How to Support Growth Hormone After Midlife Without Taking Hormones
Growth hormone usually enters the longevity conversation through body composition.
That makes sense.
Growth hormone affects muscle, bone, fat metabolism, and tissue repair. But growth hormone also sits inside a larger overnight process that includes sleep depth, brain health and dementia.
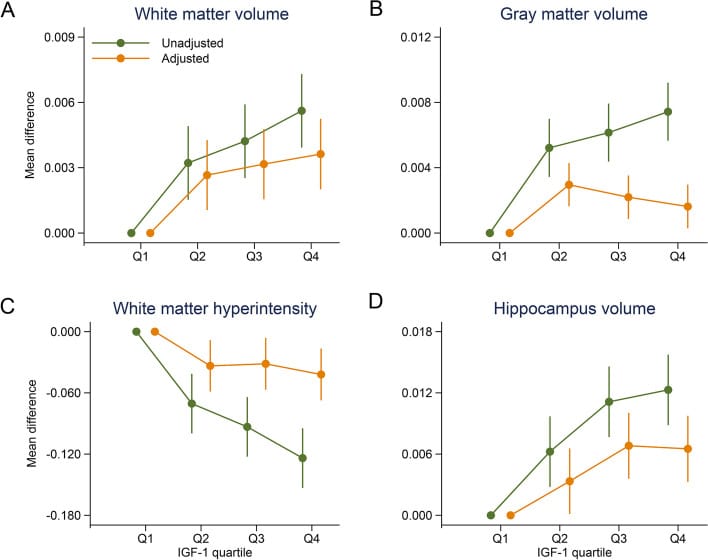
In a UK Biobank analysis of 369,711 adults, IGF-1 – the hormone the liver produces in response to growth hormone – tracked hippocampal volume.
Specifically, lower IGF-1 was associated with
- a smaller hippocampus,
- less white matter volume, and
- more white matter lesions.
The study adjusted for sex, age, ethnicity, educational attainment, smoking status, alcohol intake frequency, vegetable and fruit intake, body mass index, hypertension, diabetes, total cholesterol, and C-reactive protein.
In a separate long-term study, adults in the lowest IGF-1 quartile had a 51% higher risk of developing Alzheimer’s dementia over 7.4 years.
Growth hormone is also tied to the part of sleep that matters for overnight restoration. The largest growth hormone release of the 24-hour cycle usually occurs during the first 60-90 minutes of sleep, when slow-wave sleep is deepest.
During slow-wave sleep, cerebrospinal fluid movement through the glymphatic pathway increases. Recent human data show that sleep-active clearance helps move amyloid beta and tau (proteins involved in Alzheimer’s disease pathology) from brain tissue.
But is more growth hormone better?
In this article we’ll discuss:
- what the longevity data supports in relation to growth hormone
- which factors support healthy growth hormone function
- which factors disrupt growth hormone release or reduce IGF-1 ( insulin-like growth factor-1) response
- 9 things you can do today, to improve growth homone function for optimal brain health & longevity without using peptides or exogenous hormones, regardless of your age
Let’s get started.
Section 2. The IGF1 (insulin-like growth factor-1) paradox – why longevity data does not support maximizing growth hormone
The assumption behind peptide use is that restoring youthful growth hormone output is desirable.
The longevity data is more nuanced.
Insulin-like growth factor-1 is the primary liver-produced hormone that responds to growth hormone. It carries many of the tissue-level effects associated with growth hormone: muscle protein synthesis, bone density maintenance, and cellular repair.
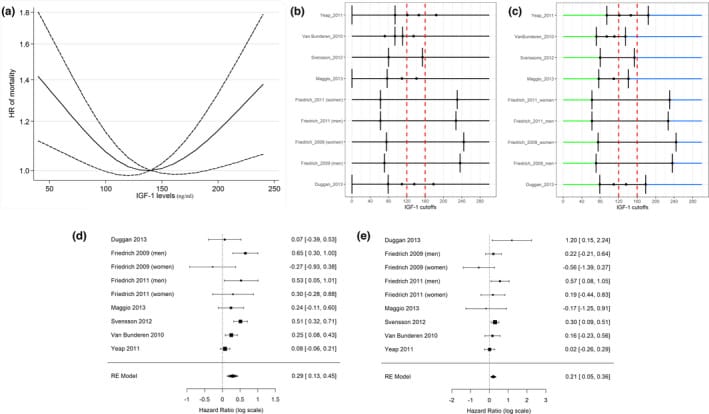
Large pooled analyses suggest that insulin-like growth factor-1 has a U-shaped association with all-cause mortality.
