Melatonin’s role extends well beyond sleep onset. It accumulates in mitochondria, scavenges reactive oxygen and nitrogen species, modulates immune function, and interacts with hormonal pathways—including those governing glucose metabolism, memory, and reproductive hormones. Whether that range of activity is protective or disruptive depends on dose, timing, and the individual’s underlying health status.
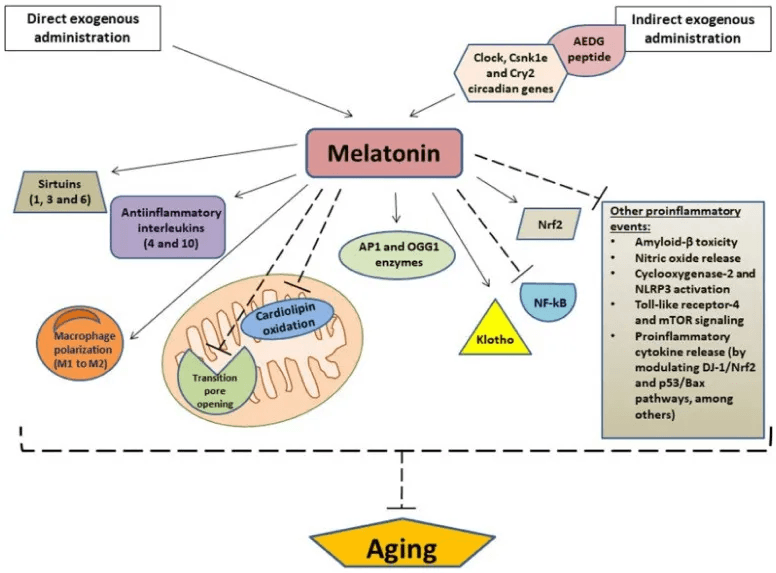
- At physiological levels, melatonin upregulates antioxidant enzymes—including glutathione peroxidase, superoxide dismutase, and catalase—and activates sirtuins SIRT1 and SIRT3, linked to mitochondrial resilience and reduced inflammaging across cardiovascular and neural tissue.
- In a controlled study of 21 healthy adults, a 5 mg dose before a glucose tolerance test raised glucose AUC by approximately 186% in the morning and 54% in the evening, with morning doses reducing insulin release and evening doses reducing insulin sensitivity.
- A double-blinded, placebo-controlled crossover trial of 17 men with type 2 diabetes found that 10 mg nightly for three months reduced insulin sensitivity by approximately 12%, assessed by hyperinsulinemic-euglycemic clamp—a gold-standard measurement method.
- In mouse hippocampal models, melatonin at 100 nM reduced long-term potentiation by approximately 50% through MT₂ receptor-mediated inhibition of the cAMP/PKA pathway, raising questions about high-dose effects on memory consolidation.
- Some data suggest possible interference with reproductive hormones and immune homeostasis in vulnerable populations; its antioxidant action can also flip to pro-oxidant activity under specific metabolic conditions.
What Are Melatonin’s Effects Beyond Sleep on Mitochondria, Immune Function, and Hormones?
We’ve been taught to think of melatonin as the body’s sleep hormone—a messenger of darkness, signaling when it’s time to rest.
But that framing barely scratches the surface.
Melatonin doesn’t just tell the brain it’s night.
It moves through nearly every major system: reducing mitochondrial oxidative stress, modulating immune function, suppressing inflammatory signaling, and interacting with hormonal pathways few associate with sleep. In certain contexts, its actions resemble those of a regulatory hormone more than a simple chronobiotic.
This complexity is both its promise and its problem.
In many cases, melatonin can support redox balance, mitochondrial resilience, and age-related defense. But in non-physiological or pharmacological doses—or when used chronically—it can disrupt glucose metabolism, suppress synaptic plasticity, and alter reproductive signaling.
➔ Even its antioxidant behavior, often considered universally protective, can flip to pro-oxidant in vitro under specific metabolic conditions.
➔ Mistimed or high-dose use has been shown to impair insulin sensitivity and blunt hippocampal signaling essential for memory.
➔ Some data suggest possible interference with reproductive hormones and immune homeostasis—especially in vulnerable populations.
What follows is a layered examination of melatonin’s dual nature: why it has drawn interest as a longevity molecule, where its benefits seem compelling, and where its risks suggest caution.
Table of Contents
What Is Melatonin’s Role as a Longevity Molecule Beyond Sleep?
