia in late life.
- How deep-learning models that estimate your “sleep age” from overnight studies connect to life expectancy.
All of this will stay grounded in what you can actually do with this knowledge: how to think about getting a sleep study, how to protect the parts of sleep that seem most tightly linked to brain and heart health, and how to view these new AI tools in a measured helpful way.
Let’s get started.
Study 1 – 2026 SleepFM model learning from clinical sleep studies
Study 1 link: “A multimodal sleep foundation model for disease prediction.”
In 2026, a Stanford led team introduced SleepFM, a large AI model trained on more than 585,000 hours of overnight polysomnography from over 65,000 people across multiple sleep cohorts. These cohorts ranged from general sleep-clinic populations to community studies of older adults, where participants are mostly in midlife and beyond.
Instead of scoring each sleep study into a small set of summary numbers and diagnoses, SleepFM takes in the full minute-by-minute recordings: brain waves (EEG), breathing effort, oxygen levels, heart rhythm, and muscle tone. It uses self-supervised learning to compress these into a single 128-dimensional representation for each person, then fine-tunes lightweight prediction “heads” on top of that representation for different outcomes (sleep staging, apnea, age, and disease risk).
The most interesting part of SleepFM is what it can see from one overnight sleep study: future risk for death, dementia, cardiovascular events, kidney disease, and more. When the authors evaluated 1,041 disease phenotypes using electronic health record follow-up, SleepFM could predict 130 conditions with a concordance index (C-index) of at least 0.75.
That means, for those conditions, if you randomly pick two people, the model puts the higher-risk person above the lower-risk person at least 75% of the time.
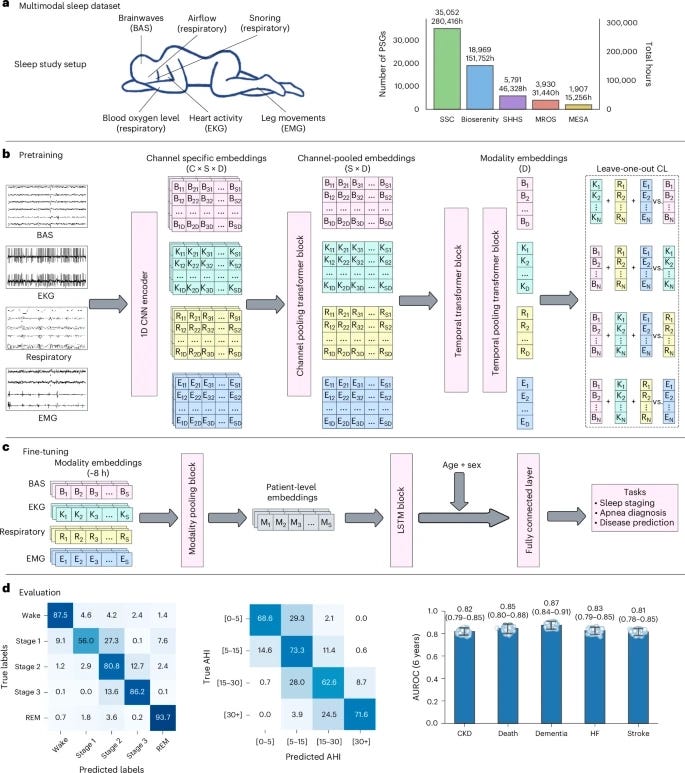
For key outcomes that matter in later life, performance was higher:
- For all-cause mortality, C-index ~0.84.
- For dementia, C-index ~0.85.
- For myocardial infarction, heart failure, chronic kidney disease, and stroke, C-indices ranged from about 0.78 to 0.81.
These results were not just driven by age and standard risk factors.
SleepFM was compared against two baselines: a model that used only simple demographic information (age, sex, BMI, race/ethnicity), and a “from-scratch” network trained directly on the raw sleep recordings without pretraining.
Key findings for SleepFM and older adults
- Disease prediction from one overnight study.
Using a single overnight sleep study per person, SleepFM predicted risk for 1,041 electronic-health-record phenotypes. It achieved a C-index ≥0.75 for 130 conditions, spanning dementia, cardiovascular disease, kidney disease, and mortality. This suggests that overnight sleep contains much richer information about long-term health than traditional scoring taps into.
- Better than “age and risk factors” alone.
