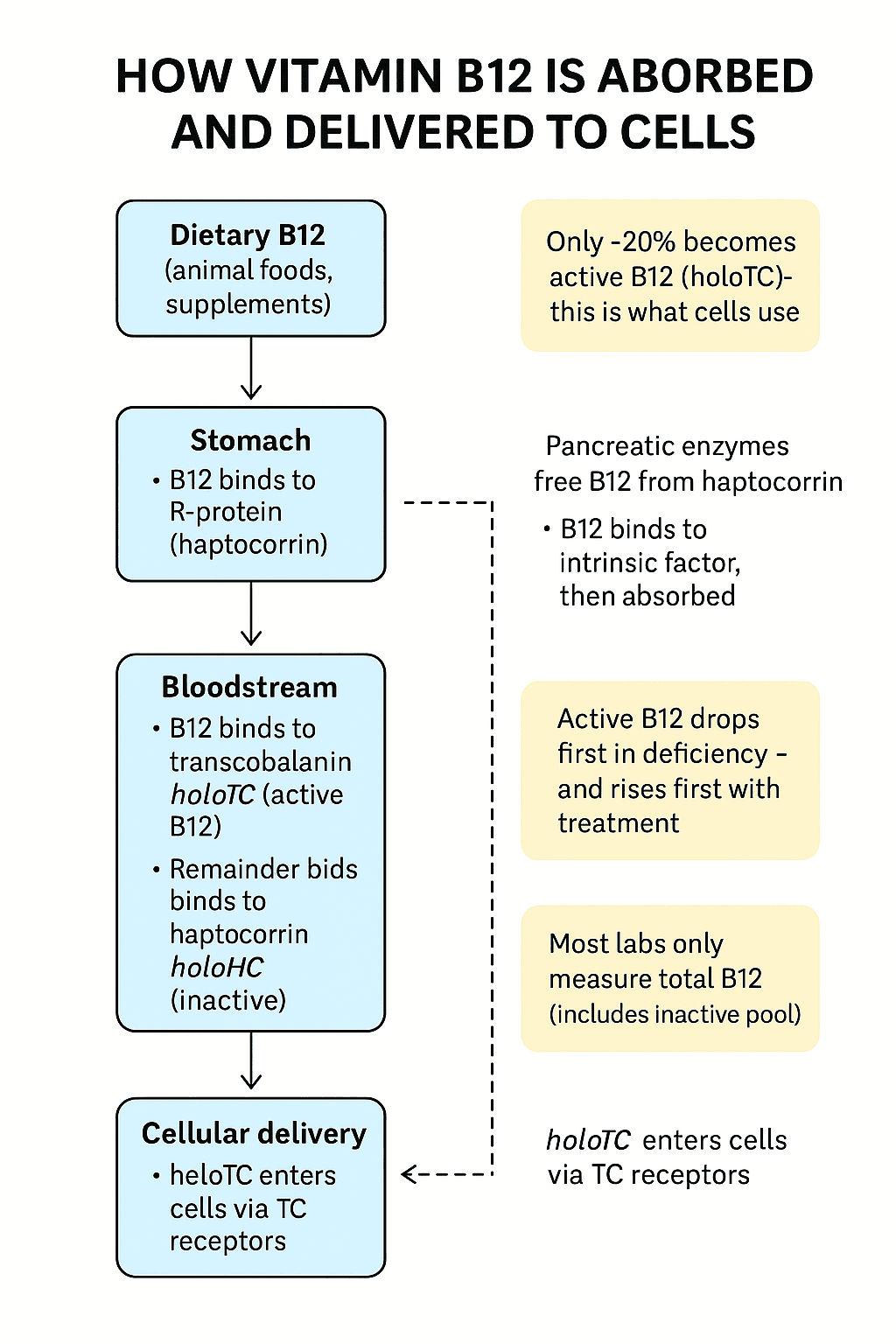
The standard vitamin B12 blood test measures total circulating B12—but the larger share of that is biologically inactive. The fraction that reaches cells is active B12 (holotranscobalamin), which makes up an average of ~20% of total serum B12 and is rarely measured by default. A 2025 UCSF study found that participants with lower active B12 had more white matter damage, slower cognitive processing, and higher neurodegeneration markers—despite normal total B12.
- About 70–90% of circulating B12 is bound to haptocorrin, which carries B12 back to the liver rather than to cells; the remaining 10–30% is holotranscobalamin—the bioavailable fraction cells can absorb.
- Active B12 (holotranscobalamin) is the first marker to drop in deficiency and the first to recover with repletion—making it a more dynamic early indicator than total B12; one study found increasing B12 intake led to a ~15% rise in holoTC, compared to just 9% for total B12.
- Methylmalonic acid (MMA) is a highly specific marker of functional B12 deficiency in people with normal kidney function; MMA above 0.4 µmol/L indicates B12 is not functioning at the mitochondrial level even when serum B12 looks normal.
- A homocysteine blood test adds context—levels between 10–15 µmol/L are linked to white matter changes and early cognitive slowing; levels above 15 µmol/L are linked to dementia risk.
- Up to 40% of adults in Western populations fall in the marginal B12 zone of 148–221 pmol/L—below ranges that support cognitive longevity, but not low enough to trigger a practical deficiency diagnosis.
Why Does the Standard Vitamin B12 Blood Test Miss Deficiency?