- At the low end, lower insulin-like growth factor-1 is linked with frailty, sarcopenia, cognitive decline, and bone vulnerability.
- At the high end, higher insulin-like growth factor-1 is linked with proliferative concerns, with acromegaly as the human example of chronic growth hormone and insulin-like growth factor-1 excess.
The longevity target is the middle of the range.
There is also a timing question.
Growth hormone is normally released in pulses, with the largest release tied to early slow-wave sleep.
A single lab value does not show whether growth hormone was released at the expected time, whether insulin-like growth factor-1 stayed in a physiologic range, or whether the tissue response was optimal.
So what affects whether the nightly growth hormone release happens at the right time?
Section 3. Factors that support healthy growth hormone function
1. Sleep depth and continuity
The largest growth hormone release of the day usually occurs during the first episode of slow-wave sleep. In one study of healthy men aged 16-83, slow-wave sleep fell from 18.9% of total sleep time in young adulthood to 3.4% by midlife, while growth hormone release fell in parallel.
What matters is more than total sleep duration. The depth and continuity of the first sleep block determine whether the early sleep-linked growth hormone release has a place to occur.
Sleep fragmentation can reduce the early growth hormone release even when total sleep time looks adequate. Chronic sleep restriction can also move growth hormone release into smaller events later in the sleep period.
Across midlife, the growth hormone change tracks the loss of slow-wave sleep closely.
By the way, if sleep after 40 no longer responds to the usual strategies, I’m opening VIP early access to a mobile sleep intelligence product for men 40+, coming to iOS and Android.
It’s designed around the sleep physiology of men 40+ – including changes in testosterone, growth hormone, and GABA – and turns your sleep patterns, sleep architecture, and daytime activity into personalized daily insights.
Join the waitlist – or share this with someone you care about who could benefit.
2. Circadian alignment
In the early sleep period, growth hormone-releasing hormone activity is more supportive of release, while somatostatin restraint tends to be lower.
Both circadian timing and sleep onset contribute to growth hormone release. The circadian component means a late or biologically mismatched sleep period can change the size and timing of the release.
Adequate sleep at a mismatched time can still contain slow-wave sleep, but the hormonal context around that sleep is different.
When sleep onset moves across a wide range, the circadian clock has a harder time aligning sleep pressure, melatonin timing, and the growth hormone-releasing hormone/somatostatin balance.
3. Insulin
Growth hormone release is lower when fasting insulin and abdominal visceral fat are higher.
When insulin drops between meals, the pituitary is less exposed to one of the metabolic conditions associated with lower growth hormone release. When insulin stays elevated through frequent eating, late-night intake, or caloric excess, the first deep sleep block begins under a different endocrine state.
Free fatty acids also matter. Abdominal visceral fat can raise free fatty acid delivery to the liver and pituitary, which can further reduce growth hormone release.
4. Testosterone and estrogen
Testosterone and estrogen help shape growth hormone pulse size and the downstream insulin-like growth factor-1 response.
- In men, testosterone can support growth hormone release directly. But the insulin-like growth factor-1 response also depends partly on aromatization to estradiol. When aromatization is blocked, testosterone can still affect growth hormone release, while the insulin-like growth factor-1 response may be smaller.
- In women, estrogen can support growth hormone release by reducing somatostatin tone at the hypothalamus.
In midlife, testosterone function becomes more easily disrupted in men, and estrogen changes through perimenopause and menopause in women.
Section 4. The factors that disrupt healthy growth hormone function
1. Somatostatin affects growth hormone release and timing
Somatostatin is the hypothalamic hormone that suppresses growth hormone release.
It has appeared throughout this article because sleep timing, insulin exposure, cortisol physiology, inflammation, alcohol, and energy state all interact with the balance between growth hormone-releasing hormone and somatostatin.
