Waking to urinate multiple times at night is often framed as a bladder or hydration problem—but nocturia is primarily an ADH and sleep problem. ADH (antidiuretic hormone, also called vasopressin) should rise during sleep to concentrate urine and slow kidney filtration; when sex hormone changes or sleep fragmentation disrupt this rise, the kidneys continue producing urine at daytime rates all night.
- Sex hormone changes in midlife directly affect ADH function. Testosterone and estrogen receptors exist in both brain and kidney tissue. When testosterone function is impaired, overnight urine production increases and sleep becomes more arousable. When estrogen function falters, the body becomes less efficient at concentrating urine overnight.
- Progesterone supports deeper sleep and reduces nighttime arousal. When progesterone support weakens, sleep becomes shallower—and small bladder sensations that a deeply asleep brain wouldn’t register become enough to wake you.
- The “phantom urge” is a sleep depth problem. Waking with a strong urge to urinate but passing little often happens because light sleep lets the brain register sensations that deeper sleep would not. The absence of deep sleep allows you to feel the urge—not the other way around.
- Fluid restriction does not address the underlying cause. When ADH function is intact, the kidneys conserve fluid overnight regardless of intake. When ADH is disrupted, restricting fluids may reduce output but leaves the endocrine and sleep architecture issue unaddressed.
Waking to urinate multiple times per night—nocturia—often gets mislabeled as a bladder problem when it’s a sleep and endocrine problem. Nocturnal urine output is governed by ADH (antidiuretic hormone, also called vasopressin), which rises during sleep to reduce kidney filtration and preserve fluids overnight. When ADH rhythms are disrupted—due to circadian misalignment, fragmented sleep, or changes in sex hormone status (testosterone decline, estrogen/progesterone adjustments)—the kidneys continue filtering urine at daytime rates throughout the night, forcing you to wake and empty your bladder repeatedly. The issue isn’t bladder capacity or overhydration; it’s that your endocrine system isn’t cueing the kidneys to conserve fluid during sleep. Medications that increase ADH (desmopressin) can help temporarily, but addressing the underlying sleep fragmentation and hormone rhythm patterns tends to produce more sustainable improvement.
Key Takeaways:
- Nocturia stems from disrupted ADH (vasopressin) rhythms, not primarily from bladder size or fluid intake
- Sex hormone changes (declining testosterone, altering estrogen/progesterone) affect ADH secretion and overnight urine production
- Sleep fragmentation and circadian misalignment blunt ADH’s nighttime rise—causing continued daytime-level urine production through the night
- Fixing nocturia requires addressing underlying sleep architecture and endocrine rhythm issues—not just bladder management
Recently, a CEO client said: “I’ve optimized everything—nutrition, training, biomarkers. But every night I wake up to pee and can’t get back to sleep. I’ve tried everything. This one thing I can’t solve is driving me crazy.”
Melatonin, valerian, prescription aids, zero liquids after 7. Nothing worked.
Here’s what I told him: It’s not just your bladder. It’s your brain.
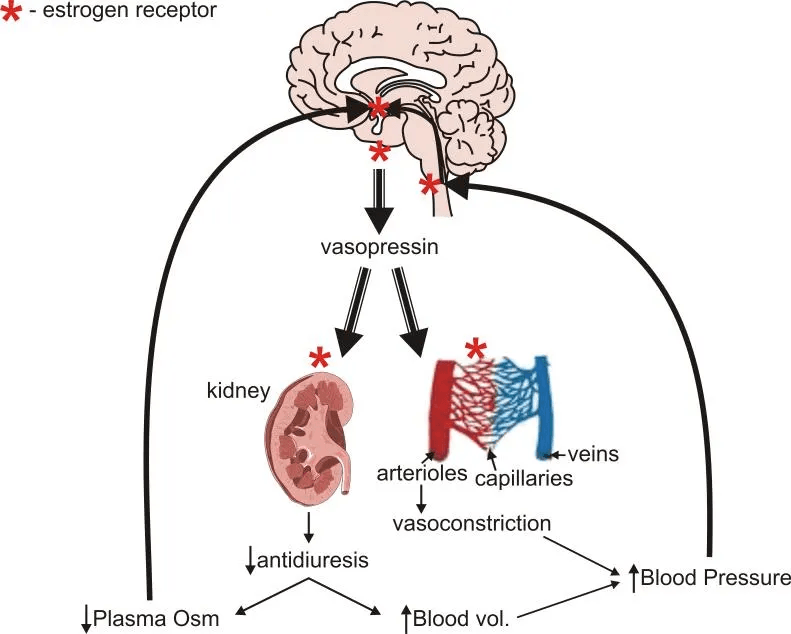
To understand what that means, we have to first look at our anti-diuretic hormone (ADH), also called vasopressin.