Your B12 level came back “normal.”
But that doesn’t mean it’s doing its job.
Standard blood tests report the total amount of B12 in your blood. But most of that is locked away—bound to a form your cells can’t use. The B12 that actually reaches your brain, nerves, and mitochondria? That part is rarely measured.
And that’s where problems begin.
As we explained in our previous article, the 2025 UCSF study in healthy individuals showed that even when total B12 appears adequate, early brain changes can already be underway. Participants with lower active B12 had more white matter damage, slower cognitive processing, and higher neurodegeneration markers—despite having “normal” serum B12.
Get Weekly Insights to Personalize Your Own Longevity Roadmap
For anyone focused on cognitive longevity or performance longevity, that’s not a detail to ignore.
The test most people get—total serum B12—can stay in range even when “biological insufficiency” is already affecting memory, energy, and repair capacity. That’s because it includes both active and inactive forms. The active portion, holotranscobalamin, is the only kind your cells can use—and it’s often the first to drop.
You don’t need anemia. You don’t need tingling hands.

If you’re optimizing for cognitive health, energy, or aging resilience, you need the right data.
This guide explains:
- Why “normal” B12 often misses early brain and metabolic changes
- Why a basic vitamin B12 blood test misses early signs and symptoms of vitamin B12 deficiency
- What active B12, MMA, and homocysteine reveal
- How B12 is absorbed and transported—and why it affects test accuracy
- What numbers matter—and when to act, even if you’re symptom-free
- Why longevity-focused adults should test regardless of diet, age, or diagnosis
If you’re relying on total vitamin B12 to assess your cognitive health or aging risk, the full picture might be missing.
This article shows you what to test instead—and how to understand and act on the results.
Table of Contents
What Are the Signs of Vitamin B12 Deficiency in Longevity-Focused Adults?
Clinical deficiency may be rare.
But functional B12 insufficiency—the kind that affects brain repair, metabolism, and cognitive resilience—isn’t.
In high-income countries like the U.S., U.K., and Canada, 3–6% of adults fall below the clinical cutoff of 148 pmol/L. But up to 40% in Western populations land in the marginal zone of 148–221 pmol/L, without hematologic symptoms.
These levels aren’t low enough to trigger a diagnosis.
But they’re common in certain groups:
- Adults 60+, who absorb less B12 due to low stomach acid or gut changes
- Vegans and vegetarians, unless consistently supplementing
- People on long-term metformin, PPIs, or H2 blockers, where B12 transport is impaired
- Anyone with unexplained fatigue, brain fog, or neuropathy
- Health-conscious adults on low animal foods diet
The most overlooked sign and symptoms of vitamin B12 deficiency—like low energy, mood swings, and poor focus—often occur without anemia.
Numbness, tingling, or strange vibration sensations in the feet can show up while blood markers still read as “normal.” These are often written off as stress, aging, or overtraining—especially when only total B12 is measured.
And subclinical deficiency is becoming more common.

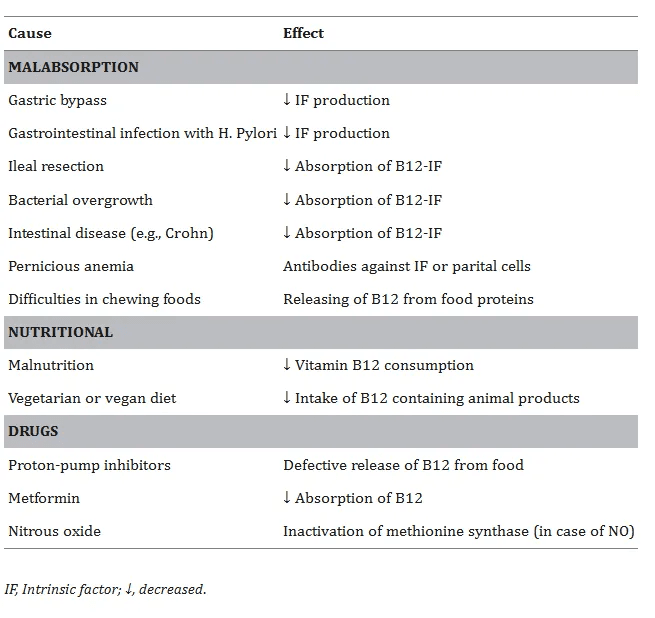
By age 65, up to 20–40% of adults show reduced B12 absorption due to atrophic gastritis and declining stomach acid (Allen, 2008). Common medications like metformin, proton pump inhibitors (PPIs), and H2 blockers further impair uptake. One U.S. study found that long-term metformin use raised B12 deficiency risk by 19% (de Jager et al., 2010), and multiple trials show that PPI use consistently lowers B12 over time (Lam et al., 2013).
The issue is compounded by testing practices. Most doctors only order serum B12, which doesn’t distinguish between active and inactive forms. Active B12 (holotranscobalamin) and MMA—the markers that reflect what’s reaching cells—are rarely automatically included.
These patterns don’t always show up on a CBC.
But they matter—because both MMA and homocysteine are linked to brain atrophy, cognitive decline, and higher mortality, even in adults with no anemia and “normal” total B12. For longevity-driven individuals—especially those over 50, on medication, don’t wait for the sign and symptoms of vitamin B12 deficiency—functional testing offers a better early-detection system.