The human data are more nuanced than the old shorthand. Growth hormone-releasing hormone stimulates release. Somatostatin reduces the size of that release. Ghrelin and related ligands can amplify the pituitary response.
Aging tends to involve
- lower growth hormone-releasing hormone activity,
- altered ghrelin rhythm, and
- higher somatostatin restraint.
That combination can reduce growth hormone pulse size even when the pituitary can still respond.
2. Abdominal visceral fat and fasting insulin (effect independent of age)
Human 24-hour sampling data put abdominal visceral fat and fasting insulin near the center of growth hormone physiology.
In 102 healthy adults, abdominal visceral fat and fasting insulin explained a large share of the variance in 24-hour growth hormone concentrations, independent of age group, sex, percent body fat, testosterone and estrogen, and aerobic fitness.
The short-term insulin evidence is more direct. In a controlled inpatient overfeeding study, seven lean men ate about 4,000 calories per day for two weeks. Average growth hormone concentration fell by about 80% by day 3, before body weight or fat mass changed. Insulin nearly doubled at the same time.
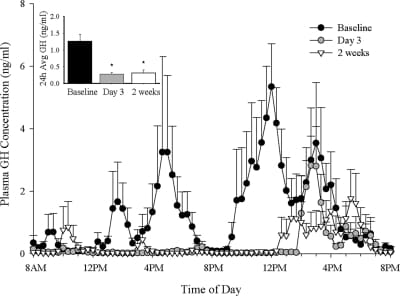
A follow-up overfeeding study adds an important nuance: growth hormone suppression during caloric excess may be partly protective. When growth hormone was maintained during overfeeding, insulin resistance and fasting lipid markers worsened.
3. Cortisol, corticotropin-releasing hormone, and overtraining
Cortisol physiology is timing-dependent. Acute cortisol responses are part of normal adaptation. Chronic glucocorticoid exposure and sustained corticotropin-releasing hormone activity can reduce growth hormone responsiveness.
Human pharmacology studies suggest two related pathways.
- Glucocorticoids can increase beta-adrenergic responsiveness in hypothalamic somatostatin neurons, which increases somatostatin restraint on growth hormone release.
- Corticotropin-releasing hormone can also reduce the growth hormone response to growth hormone-releasing hormone, likely through a hypothalamic pathway rather than cortisol alone.
That distinction matters because “high cortisol” is too broad. The practical concern is late-evening arousal, chronic corticotropin-releasing hormone activity, glucocorticoid exposure, and training stress that reduces recovery capacity.
Overtraining is another underappreciated disruptor.
Human overtraining studies and reviews report blunted growth hormone responses during dynamic testing in athletes with overtraining syndrome. That does not make exercise the issue. It means training volume without enough recovery can reduce hypothalamic-pituitary responsiveness.
For the overnight growth hormone release, the relevant question is whether arousal physiology is still elevated when the first deep sleep block should be occurring.
4. Inflammation and alcohol can reduce insulin-like growth factor-1 response and disrupt overnight growth hormone release
Inflammation creates a different issue from low growth hormone release. Inflammatory cytokine activity can reduce the tissue response to growth hormone, which means insulin-like growth factor-1 can stay lower even when growth hormone is present.
Inflammatory cytokines can reduce growth hormone receptor sensitivity, alter insulin-like growth factor binding proteins, and increase SOCS proteins. SOCS3 can interfere with the JAK/STAT pathway that helps translate growth hormone receptor activation into liver-produced insulin-like growth factor-1.
Growth hormone can be present while insulin-like growth factor-1 activity remains under-supported.
Human muscle biopsy work also links age-related inflammatory activity with higher SOCS3 and altered insulin-like growth factor-1-related muscle stem cell activity.
Alcohol has a separate human evidence base.
In a sleep-lab study, alcohol at 0.8 grams per kilogram suppressed nighttime plasma growth hormone values by 70-75%. Interestingly, a later dose-response study found that ethanol lowered nocturnal growth hormone while plasma growth hormone-releasing hormone did not change.
5. Energy balance changes growth hormone release
Ghrelin, produced primarily by the stomach, works with growth hormone-releasing hormone to increase growth hormone release, especially during fasting.