Compared with a model that used only age, sex, BMI, and race/ethnicity, SleepFM achieved higher C-indices across many diseases. For mortality in particular, a SleepFM model fine-tuned with limited labeled data outperformed the demographics model trained on several times more data. This suggests that detailed overnight patterns carry extra information beyond what a standard clinic history provides.
- Competitive on standard sleep tasks.
While disease prediction is the headline, SleepFM also matched or exceeded state-of-the-art models on sleep staging and sleep apnea classification, with F1 scores around 0.70–0.78 for staging and accuracies of about 69% for four-level apnea severity and 87% for the presence of at least moderate sleep apnea across several cohorts.
For you as an individual, the key takeaway is not that SleepFM can tell you exactly what will happen from one sleep study.
Rather, it shows that your overnight sleep patterns carry a surprisingly rich imprint of future health risk—one that can be uncovered when you combine large datasets with modern AI methods.
At the same time, its important to note that the authors present SleepFM as a research model: they detail important limitations, highlight the need for further work on interpretability and generalization, and do not frame it as a ready-to-use clinical test. Any move toward routine clinical use will require additional validation, prospective studies, and careful ethical and implementation work.
Study 2 – 2026 Berkeley study on slow-wave sleep and anxiety in older adults
Study 2 link: “Impaired slow-wave sleep accounts for brain aging-related increases in anxiety.”
In 2026, Eti Ben Simon, Matthew Walker, and colleagues asked a different but related question: How does deep non-REM slow-wave sleep help older adults keep their emotions steady?
They studied 61 cognitively healthy older adults, all over 65, from the Berkeley Aging Cohort. Each participant spent a night in the sleep lab with full polysomnography. The researchers measured slow-wave activity (SWA) during non-REM sleep—the large, synchronized brain waves that dominate deep sleep. The next morning, participants underwent structural MRI to measure atrophy in emotion-sensitive brain regions and completed anxiety assessments. A subset of 24 individuals returned for follow-up assessments over about four years.
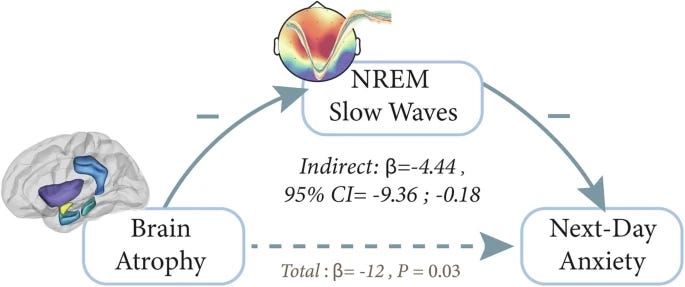
The central idea was simple but powerful: as the brain ages, some regions involved in emotional regulation shrink. At the same time, older adults often report more anxiety and disturbed sleep. The team wanted to know whether changes in slow-wave activity form a bridge between these two trends.
They found that older adults with lower SWA during the lab sleep study tended to experience higher anxiety the next day, and this association held up both at baseline and at follow-up. MRI showed that atrophy in specific emotion-processing areas was tied to lower SWA.
Key findings for deep sleep and emotional stability
- Less slow-wave activity linked to higher anxiety.
Individuals with more impaired SWA during non-REM sleep showed higher next-day anxiety scores at baseline, and the same pattern appeared at follow-up. This suggests that deep sleep is not only about memory and physical restoration but also about overnight emotional recalibration.
- Brain atrophy tied to weaker slow waves.
Structural MRI revealed that shrinkage in key emotion-processing regions was associated with reduced capacity to generate high-quality slow waves. In other words, structural brain aging and changes in deep sleep went hand in hand.
- SWA as the bridge between brain structure and anxiety.
Mediation analysis indicated that SWA largely carried the relationship between regional atrophy and next-day anxiety. Once SWA was included in the model, the direct link from atrophy to anxiety was greatly reduced. That frames deep sleep as a potential buffer: when SWA is preserved, emotional stability may be better protected even in the presence of brain aging.
The authors highlight that late-life anxiety and emotional dysregulation are tied to faster cognitive decline and higher dementia risk. While this particular study did not follow people long enough to track dementia directly, it strengthens the idea that protecting deep non-REM sleep is one way to protect both emotional and cognitive health in later life.