How Common Is Vitamin B12 Deficiency Across Different Populations?
| Group | Deficient (%)(~<148 pmol/L) | Marginal/Low (%)(~148–258 pmol/L) | Region | Notes |
|---|---|---|---|---|
| General adult population | 3–6% | Up to 40% | US / UK | NHANES data; marginal status includes many asymptomatic cases |
| Older adults (60+) | 4–15% | Up to 43% | US / Canada / Europe | Due to reduced absorption; higher rates with age |
| Vegans (unsupplemented) | 40–52% | Up to 70% | UK / US / Germany | Serum B12 often <150 pmol/L without supplements |
| Vegetarians (low dairy/egg) | 7–30% | Up to 47% | Western countries / India | Risk varies by intake and fortification |
| General population in India | ~47% | Up to 75% | India | Driven by widespread vegetarian diets and lack of fortification |
| Latin America | ~40% | – | Varies | High due to low animal food intake |
How Is Vitamin B12 Transported, and Why Does the Standard Test Miss Active B12?
The most common vitamin B12 blood test measures total serum B12. It’s what most doctors order—and what most people assume is enough. But total B12 gives you a bulk number. It doesn’t tell you how much B12 your cells can use.
About 70–90% of circulating B12 is bound to haptocorrin—a transport protein that carries B12 back to the liver or binds non-functional B12 analogues. The remaining 10–30% is bound to transcobalamin, forming active vitamin B12 holotranscobalamin (holoTC)—the active vitamin B12 that cells can use.
This active fraction makes up an average of ~20% of total serum B12, but that ratio varies widely between individuals. And importantly, holoTC is the first to be depleted in deficiency—and the first to respond to repletion. That’s because the body prioritizes delivery over storage: when intake drops, it protects tissue supply as long as possible. When intake resumes, holoTC levels rebound rapidly—making the active vitamin B12 blood test a sensitive, dynamic marker of B12 status.
One study found that increasing B12 intake led to a ~15% rise in holoTC—compared to just 9% for total B12—highlighting how quickly active B12 reflects real-time intake. By contrast, total B12 is slower to shift—and easily skewed by changes in haptocorrin or liver output. This is why relying on total B12 can delay detection—especially when comparing active B12 vs total B12.
⚠️ The difference between total B12 and active B12 isn’t minor. Total B12 includes forms that can’t be used by your cells. Active B12 (holotranscobalamin) is the fraction deliverable to tissues—and it’s what determines whether your brain, nerves, and mitochondria are getting what they need.
That distinction matters.
For example, a person may have a total B12 of 300 pmol/L (which looks fine), but if most of it is bound to haptocorrin, their active vitamin B12 may already be critically low. Adding to this complexity: holotranscobalamin has a short half-life—it turns over quickly, responding in near real-time to changes in intake and absorption. That makes it a more sensitive indicator than total B12.
There are also rare cases where total B12 appears normal due to elevated holohaptocorrin, while holoTC is low. This can occur in genetic transcobalamin deficiency or other cases involving protein imbalances.
What Do the Different Vitamin B12 Blood Tests Measure?
To assess B12 status properly, most people need more than one marker. Active B12 (holotranscobalamin) is the most direct—and markers like MMA and homocysteine can still reveal problems that active B12 might miss.
Here’s what each test measures—and what it can (and can’t) tell you.
What Does the Total Vitamin B12 Blood Test Measure?
This is the most commonly ordered test. It measures the total amount of B12 in the bloodstream, including both active and inactive forms.
Most of this B12 is bound to haptocorrin, a protein that delivers B12 back to the liver—not to cells. As a result, total B12 can appear normal when active B12 is low.
- What it measures: All circulating B12—both usable and unusable
- When it’s helpful: As a general screen or baseline in combination with other markers
- Gray zone: 148–258 pmol/L (some labs use 200–300 pg/mL)
- Deficiency cutoff: <148 pmol/L (or <200 pg/mL)
Why Is the Active Vitamin B12 Test (Holotranscobalamin) Important?
Holotranscobalamin—or active B12—is the fraction of B12 that’s available for uptake by cells. Unlike total B12, this marker isolates the biologically active form bound to transcobalamin.
Because it reflects real-time tissue supply, holotranscobalamin is more sensitive to recent changes in intake or absorption.
- What it measures: The bioavailable form of B12 that cells can absorb
- Typical reference range:
- Normal: >55 pmol/L
- Borderline: 35–55 pmol/L
- Deficient: <35 pmol/L
- When to use it: Early detection; useful when total B12 is borderline
- Advantages: Not skewed by changes in haptocorrin or liver disease
(Pathways for Hcy and MMA metabolism in humans)
What Does the Methylmalonic Acid Test Reveal About Vitamin B12 Function?
Methylmalonic acid (MMA) is a metabolic marker that rises when B12 is not functioning at the cellular level. It reflects activity inside mitochondria—specifically, the B12-dependent conversion of methylmalonyl-CoA to succinyl-CoA.
Because this pathway doesn’t depend on other vitamin, this makes the test for methylmalonic acid (MMA) the most specific test for functional B12 deficiency in people with normal kidney function.