In human studies, two days of fasting increased 24-hour endogenous growth hormone production about five-fold through more frequent and larger release events. A five-day fast also increased 24-hour integrated growth hormone output.
But the growth hormone rise during fasting is not purely ghrelin-mediated. Falling insulin removes one suppressive input. Changing free fatty acid dynamics modify another. Nutrient sensing pathways at the hypothalamus contribute independently. Growth hormone is responding to the full metabolic context, not to a single hormone.
So what can you do to support optimal growth hormone release and function?
Section 5. 9 things you can do today, to improve growth homone function for optimal brain health & longevity
….without using peptides or exogenous hormones, regardless of your age.
1. Sleep depth and continuity: protect the first deep sleep block
Start with the first 90 minutes after sleep onset. That is when the largest growth hormone release usually occurs.
The goal is an uninterrupted first sleep block; the sleep score is secondary. Late caffeine, late intense work, late heavy meals, heat, bright light, and unresolved arousal can all make early sleep lighter or more fragmented. Alcohol can also change sleep stage dynamics and directly suppress nighttime growth hormone.
Track whether the first sleep block stays intact. Total sleep duration can look adequate while the early slow-wave sleep window remains fragmented.
2. Circadian alignment
Growth hormone release is tied to sleep, but circadian timing shapes the hormonal context around that sleep.
Morning outdoor light helps anchor the day-night rhythm. A repeatable wake time and a repeatable sleep onset time help the circadian clock coordinate sleep pressure, melatonin timing, and the growth hormone-releasing hormone/somatostatin balance.
Evening light matters because delayed melatonin timing can delay sleep onset.
3. Insulin exposure: create regular periods when insulin can fall between meals
Fasting insulin and abdominal visceral fat predict lower 24-hour growth hormone release. Short-term overfeeding can suppress growth hormone within days, before body weight changes.
The practical target is regular time between meals so insulin can return toward baseline. Meals built around protein, fiber-rich plants, and enough energy earlier in the day reduce the need for late intake.
A late pattern of grazing, dessert, and alcohol can keep insulin, liver metabolism, and free fatty acid dynamics active into the first sleep block. Fasting glucose, fasting insulin, waist measurement, and triglycerides are more useful tracking anchors than a single growth hormone value.
4. Abdominal visceral fat
Abdominal visceral fat is not only body composition. It is an endocrine condition that changes insulin exposure, free fatty acid delivery, inflammatory activity, and growth hormone release.
Resistance training, adequate protein, walking after meals, and a consistent sleep window all help lower the metabolic conditions that suppress growth hormone.
Recent fasting analyses also suggest that growth hormone changes can occur independent of weight change.
5. Testosterone and estrogen function
Testosterone and estrogen shape growth hormone release and the downstream insulin-like growth factor-1 response.
In men, testosterone function should be read alongside sleep fragmentation, abdominal visceral fat, fasting insulin, and alcohol intake. In women, estrogen route matters because oral estrogen can raise growth hormone while lowering liver-produced insulin-like growth factor-1, while transdermal estrogen is less likely to reduce insulin-like growth factor-1 through first-pass liver metabolism.
Testosterone and estrogen do not fix growth hormone. They shape how strongly the growth hormone release process responds.
6. Cortisol and corticotropin-releasing hormone physiology: lower late-evening arousal before the first sleep block
Corticotropin-releasing hormone can blunt the growth hormone response to growth hormone-releasing hormone in humans. Glucocorticoids can also increase somatostatin restraint.
That makes late-evening arousal a practical target. Demanding work, conflict, intense exercise, and emotionally activating inputs are better placed earlier when possible. A repeatable wind-down works when it lowers arousal, not because it checks off a long sleep hygiene list.
If early waking is the dominant pattern, the relevant question is which physiology is active at that time: cortisol and corticotropin-releasing hormone, blood sugar, temperature, inflammation, or another profile.
7. Inflammation and alcohol: separate chronic inflammatory load from the acute alcohol effect
Inflammation can reduce growth hormone receptor sensitivity and insulin-like growth factor-1 activity through cytokines, SOCS proteins, and changes in insulin-like growth factor binding proteins.
The useful starting points are measurable and concrete: sleep-apnea testing when relevant, oral health, resistance training, fiber intake, omega-3-rich foods, and reducing abdominal visceral fat. Broad anti-inflammatory language is less useful than naming the inputs that can be verified.