Why Is Nighttime Urination a Hormonal and Sleep Problem, Not a Bladder Problem?
The Anti-Diuretic Hormone (ADH) is your body’s water-retention signal. It follows a circadian pattern: levels should rise at night so the kidneys concentrate urine and slow bladder filling.
In midlife, this nighttime fluid processing pattern often becomes unstable — and when it does, two things happen:
- Light Sleep: Your sleep depth is more fragile, so your “wake threshold” is lower.
- Altered ADH: Midlife reproductive-hormone shifts change how ADH behave meaning your kidneys process water differently at night.
Put together, that can sometimes appears as what I call the phantom urge:
You feel a need to pee every few hours, but when you go, you barely pass any urine—and then can’t get back to sleep. The problem isn’t an overactive bladder.
Because the sleep is light and unstable, the brain registers tiny signals from the bladder that a deeply asleep brain would ignore.
The urge didn’t pull you out of deep sleep. The lack of deep sleep allowed you to feel the urge + to prevent you from falling back asleep.
[Note: Here I’m focusing on functional and non-medical contributors. Sudden or major changes in urination frequency or volume should always be evaluated by a physician.]
How Do Sex Hormones, Sleep Fragmentation, and Antidiuretic Hormone Timing Drive Nighttime Urination?
I used to wake up every night to pee too—and assumed that was the main issue. Like most, I started with no fluids after 5/6/7 PM. I even tried no fluids after 3pm. None worked.
It was so disruptive I went to a urologist. Then a second, for another opinion. I did the full workup—bladder ultrasound and the flow-measurement chair—twice.
The result each time: “Everything is normal.” The conclusion: “Too much water at night—stop drinking after 7 p.m.” After 12 months of tests and 2 specialists, I was back where I started.
I found my solution when I stopped focusing on preventing the “need to pee” and started addressing why I had the phantom urge + why I couldn’t return to sleep.
Today, I still wake up and need to pee—every night—mostly because I genuinely feel better having (quite a bit of) liquids with and after dinner—but I fall back asleep without effort also, every night. My bladder didn’t get bigger or become less active and I have fluids in the evening, often past 8 p.m. (plus, I really enjoy tea after dinner).
The difference is that my sleep has become high quality, deeper, and more robust.
So in the rest of this article, we’ll look at three pieces:
- why nighttime urination—nocturia—isn’t always about fluid overload,
- why this pattern shows up so often in midlife, and
- how I have helped others turn nighttime bathroom trips into non-events—and how you can achieve the same.
Why Do Declining Testosterone and Disrupted Circadian Rhythm Cause Nighttime Urination?
How Do Testosterone and Estrogen Changes Affect Antidiuretic Hormone and Bladder Behavior?
The pattern many notice in midlife—lighter sleep, louder bladder signals, faster urine production—doesn’t begin with hydration. It begins with reproductive hormone changes:
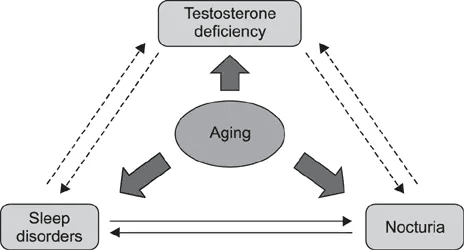
How Does Testosterone Affect Sleep and Nighttime Urination?