- What it measures: Accumulation of methylmalonic acid due to inactive B12 pathways
- Typical reference range:
- Normal: <0.27–0.4 µmol/L
- Elevated: >0.4–0.6 µmol/L
- Markedly elevated: >0.7–1.0 µmol/L
- Key caveat: Methylmalonic acid (MMA) rises with impaired kidney function, so interpret your methylmalonic acid test alongside eGFR or creatinine
What Does a Homocysteine Blood Test Reveal About B12 and Brain Health?
A homocysteine blood test measures an amino acid that builds up when B12 and folate are insufficient for proper methylation. It’s not specific to B12—folate and B6 deficiencies also raise it—but it adds useful context when interpreted with methylmalonic acid (MMA).
The homocysteine blood test is sensitive but non-specific marker that helps contextualize B12 status when paired with MMA.
- What it measures: A byproduct of disrupted one-carbon metabolism
- Typical reference range:
- Optimal: <10 µmol/L
- Borderline: 10–15 µmol/L
- Elevated: >15 µmol/L
- Note: Influenced by folate, B6, kidney function, and genetics
Test Your B12 Essentials—No Doctor Required
Most doctors skip MMA and homocysteine—and insurance may not cover them. Catch functional B12 loss early. Choose the test you need from major US labs.
How Does Holohaptocorrin (Inactive B12) Skew Your Vitamin B12 Blood Test?
This is the portion of B12 bound to haptocorrin—biologically inactive and not absorbed by most tissues. It isn’t measured directly in standard panels, but high holohaptocorrin can inflate total B12 and give a false sense of sufficiency.
- Why it matters: Explains misleading total B12 results
- Common in: Liver disease, inflammation, some cancers
- Tip: If total B12 is high but symptoms persist, check holoTC and MMA
Vitamin B12 Blood Test Markers Explained: What Each Test Measures
| Marker | What It Measures | Bioavailability | When It Rises or Falls | Best Use |
|---|---|---|---|---|
| Total B12 | All B12 in blood (active + inactive) | ❌ Mostly inactive | Skewed by liver, binding protein changes | General screen (limited by itself) |
| Active B12 (HoloTC) | B12 delivered to tissues | ✅ Bioavailable | Drops first in deficiency, rises quickly | Early detection, borderline cases |
| Methylmalonic Acid | Metabolic buildup when B12 is lacking | ✅ Functional marker | Rises early, more specific than serum B12 | Most sensitive test (with normal kidneys) |
| Homocysteine | One-carbon pathway impairment (B12/folate/B6) | ⚠️ Indirect | Elevated in multiple deficiencies or renal issues | Add-on marker for context |
| Holohaptocorrin | B12 bound to non-usable protein (haptocorrin) | ❌ Inactive | Not measured directly; inflates total B12 | Explains false-high or false-normal readings |
How Should You Interpret Your Vitamin B12 Blood Test Results for Longevity?
Getting a vitamin B12 blood test is easy.
Optimizing your vitamin B12 status—especially for cognitive longevity and aging well—takes more effort context.
As explained earlier, active B12 (holotranscobalamin) is the form that can reach your cells. Total B12, by contrast, is mostly made up of inactive forms.
What Is the Normal Range for Active Vitamin B12 (Holotranscobalamin)?
When tested directly, active vitamin B12 levels are typically interpreted like this:
- Above 55 pmol/L → healthy
- 35–55 pmol/L → borderline
- Below 35 pmol/L → functionally low

Why Do Methylmalonic Acid and Homocysteine Tests Matter for Longevity?
If active B12 testing isn’t available, functional markers offer a more reliable snapshot of how your body is using B12.
- Methylmalonic acid (MMA) rises when intracellular B12 is too low to support mitochondrial function. It’s highly specific—when elevated (above the typical methylmalonic acid normal range of 0.4 µmol/L), it often signals early deficiency, even with a normal serum B12.
- A homocysteine blood test detects when one-carbon metabolism is under strain. It’s influenced by B12, folate, and B6—and has been linked to vascular risk, poor detox capacity, and cognitive decline.
Together, these two markers provide a deeper readout of how B12 is functioning across brain and metabolic systems.
Even modest elevations in homocysteine—between 10 and 15 µmol/L—have been associated with white matter changes and early cognitive slowing. Higher levels (>15 µmol/L) are linked to dementia risk and systemic aging.
This visual shows how homocysteine disrupts brain metabolism at multiple points—impacting oxidative stress, the blood–brain barrier, and neuroinflammatory cascades:

When methylmalonic acid and homocysteine are both elevated, the likelihood of functional B12 deficiency is likely much higher—even when total B12 is mid-range.
How Should You Think About Your Vitamin B12 Results for Longevity?
You’re not just trying to rule out a deficiency—you’re trying to protect your brain for the long haul.
That’s why the standard cutoffs on most lab reports often fall short. For longevity, the goal is to catch subtle signs early, track trends over time, and stay in the range where B12 supports cognition, energy, and nerve repair.
To do that, you need to understand what each marker means, how to use them together, and when—and how—to request the right tests.
- Normal B12, elevated MMA → you may have functional deficiency
- High total B12, low active B12 → classic active B12 vs total B12 mismatch—your body isn’t delivering it to cells
- Slightly high homocysteine + borderline MMA → early methylation stress
- Low active vitamin B12 levels, normal MMA → early depletion; watch for trends and symptoms
These nuances illustrate why the standard vitamin B12 blood test is often insufficient—and why a proactive approach matters if your goal is cognitive longevity and performance longevity, not just avoiding a deficiency label.