Alcohol sits in a separate category because human sleep-lab data directly show suppression of nighttime growth hormone. If growth hormone is the concern, alcohol timing and dose belong in the same review as sleep depth, insulin exposure, and early waking.
8. Somatostatin
Somatostatin is the hypothalamic hormone that suppresses growth hormone release.
The practical work is not to micromanage somatostatin directly. The practical work is to reduce the conditions that tend to increase somatostatin restraint or reduce growth hormone-releasing hormone activity: late arousal, mismatched sleep timing, elevated insulin exposure, inflammation, alcohol, and chronic caloric excess.
9. Energy balance and meal timing: avoid chronic excess
Fasting studies show that growth hormone release responds strongly to energy state. Two days of fasting can raise growth hormone production several-fold. Overfeeding can suppress growth hormone within days.
That does not make extended fasting the universal action step. The practical emphasis is caloric balance, fewer late calories, and enough protein and resistance training to protect lean tissue.
If fasting is used, the useful frame is evidence that growth hormone release responds to insulin exposure, ghrelin rhythm, and energy state. The goal is not a fasting-induced spike. The goal is a nightly release that occurs in a supportive metabolic context.
Section 6. Longevity data does not support maximizing growth hormone
The longevity data does not support maximizing growth hormone. It supports enough growth hormone activity to maintain tissue function, with insulin-like growth factor-1 staying in a physiologic range.
Across midlife, the first question is usually not whether growth hormone can be forced higher. The better question is whether the conditions for timed release are still present.
Each condition gives you a more useful question than “How do I raise growth hormone?”
Supporting these conditions may or may not produce a specific growth hormone number on a lab test.
But it addresses the sleep, metabolic, inflammatory, and neuroendocrine conditions that determine whether growth hormone is released at the right time and translated into healthy hormone function that supports brain health, prevents frailty and promotes longvevity.
- Kat
P.S. If you’re struggling to get consistent, high-quality sleep – or still waking with low energy – I’m opening private early access to a mobile sleep intelligence product for men 40+, built for iOS and Android.
Using your sleep patterns, sleep architecture, and daytime activity, it delivers daily guidance for men 40+ focused on brain health, and longevity.
Join the waitlist – or share this with someone who could benefit.
Personalized sleep intelligence for men 40+, delivered daily on iOS & Android:

References
- Cao, Z., Min, J., Tan, Q., Si, K., Yang, H., & Xu, C. (2023). Circulating insulin-like growth factor-1 and brain health: Evidence from 369,711 participants in the UK Biobank. Alzheimer’s Research & Therapy, 15(1), 140.
- Dagum, P., Elbert, D. L., Giovangrandi, L., et al. (2026). The glymphatic system clears amyloid beta and tau from brain to plasma in humans. Nature Communications, 17(1), 715.
- Rahmani, J., Montesanto, A., Giovannucci, E., et al. (2022). Association between IGF-1 levels ranges and all-cause mortality: A meta-analysis. Aging Cell, 21(2), e13540.
- Freda, P. U., Bruce, J. N., Jin, Z., Kostadinov, J., Khandji, A. G., Cremers, S., & Post, K. D. (2025). Prospective, longitudinal study of cancer predictors and rates in a New York City cohort of 598 patients with acromegaly. The Journal of Clinical Endocrinology & Metabolism, 110(5), 1247-1257.
- Van Cauter, E., Leproult, R., & Plat, L. (2000). Age-related changes in slow wave sleep and REM sleep and relationship with growth hormone and cortisol levels in healthy men. JAMA, 284(7), 861-868.
- Morris, C. J., Aeschbach, D., & Scheer, F. A. (2012). Circadian system, sleep and endocrinology. Molecular and Cellular Endocrinology, 349(1), 91-104.
- Veldhuis, J. D., Evans, W. S., Iranmanesh, A., Weltman, A. L., & Bowers, C. Y. (2004). Short-term testosterone supplementation relieves growth hormone autonegative feedback in men. The Journal of Clinical Endocrinology & Metabolism, 89(3), 1285-1290.