Androgen (testosterone) receptors —where cells receive testosterone for it to act— exist in both the brain’s control center and kidney tissue. When testosterone function is impaired—not just low, but poorly utilized—two things happen simultaneously: more urine is produced at night, and the brain remains in a lighter, more arousable state.
How Does Estrogen Affect Sleep and Nighttime Urination?
Estrogen receptors —where cells receive estrogen for it to act—regulate ADH signaling and kidney behavior. When estrogen function is adequate—not just present, but properly received at the receptor—the body runs “night mode” on water: less urine, more concentrated, slower bladder filling. When function is impaired, sleep gets lighter and the system that concentrates urine at night becomes less efficient.
How Does Progesterone Affect Sleep Depth and Nighttime Urination?
Progesterone supports deeper sleep and lowers nighttime arousal. When progesterone support weakens, sleep becomes shallower—and small bladder signals that a deeply-asleep brain would ignore now feel loud enough to wake you.
What Is the Combined Effect of Hormonal Changes on Nighttime Urination in Midlife?
Even with normal water intake, the bladder fills at the wrong time and the sleep is too light for the brain to ignore it. This is why “stop drinking water before bed” doesn’t actually stop the 3am wakeups, nor does it improve your sleep.
That logic is reversed.
Instead, when you improve your sleep, you can drink water before bed and take bathroom trips without it impacting and how satisfied you are with your sleep.
One SleepOS member summed it up like this when I asked how well he was sleeping:
If you’re ready to stop treating the bladder and get a jump start—before the holidays—on the kind of hormone-stabilized sleep that protects your brain long term & lets you head into the season feeling good, this is the Trio Hormones Sleep Solution this member used.
👉 Start SleepOS Hormones Trio Hormone Sleep Solution Here
Can This Hormonal Pattern Apply Even If Your Situation Feels Different?
You might be thinking, “Okay, that sounds nice, but I’ve been waking up at 3 a.m. for over a decade. My body just doesn’t do that.”
Fair. Here’s what transformation looks like for someone else—same person, 3 months apart after working through Sleep OS Hormones:
Before Sleep OS (July): Here, this individual is getting 7 hours, but they perceive their sleep is not much better after trying the usual tips. The bladder signal wakes them up, and because the sleep depth is shallow, they stay awake.
After Sleep OS (October): Fast forward three months, his update looked very different. We didn’t shrink his liquid intake window or give him a bigger bladder.
But look at the result: “Back to sleep in seconds.”
This is the shift.
When you support the hormonal pathways—foundationally improving the Sleep Depth & Quality—the bathroom trip stops being a prolonged wake-up and becomes a non-event
You wake, you go, you return to bed, and your brain drops back into sleep almost immediately.
Here is another Sleep OS member (professional, mother, HRT user) describing that same state after Sleep OS Hormones:
That is the goal of SleepOS Hormones.
Warmly, Kat
P.S. If your sleep has changed since midlife—waking at 3 a.m., whether you need to pee or not, and then not falling back asleep—sleep hygiene is not enough. The midlife hormonal changes affects how your body regulates sleep and fluid processing, but that function can be supported at any age.
My digital program, Sleep OS: Hormones was designed for this. It offers a self-paced, step-by-step process to strengthen hormonal pathways that stabilize sleep—without supplements, prescriptions, hormone therapy or lab testing.
👉 You can learn more about my most popular program, the Trio Hormone Sleep Improvement Solution here. (code: TRIO15 until 12/1)
👉 Or, explore the foundational Sleep & Stress Single Hormone Sleep Improvement Solution here

FAQ
Q: Is waking to pee at night ever just a urinary tract issue, not a hormone or sleep issue? A: UTIs and bladder dysfunction can cause nocturia, but chronic nocturia in otherwise healthy individuals—especially waking 2+ times per night—is usually driven by ADH rhythm disruption and sleep fragmentation, not bladder capacity issues. If urine cultures are normal and bladder imaging is unremarkable, the problem is endocrine and sleep-related. A urologist’s clearance is useful to rule out structural issues.