Illustrative Reference Ranges for Vitamin B12 Blood Tests, Homocysteine, and Methylmalonic acid (MMA)
| Marker | Optimal Range | Borderline / Watch Zone | Likely Deficient | Notes |
|---|---|---|---|---|
| Total B12 (pmol/L) | > 300 | 200–300 | < 200 | Easily skewed by haptocorrin. Poor sensitivity. Not reliable on its own. |
| Holotranscobalamin (holoTC, pmol/L) | > 55 | 35–55 | < 35 | Best early indicator. Bioavailable B12. |
| Methylmalonic Acid (MMA, µmol/L) | < 0.27 | 0.27–0.4 | > 0.4 | Highly specific. Check kidney function if elevated. |
| Homocysteine (µmol/L) | < 10 | 10–15 | > 15 | Sensitive, but not specific. Also influenced by folate, B6, kidney. |
Note: Ranges may vary slightly by lab. Always interpret with your health care provider.
What Should You Ask Your Doctor About Homocysteine, Methylmalonic Acid, and B12 Testing?
If you’ve already done a vitamin B12 blood test, it was most likely total B12. You can confirm this by checking your lab report—active B12 (holotranscobalamin) is rarely included by default.
If the result looks normal but you’re dealing with brain fog, low energy, or other subtle shifts, it’s worth asking your doctor for a deeper panel.
Here’s what to ask for:
- Methylmalonic acid (MMA) – shows how well B12 is working inside your cells
- Homocysteine blood test – reflects one-carbon metabolism and also depends on folate and B6
- Creatinine or eGFR – helps interpret MMA, especially if it’s elevated
Active B12 is useful if your provider offers it—but it’s not available everywhere. In that case, the test for methylmalonic acid and the homocysteine blood test are more accessible and give valuable insight. They don’t just assess B12 status—they also reflect other systems involved in energy, methylation, and long-term brain health.
If your doctor is unfamiliar with these markers, keep it simple:
“This test only showed total B12, but I’d like to understand how B12 is actually functioning. Can we order a test for methylmalonic acid and a homocysteine blood test?”
And if these tests aren’t available through your provider, they can often be ordered directly.
These tests aren’t just about cognition. Early B12 decline also affects testosterone, sperm motility, and DNA integrity. See how low vitamin B12 plays out in men’s fertility →
When Should You Order an Active B12, Homocysteine, or Methylmalonic Acid Test?
If you’re optimizing for cognitive performance, cognitive longevity or performance longevity, these tests shouldn’t be reserved for crisis management.