- Cook, D. M., Ludlam, W. H., & Cook, M. B. (1999). Route of estrogen administration helps to determine growth hormone (GH) replacement dose in GH-deficient adults. The Journal of Clinical Endocrinology & Metabolism, 84(11), 3956-3960.
- Clasey, J. L., Weltman, A., Patrie, J., Weltman, J. Y., Pezzoli, S., Bouchard, C., Thorner, M. O., & Hartman, M. L. (2001). Abdominal visceral fat and fasting insulin are important predictors of 24-hour GH release independent of age, gender, and other physiological factors. The Journal of Clinical Endocrinology & Metabolism, 86(8), 3845-3852.
- Cornford, A. S., Barkan, A. L., & Horowitz, J. F. (2011). Rapid suppression of growth hormone concentration by overeating: Potential mediation by hyperinsulinemia. The Journal of Clinical Endocrinology & Metabolism, 96(3), 824-830.
- Cornford, A. S., Barkan, A. L., Hinko, A., & Horowitz, J. F. (2012). Suppression in growth hormone during overeating ameliorates the increase in insulin resistance and cardiovascular disease risk. American Journal of Physiology-Endocrinology and Metabolism, 303(10), E1264-E1272.
- Horne, B. D., Anderson, J. L., May, H. T., Bair, T. L., Le, V. T., Iverson, L., Knowlton, K. U., & Muhlestein, J. B. (2024). Insulin resistance reduction, intermittent fasting, and human growth hormone: Secondary analysis of a randomized trial. npj Metabolic Health and Disease, 2(1), 26.
- Horne, B. D., Anderson, J. L., May, H. T., Bair, T. L., Le, V. T., Iverson, L., Knowlton, K. U., & Muhlestein, J. B. (2024). Weight loss-independent changes in human growth hormone during water-only fasting: A secondary evaluation of a randomized controlled trial. Frontiers in Endocrinology, 15, 1401780.
- Lima, L., Arce, V., Diaz, M. J., Tresguerres, J. A., & Devesa, J. (1993). Glucocorticoids may inhibit growth hormone release by enhancing beta-adrenergic responsiveness in hypothalamic somatostatin neurons. The Journal of Clinical Endocrinology & Metabolism, 76(2), 439-444.
- Raza, J., Massoud, A. F., Hindmarsh, P. C., Robinson, I. C., & Brook, C. G. (1998). Direct effects of corticotrophin-releasing hormone on stimulated growth hormone secretion. Clinical Endocrinology, 48(2), 217-222.
- Cadegiani, F. A., & Kater, C. E. (2017). Hypothalamic-pituitary-adrenal (HPA) axis functioning in overtraining syndrome: Findings from Endocrine and Metabolic Responses on Overtraining Syndrome (EROS)-EROS-HPA Axis. Sports Medicine – Open, 3(1), 45.
- Cadegiani, F. A., & Kater, C. E. (2017). Hormonal aspects of overtraining syndrome: A systematic review. BMC Sports Science, Medicine and Rehabilitation, 9, 14.
- Bioletto, F., Varaldo, E., Gasco, V., Maccario, M., Arvat, E., Ghigo, E., & Grottoli, S. (2025). Central and peripheral regulation of the GH/IGF-1 axis: GHRH and beyond. Reviews in Endocrine and Metabolic Disorders, 26(3), 321-342.
- Montero-Hidalgo, A. J., Del Rio-Moreno, M., Perez-Gomez, J. M., Luque, R. M., & Kineman, R. D. (2025). Update on regulation of GHRH and its actions on GH secretion in health and disease. Reviews in Endocrine and Metabolic Disorders, 26(3), 305-320.
- Witkowska-Sedek, E., & Pyrzak, B. (2020). Chronic inflammation and the growth hormone/insulin-like growth factor-1 axis. Central-European Journal of Immunology, 45(4), 469-475.
- McKay, B. R., Ogborn, D. I., Baker, J. M., Toth, K. G., Tarnopolsky, M. A., & Parise, G. (2013). Elevated SOCS3 and altered IL-6 signaling is associated with age-related human muscle stem cell dysfunction. American Journal of Physiology-Cell Physiology, 304(8), C717-C728.
- Cammisa, I., Rigante, D., & Cipolla, C. (2025). A theoretical link between the GH/IGF-1 axis and cytokine family in children: Current knowledge and future perspectives. Children, 12(4).