Q: Does the amount of fluid I drink before bed matter if my ADH is working? A: Less than you’d think—when ADH function is intact, your kidneys conserve fluid overnight regardless of daytime intake. When ADH rhythm is disrupted (due to sleep fragmentation or circadian misalignment), even moderate hydration can result in nocturia. So if your ADH is disrupted, restricting fluids is a band-aid; fixing the endocrine rhythm is the more durable solution.
Q: Can declining testosterone directly cause nocturia? A: Often, yes—testosterone supports ADH secretion timing and also improves sleep architecture quality, which secondarily supports ADH’s nighttime rise. Low testosterone may reduce ADH sensitivity or disrupt its circadian peak. This is one reason nocturia is more common in men with lower or declining testosterone, and why testosterone optimization sometimes improves nighttime urine patterns.
Q: Will fixing my sleep alone improve nocturia, or do I also need hormone treatment? A: In many cases, improving sleep continuity and circadian alignment alone can significantly reduce nocturia—because better sleep directly supports ADH secretion patterns. However, if testosterone is substantially low, hormone optimization may be necessary alongside sleep improvements. The two work together; neither fully compensates for the other being compromised.
Q: How quickly does nocturia improve if I fix my sleep fragmentation? A: Many individuals see improvement within 1-2 weeks of better sleep architecture—fewer nighttime waking episodes often means less total urine production overnight. More complete improvement (fewer overall bathroom trips) may take 4-6 weeks as ADH rhythm and circadian alignment stabilize. The speed depends on how disrupted your baseline patterns were and whether other factors (low testosterone, fluid intake timing) are also being addressed.
References
- Pauwaert K, Goessaert AS, Robinson D, Cardozo L, Bower W, Calders P, Mariman A, Abrams P, Tubaro A, Dmochowski R, Weiss JP, Hervé F, Depypere H, Everaert K. Nocturia in Menopausal Women: The Link Between Two Common Problems of the Middle Age. Int Urogynecol J. 2024 May;35(5):935-946. doi: 10.1007/s00192-024-05743-1. Epub 2024 Mar 4. PMID: 38436669.
- Shigehara K, Izumi K, Mizokami A, Namiki M. Testosterone Deficiency and Nocturia: A Review. World J Mens Health. 2017 Apr;35(1):14-21. https://doi.org/10.5534/wjmh.2017.35.1.14
- Song QX, Suadicani SO, Negoro H, Jiang HH, Jabr R, Fry C, Xue W, Damaser MS. Disruption of circadian rhythm as a potential pathogenesis of nocturia. Nat Rev Urol. 2025 May;22(5):276-293. doi: 10.1038/s41585-024-00961-0. Epub 2024 Nov 14. PMID: 39543359; PMCID: PMC12285584.
- Pauwaert K, Goessaert AS, Robinson D, Cardozo L, Bower W, Calders P, Mariman A, Abrams P, Tubaro A, Dmochowski R, Weiss JP, Hervé F, Depypere H, Everaert K. Nocturia in Menopausal Women: The Link Between Two Common Problems of the Middle Age. Int Urogynecol J. 2024 May;35(5):935-946. doi: 10.1007/s00192-024-05743-1. Epub 2024 Mar 4. PMID: 38436669.
- Sladek CD, Somponpun SJ. Estrogen receptors: their roles in regulation of vasopressin release for maintenance of fluid and electrolyte homeostasis. Front Neuroendocrinol. 2008 Jan;29(1):114-27. doi: 10.1016/j.yfrne.2007.08.005. Epub 2007 Oct 12. PMID: 18022678; PMCID: PMC2274006.