They’re most valuable when used early—and regularly.
You don’t have to be in a “high-risk group” to benefit. B12 status can shift without signs, especially if you’ve:
- Cut back on animal foods
- Made recent changes to your supplement stack or macros
- Been tracking intake through apps but not verifying absorption
Serum B12 can stay within range while the test for methylmalonic acid (MMA) and homocysteine blood test creep up.
Running a yearly panel —especially one that includes an active vitamin B12 blood test—gives you that deeper readout—and helps prevent gradual depletion from becoming structural damage.
Whether you’re optimizing or troubleshooting, functional testing closes the gap between “normal” and truly well.
What If Vitamin B12 Is Normal but Methylmalonic Acid or Homocysteine Are High?
This is one of the most common patterns we see—especially in proactive adults who test regularly.
Even when total B12 appears normal—or even high-normal—that doesn’t always mean B12 is reaching cells or supporting function. The UCSF 2025 study showed that cognitive slowing and white matter changes were observed in individuals with B12 levels above 400 pmol/L, especially when active B12 was low or inactive forms were dominant.
This visual from the UCSF study shows how low active B12 and elevated holohaptocorrin correlate with neurodegeneration markers—even in people with B12 levels above clinical cutoffs:
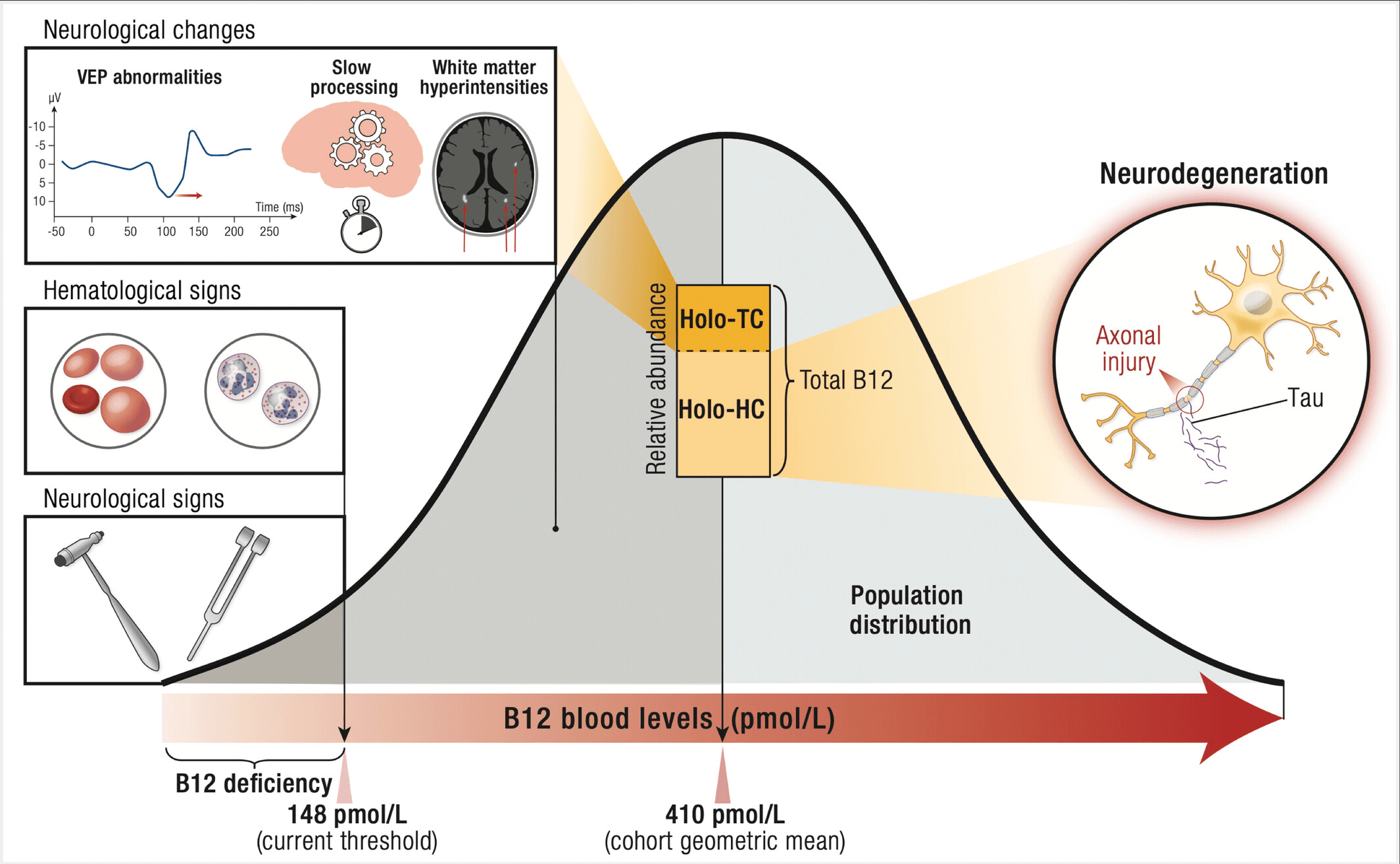
So it’s not just the low end of the range that carries risk.
If methylmalonic acid test results or homocysteine blood test results are elevated—even with total B12 in the 300–500 range—it can still reflect early or functional deficiency.
What to check:
- Methylmalonic acid normal range: <0.27–0.4 µmol/L
- Homocysteine: ideally <10 µmol/L
- Active B12 (if available): >55 pmol/L is within the active vitamin B12 normal range
If methylmalonic acid test (MMA) is >0.4 µmol/L and homocysteine blood test is elevated, even with decent-looking B12 levels, support is warranted.
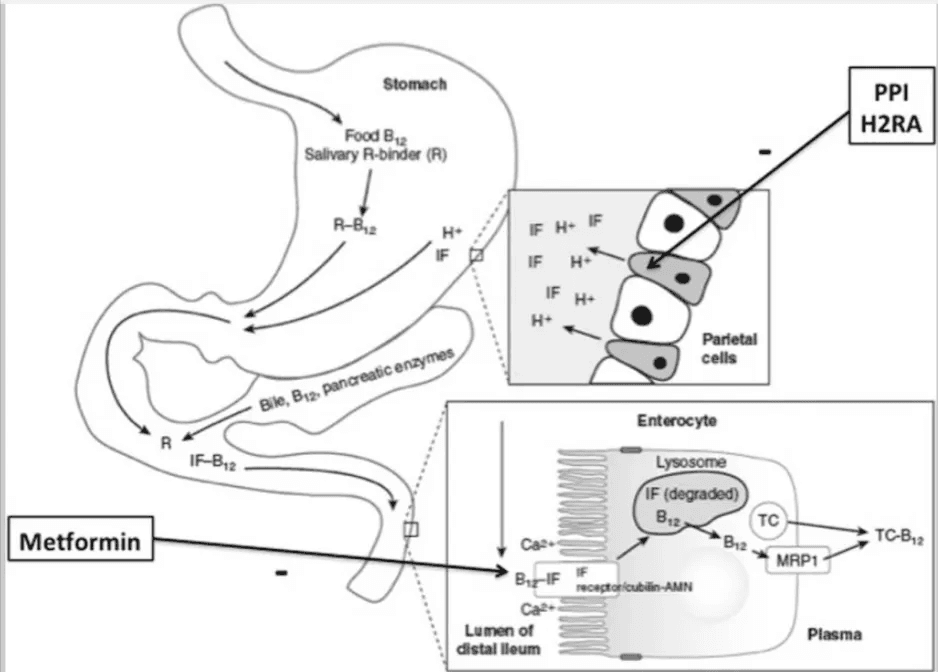
How Does Metformin Affect Vitamin B12 Levels Over Time?
Metformin is increasingly used off-label for longevity, glucose regulation, and age-related disease prevention—but long-term use can impair B12 absorption.
Studies show that B12 and metformin interact over time, even at moderate doses.
You may stay in the normal range for years—until symptoms emerge or a stressor (like surgery or illness) reveals the deficit.