Study 3 – 2023 slow-wave sleep loss and later dementia
Study 3 link: “Association Between Slow-Wave Sleep Loss and Incident Dementia.”
In 2023, an analysis from the Framingham Heart Study took a longer view: Does losing deep slow-wave sleep over time predict who will go on to develop dementia?
The researchers focused on 346 participants (mean age about 69 years; range 60–87) who had two overnight polysomnography studies spaced several years apart. By comparing these two studies, they calculated the annual change in the percentage of sleep spent in slow-wave sleep (SWS). They then followed participants for up to 17 years to see who developed dementia.
The main result: each percentage-point per year drop in SWS was associated with a meaningful increase in dementia risk, even after adjusting for age, sex, genetic risk (APOE ε4), medication use, and other factors. SWS declined naturally with age, but those with steeper declines were more likely to develop dementia.
The team also examined whether Alzheimer’s genetic risk or hippocampal volume (from MRI) was related to SWS loss. They found that carriers of the APOE ε4 allele lost SWS more quickly, but hippocampal volume at baseline did not show a clear association with SWS change.
Key findings for SWS and dementia
- Steeper loss of SWS linked to higher dementia risk.
After adjusting for demographic and clinical factors, each 1-percentage-point decline in SWS per year was associated with a 27% higher risk of dementia (hazard ratio 1.27; 95% CI 1.06–1.54).
- Genetic risk accelerates SWS loss.
Participants carrying at least one APOE ε4 allele showed faster SWS loss with aging than non-carriers, even though hippocampal volume did not explain this difference. This points to an interaction between genetic risk and sleep architecture.
- Potentially modifiable risk factor.
The authors conclude that SWS loss with aging may be a modifiable risk factor for dementia. That doesn’t mean boosting deep sleep will definitely prevent dementia, but it strengthens the case for treating sleep as part of dementia risk management.
Study 4 – 2022 sleep-derived “age” and life expectancy
Study 4 link: “Age estimation from sleep studies using deep learning predicts life expectancy.”
In a 2022 multi institution collaboration including the Technical Univeristy of Denmark, Stanford, Mass Gen, researchers took yet another angle: What if we ask a deep-learning model to guess your age from a single overnight sleep study, and then see whether looking “older” or “younger” than your actual age relates to mortality?
Their model was trained on 2,500 polysomnography recordings and then tested on 10,699 overnight studies from seven cohorts that together covered adults between 20 and 90 years old. The model’s job was to estimate chronological age from the full overnight recordings. The difference between predicted age and actual age was called age estimation error (AEE): positive values meant the sleep study looked “older” than the person’s calendar age, negative values meant “younger.”
Compared with traditional sleep summary metrics, the deep-learning age estimator was more accurate. More importantly, AEE carried information about survival: people whose sleep looked older than their actual age tended to have higher mortality risk during follow-up, even after accounting for demographics, sleep characteristics, and health covariates.
Key findings for “sleep age” and mortality
- More accurate age estimation than standard metrics.
Traditional sleep scoring measures estimated age with an MAE of around 14.9 ± 6.3 years, whereas the deep-learning model reduced the error to about 5.8 ± 1.6 years. That suggests the overnight recordings contain a detailed imprint of biological aging that other indices do not capture.
- Age estimation error predicts mortality.
After adjusting for demographics, sleep variables, and health factors, each 10-year increment in AEE was associated with a 29% higher all-cause mortality rate. An increase in AEE from −10 to +10 years corresponded to an estimated 8.7-year shorter life expectancy.
- Sleep as a window into overall aging.
The authors interpret AEE as a proxy for “sleep age,” reflecting how old your physiology looks during sleep relative to your birth date. Someone whose sleep looks much older than their chronological age may have accumulated more health burdens, while a younger-looking sleep pattern could reflect better preserved systems.
This work and SleepFM share key ideas: use the full overnight recording, train deep models at scale, and treat certain outputs (like age or disease-risk scores) as summaries of many smaller sleep features. Together, they suggest that overnight sleep pattern is a rich, underused biomarker for aging and longevity.
The take-home message is that the way you sleep tonight is intertwined with the health of your brain, heart, and metabolism, and that detailed sleep data—interpreted carefully—could become part of future risk assessment.
If your sleep feels less restorative than it used to — and you’re wondering which part of the equation is involved — my free 3AM Decoder maps 3am disruption patterns across 5 potential contributors. It takes < 2 minutes.