- Klinc, A., Janez, A., & Jensterle, M. (2025). Long-term IGF-1 maintenance in the upper-normal range has beneficial effect on low-grade inflammation marker in adults with growth hormone deficiency. International Journal of Molecular Sciences, 26(5).
- Prinz, P. N., Roehrs, T. A., Vitaliano, P. P., Linnoila, M., & Weitzman, E. D. (1980). Effect of alcohol on sleep and nighttime plasma growth hormone and cortisol concentrations. The Journal of Clinical Endocrinology & Metabolism, 51(4), 759-764.
- Ekman, A. C., Vakkuri, O., Ekman, M., Leppaluoto, J., Ruokonen, A., & Knip, M. (1996). Ethanol decreases nocturnal plasma levels of thyrotropin and growth hormone but not those of thyroid hormones or prolactin in man. The Journal of Clinical Endocrinology & Metabolism, 81(7), 2627-2632.
- McCullar, K. S., Barker, D. H., McGeary, J. E., Saletin, J. M., Gredvig-Ardito, C., Swift, R. M., & Carskadon, M. A. (2024). Altered sleep architecture following consecutive nights of presleep alcohol. Sleep, 47(4).
- Gardiner, C., Weakley, J., Burke, L. M., et al. (2025). The effect of alcohol on subsequent sleep in healthy adults: A systematic review and meta-analysis. Sleep Medicine Reviews, 80, 102030.
- Alba-Roth, J., Muller, O. A., Schopohl, J., & von Werder, K. (1988). Arginine stimulates growth hormone secretion by suppressing endogenous somatostatin secretion. The Journal of Clinical Endocrinology & Metabolism, 67(6), 1186-1189.
- Ghigo, E., Goffi, S., Nicolosi, M., Arvat, E., Valente, F., Mazza, E., Ghigo, M. C., & Camanni, F. (1990). Growth hormone responsiveness to combined administration of arginine and growth hormone-releasing hormone does not vary with age in man. The Journal of Clinical Endocrinology & Metabolism, 71(6), 1481-1485.
- Dimaraki, E. V., Jaffe, C. A., Demott-Friberg, R., Russell-Aulet, M., Bowers, C. Y., Marbach, P., & Barkan, A. L. (2001). Generation of growth hormone pulsatility in women: Evidence against somatostatin withdrawal as pulse initiator. American Journal of Physiology-Endocrinology and Metabolism, 280(3), E489-E495.
- Oikonomakos, I., Siow, R., Bornstein, S. R., & Steenblock, C. (2025). The role of growth hormone-releasing hormone and the hypothalamic-pituitary-somatotropic axis in aging: Potential therapeutic applications and risks. Hormone and Metabolic Research, 57(11), 662-667.
- Fernandez-Garza, L. E., Guillen-Silva, F., Sotelo-Ibarra, M. A., Dominguez-Mendoza, A. E., Barrera-Barrera, S. A., & Barrera-Saldana, H. A. (2025). Growth hormone and aging: A clinical review. Frontiers in Aging, 6, 1549453.
- Ho, K. Y., Veldhuis, J. D., Johnson, M. L., Furlanetto, R., Evans, W. S., Alberti, K. G., & Thorner, M. O. (1988). Fasting enhances growth hormone secretion and amplifies the complex rhythms of growth hormone secretion in man. The Journal of Clinical Investigation, 81(4), 968-975.
- Hartman, M. L., Veldhuis, J. D., Johnson, M. L., Lee, M. M., Alberti, K. G., Samojlik, E., & Thorner, M. O. (1992). Augmented growth hormone secretory burst frequency and amplitude mediate enhanced GH secretion during a two-day fast in normal men. The Journal of Clinical Endocrinology & Metabolism, 74(4), 757-765.
- Muller, A. F., Lamberts, S. W., Janssen, J. A., et al. (2002). Ghrelin drives GH secretion during fasting in man. European Journal of Endocrinology, 146(2), 203-207.
- Khaira, F., & Sulastri, D. (2026). Meal timing and ghrelin: A chrononutritional perspective on weight regulation potential. Chronobiology International, 43(5), 606-613.