If you’re taking metformin—even in low doses—it’s wise to check active B12 or methylmalonic acid annually, and either adjust your nutrition or supplement if there’s any sign of depletion.
This diagram shows how metformin, PPIs, and H2 blockers interfere with vitamin B12 digestion and cellular uptake—through effects on stomach acid, calcium transport, and intrinsic factor binding.
Who Else Should Consider an Annual Vitamin B12, Homocysteine, and Methylmalonic Acid Panel?
Alongside the general longevity population, these groups often benefit from regular screening:
- Older adults (age 60+): absorption declines naturally with age
- Vegans and vegetarians: B12 is absent from unfortified plant foods
- Post–bariatric surgery patients: intrinsic factor loss can make deficiency permanent
- Long-term users of PPIs or H2 blockers: these drugs impair B12 release from food
In all these cases, a vitamin B12 test, homocysteine blood test, and test for methylmalonic acid are best done at least once a year.
Get Weekly Insights to Personalize Your Own Longevity Roadmap
How Often Should You Repeat the Methylmalonic Acid or Active Vitamin B12 Test?
If you’re supplementing, adjusting your diet, recovering from deficiency, or tracking a borderline case:
- Retest 4-8 weeks after starting support
- Then recheck with a test for methylmalonic acid or an active vitamin B12 blood test every 6–12 months depending on risk.
- If you’re stable and symptom-free, once yearly is usually enough
And if testing is delayed or access is limited? Start supporting now and verify later. B12 is low-risk and easy to correct, but waiting too long can cost you in cognitive performance and repair capacity.
FAQ: Vitamin B12 Blood Test, active vitamin b12 (holotranscobalamin), Homocysteine, and Methylmalonic Acid
What is a vitamin B12 blood test?
A vitamin B12 blood test measures the total amount of B12 in your blood to assess for deficiency or metabolic issues.
It typically measures total B12, which includes both active and inactive forms. But total B12 alone may miss early dysfunction. Many longevity-focused practitioners recommend pairing it with a test for methylmalonic acid, a homocysteine blood test, or an active vitamin B12 blood test—also known as holotranscobalamin.
What is a homocysteine blood test
A homocysteine blood test measures the level of homocysteine in your blood to assess cardiovascular, cognitive, and metabolic health.
High homocysteine may indicate deficiencies in B12, folate, or B6—and is often used alongside a vitamin B12 blood test or a test for methylmalonic acid to catch functional problems earlier than standard panels do.
Are vitamin B12 tests covered by insurance?
Insurance often covers basic B12 tests if there’s a medical reason—but they use standard reference ranges focused on deficiency, not prevention.
Functional markers like methylmalonic acid and homocysteine aren’t always covered. You can order them yourself without a referral, and they’re usually affordable out-of-pocket.
Does Medicare cover vitamin B12 blood tests?
Medicare may cover vitamin B12 blood tests when they’re deemed medically necessary—often based on signs and symptoms of vitamin B12 deficiency.
Typical covered indications include anemia, neuropathy, or gastrointestinal conditions that impair absorption. However, advanced markers like the test for methylmalonic acid, homocysteine blood test or active vitamin B12 holotranscobalamin may vary and often requires clinical justification.
Why are my homocysteine levels high?
The most common reason is insufficient B12, folate, or B6—but kidney function and genetics can also play a role.
If you’re using a homocysteine blood test alongside methylmalonic acid, it can help distinguish B12-related causes. When both are elevated, it usually points to a functional B12 deficit—even if your active vitamin B12 levels haven’t been tested yet.
What is the difference between active B12 and total B12?
Active B12 vs total B12 comes down to what your body can use.
Total B12 includes all circulating forms—most of which are inactive. Active B12, or holotranscobalamin, is the fraction your cells actually absorb. It’s often the first to drop in early deficiency. That’s why some prefer the active vitamin B12 blood test to catch problems total B12 can miss.
Is fasting required for a vitamin B12 test?
No, fasting is not usually required for a standard vitamin B12 blood test.
However, if your test also includes homocysteine or methylmalonic acid, your provider may recommend fasting to improve accuracy—especially for homocysteine, which can be influenced by recent meals.
Why Do You Need More Than a Vitamin B12 Blood Test?
Most people never look beyond their total B12 level. That’s a problem—because as you now know, real issues start earlier.
If you care about preserving brain function, energy, and repair capacity as you age—maximizing performance longevity and cognitive longevity—then testing for function, not just presence, is essential. You need to know what your cells are actually receiving—and how that’s showing up metabolically.
That’s what the test for methylmalonic acid, homocysteine blood test, and (if available) test for active B12 levels reveal.
Early cognitive decline doesn’t wait for your B12 to hit rock bottom. Functional testing helps you catch it while it’s still reversible.
Optimizing for longevity is about not missing the window to do something meaningful, while you still can.
Want to See What Total B12 Misses?
The B12 & Methylation Essentials panel includes homocysteine and methylmalonic acid—two key markers that detect early B12 dysfunction before symptoms appear. These tests aren’t always included in standard panels. Most doctors don’t request them, and insurance may not cover them in all cases.
References
- Office of Dietary Supplements. (n.d.). Vitamin B12: Fact sheet for health professionals. National Institutes of Health. https://ods.od.nih.gov/factsheets/VitaminB12-HealthProfessional/
- Niklewicz A, Hannibal L, Warren M, Ahmadi KR. A systematic review and meta-analysis of functional vitamin B12 status among adult vegans. Nutr Bull. 2024 Dec;49(4):463-479. doi: 10.1111/nbu.12712. Epub 2024 Oct 7. PMID: 39373282.