How to use this: 5 ways to act on AI-driven sleep science in midlife and beyond
Modern models like SleepFM show that one overnight clinical sleep study can encode meaningful health information. Smaller but carefully run cohorts in older adults show that deep non-REM slow-wave sleep is closely tied to emotional stability and dementia risk, and that deep-learning age estimates from sleep studies relate to life expectancy.
You cannot control your genes. But you can influence how proactive you are in resolving sleep problems, how you protect deep sleep, and how you think about AI tools now positioning sleep itself as a biomarker.
Here are five practical steps, grounded in the evidence above:
- Treat persistent sleep problems as a primary health pillar.
The SleepFM work shows that overnight sleep carries information about future dementia, cardiovascular disease, kidney disease, and mortality, and the “sleep age” study shows that how old your sleep looks relative to your calendar age relates to all-cause mortality risk. If you are dealing with long-standing insomnia, short sleep, 3am wakeups, gasping in sleep, or unrefreshing sleep, consider prioritizing addressing these sleep patterns so you aim for a 7-8 hours of continuous restful sleep.
- Ask whether a full overnight sleep study makes sense for you.
For people with possible sleep apnea, restless legs, or unexplained daytime sleepiness, an overnight study (in lab or with high-quality home equipment) can uncover conditions that fragment deep sleep and disrupt oxygen levels.
- Protect deep non-REM slow-wave sleep through everyday habits.
The anxiety and dementia studies both point to SWS as a key component of healthy aging—supporting emotional regulation overnight and relating to lower dementia risk when it declines more slowly. You can support SWS by keeping a regular sleep schedule, limiting alcohol close to bedtime, getting daylight exposure early in the day, and staying physically active.
- Pair emotional health care with sleep care.
In the Berkeley study, impaired slow-wave sleep linked brain atrophy to next-day anxiety in older adults, and anxiety itself is tied to faster cognitive decline. If you notice your anxiety rising along with worsening sleep, consider these as connected issues. Approaches such as cognitive behavioral therapy for insomnia, and, when appropriate, evidence-based therapy for anxiety can help ease both daytime distress and overnight arousal that disrupts deep sleep.
- Stay curious but cautious about AI tools built on sleep data.
SleepFM and the “sleep age” model show that AI can pull out health-relevant information from sleep studies that humans do not routinely use. In the coming years, we will see more AI-based scores on sleep reports or consumer devices. Use them as prompts for conversation—questions like “What might this score mean for my risk of dementia or heart disease?”—rather than as definitive answers. Always anchor decisions about testing, medication, or major lifestyle changes in research you have verified on your own and/or via discussions with a provider.
Putting everything together, the takeaway here is not that one night in a sleep lab can predict your future with certainty. It is that your sleep contains many layers of information about how your brain and body are aging—and that large models like SleepFM, along with carefully designed cohort studies, are finally starting to decode that information.
By treating sleep problems seriously, considering formal evaluation, protecting deep non-REM sleep, and engaging thoughtfully with emerging AI tools, you are investing in a future where your brain, heart, and emotional life stay as healthy as possible for as long as possible.
—Kat & Heather
References
- Thapa, R., Kjaer, M. R., He, B., Covert, I., Moore, H., Hanif, U., Ganjoo, G., Westover, M. B., Jennum, P., Brink-Kjaer, A., Mignot, E., & Zou, J. (2026). A multimodal sleep foundation model for disease prediction. Nature Medicine. Advance online publication. doi:10.1038/s41591-025-04133-4.
- Ben Simon, E., Shah, V. D., Murillo, O., Zsofia, Z., & Walker, M. P. (2026). Impaired slow-wave sleep accounts for brain aging-related increases in anxiety. Communications Psychology. Advance online publication.
- Himali, J. J., et al. (2023). Association between slow-wave sleep loss and incident dementia. JAMA Neurology, 80(1), 32–41.
- Brink-Kjaer, A., Leary, E. B., Sun, H., Westover, M. B., Stone, K. L., Peppard, P. E., Lane, N. E., Cawthon, P. M., Redline, S., Jennum, P., Sorensen, H. B. D., & Mignot, E. (2022). Age estimation from sleep studies using deep learning predicts life expectancy. npj Digital Medicine, 5(1), 103. doi:10.1038/s41746-022-00630-9.