- Henjum S, Groufh-Jacobsen S, Lindsay A, Raael E, Israelsson AM, Shahab-Ferdows S, Hampel D. Adequate vitamin B12 and folate status of Norwegian vegans and vegetarians. Br J Nutr. 2023 Jun 28;129(12):2076-2083. doi: 10.1017/S0007114522002987. Epub 2022 Sep 27. PMID: 36165126; PMCID: PMC10197078.
- Hannibal L, Lysne V, Bjørke-Monsen AL, Behringer S, Grünert SC, Spiekerkoetter U, Jacobsen DW, Blom HJ. Biomarkers and Algorithms for the Diagnosis of Vitamin B12 Deficiency. Front Mol Biosci. 2016 Jun 27;3:27. doi: 10.3389/fmolb.2016.00027. Erratum in: Front Mol Biosci. 2017 Aug 08;4:53. doi: 10.3389/fmolb.2017.00053. PMID: 27446930; PMCID: PMC4921487.
- Smith AD, Refsum H, Bottiglieri T, Fenech M, Hooshmand B, McCaddon A, Miller JW, Rosenberg IH, Obeid R. Homocysteine and Dementia: An International Consensus Statement. J Alzheimers Dis. 2018;62(2):561-570. doi: 10.3233/JAD-171042. PMID: 29480200; PMCID: PMC5836397.
- Luzzi, S., Cherubini, V., Falsetti, L., Viticchi, G., Silvestrini, M., & Toraldo, A. (2022). Homocysteine, Cognitive Functions, and Degenerative Dementias: State of the Art. Biomedicines, 10(11), 2741. https://doi.org/10.3390/biomedicines10112741
- Seshadri S, Beiser A, Selhub J, Jacques PF, Rosenberg IH, D’Agostino RB, Wilson PW, Wolf PA. Plasma homocysteine as a risk factor for dementia and Alzheimer’s disease. N Engl J Med. 2002 Feb 14;346(7):476-83. doi: 10.1056/NEJMoa011613. PMID: 11844848.
- Miller JW. Proton Pump Inhibitors, H2-Receptor Antagonists, Metformin, and Vitamin B-12 Deficiency: Clinical Implications. Adv Nutr. 2018 Jul 1;9(4):511S-518S. doi: 10.1093/advances/nmy023. PMID: 30032223; PMCID: PMC6054240.
- Gilsing AM, Crowe FL, Lloyd-Wright Z, Sanders TA, Appleby PN, Allen NE, Key TJ. Serum concentrations of vitamin B12 and folate in British male omnivores, vegetarians and vegans: results from a cross-sectional analysis of the EPIC-Oxford cohort study. Eur J Clin Nutr. 2010 Sep;64(9):933-9. doi: 10.1038/ejcn.2010.142. Epub 2010 Jul 21. PMID: 20648045; PMCID: PMC2933506.
- Singla R, Garg A, Surana V, Aggarwal S, Gupta G, Singla S. Vitamin B12 Deficiency is Endemic in Indian Population: A Perspective from North India. Indian J Endocrinol Metab. 2019 Mar-Apr;23(2):211-214. doi: 10.4103/ijem.IJEM_122_19. PMID: 31161105; PMCID: PMC6540890.
- Obeid R, Heil SG, Verhoeven MMA, van den Heuvel EGHM, de Groot LCPGM, Eussen SJPM. Vitamin B12 Intake From Animal Foods, Biomarkers, and Health Aspects. Front Nutr. 2019 Jun 28;6:93. doi: 10.3389/fnut.2019.00093. PMID: 31316992; PMCID: PMC6611390.
- de Jager J, Kooy A, Lehert P, Wulffelé MG, van der Kolk J, Bets D, Verburg J, Donker AJ, Stehouwer CD. Long term treatment with metformin in patients with type 2 diabetes and risk of vitamin B-12 deficiency: randomised placebo controlled trial. BMJ. 2010 May 20;340:c2181. doi: 10.1136/bmj.c2181. PMID: 20488910; PMCID: PMC2874129.
- Allen LH. How common is vitamin B-12 deficiency? Am J Clin Nutr. 2009 Feb;89(2):693S-6S. doi: 10.3945/ajcn.2008.26947A. Epub 2008 Dec 30. PMID: 19116323.
- Lam JR, Schneider JL, Zhao W, Corley DA. Proton pump inhibitor and histamine 2 receptor antagonist use and vitamin B12 deficiency. JAMA. 2013 Dec 11;310(22):2435-42. doi: 10.1001/jama.2013.280490. PMID: 24327038.
- van Wijngaarden JP, Dhonukshe-Rutten RAM, Brouwer-Brolsma EM, Enneman AW, Swart KMA, van Dijk SC, In ‘t Veld PH, van Schoor NM, van der Velde N, de Jonge R, Lips P, Uitterlinden AG, de Groot LCPGM. Vitamin B12 Intake and Related Biomarkers: Associations in a Dutch Elderly Population. J Nutr Health Aging. 2017;21(10):1268-1276. doi: 10.1007/s12603-017-0911-6. PMID: 29188889.