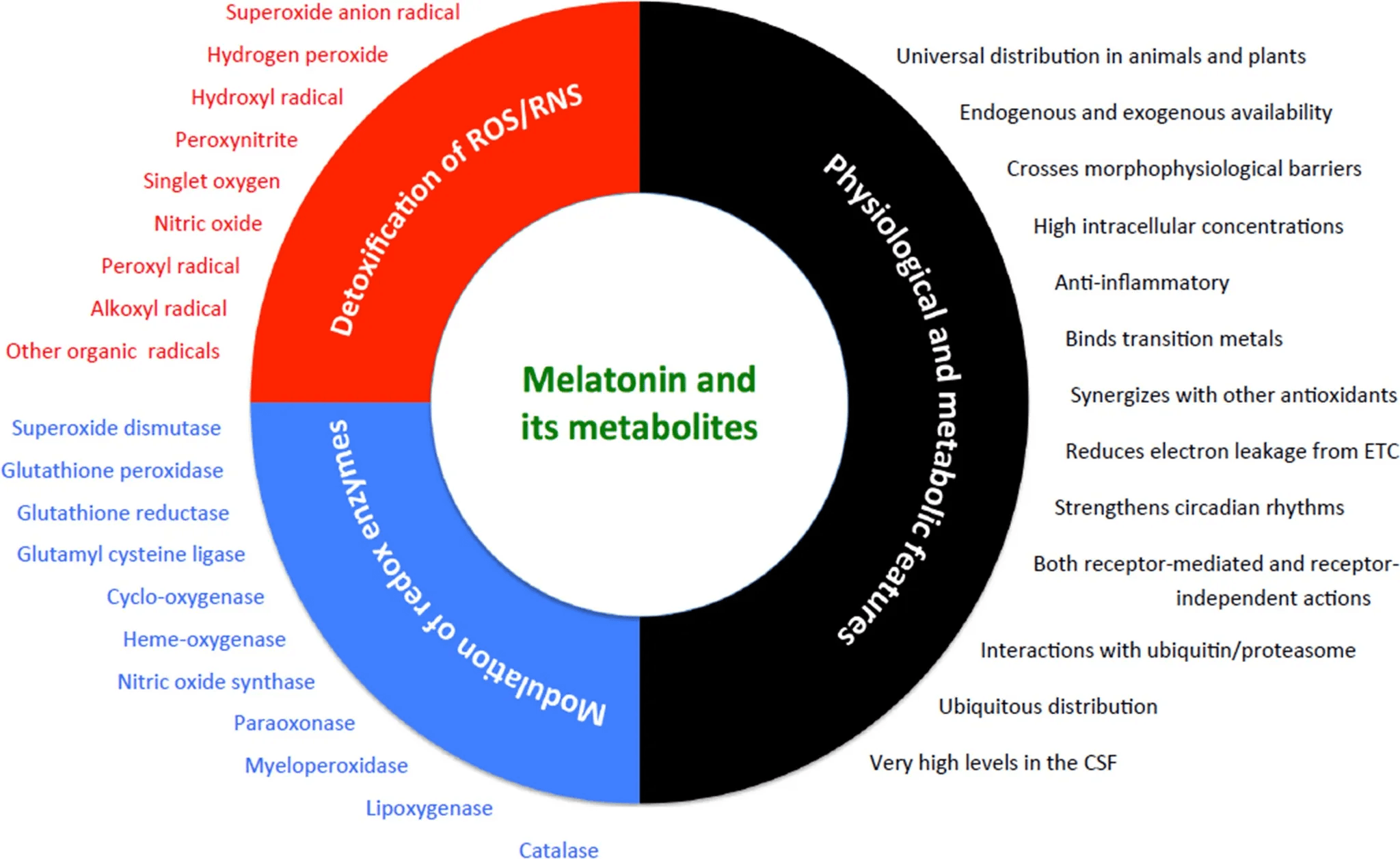
Melatonin exhibits a robust capacity to reduce oxidative stress across a wide range of cellular environments. It acts through both direct and indirect mechanisms:
➔ Directly neutralizing reactive oxygen species (ROS) and reactive nitrogen species (RNS), including some of the most damaging radicals in biological systems.
➔ Indirectly enhancing the cell’s antioxidant defense by increasing the activity of endogenous antioxidant enzymes and reducing the activity of enzymes that generate free radicals.
Additionally, melatonin is capable of chelating transition metals that participate in the Fenton and Haber-Weiss reactions—critical drivers of hydroxyl radical formation. This allows melatonin to suppress oxidative cascades before they can propagate significant cellular damage.
Its intracellular distribution is also relevant. Melatonin accumulates in mitochondria—one of the primary sources of ROS production in the cell. This localization contributes to its ability to prevent mitochondrial dysfunction and reduce apoptosis under oxidative stress conditions. These characteristics support the classification of melatonin as a mitochondria-targeted antioxidant.
How Does Melatonin Protect Mitochondria Through Antioxidant Actions?
Beyond its role in circadian regulation, melatonin functions as a broad-spectrum antioxidant. It directly scavenges harmful radicals such as hydroxyl, peroxyl, and nitric oxide species. It also amplifies the body’s natural antioxidant systems by upregulating enzymes like glutathione peroxidase, superoxide dismutase, and catalase, while simultaneously suppressing pro-oxidant enzymes like nitric oxide synthase and lipoxygenase .
These functions have been documented in both in vitro and in vivo models. For example, melatonin has demonstrated protective effects against oxidative injury in the brain during ischemic stroke, in the myocardium during infarction, and in models of drug-induced toxicity. Across these systems, its antioxidant actions are consistently linked to reductions in inflammation and preservation of mitochondrial membrane integrity .
Melatonin’s antioxidant capacity is not limited to membrane compartments. It crosses cell membranes and enters subcellular structures, including mitochondria, where it directly counters oxidative stress at its point of origin. This has led to comparisons with vitamin E—another lipid-soluble antioxidant. In head-to-head assessments, melatonin has been shown to provide more comprehensive protection across aqueous and lipid environments.
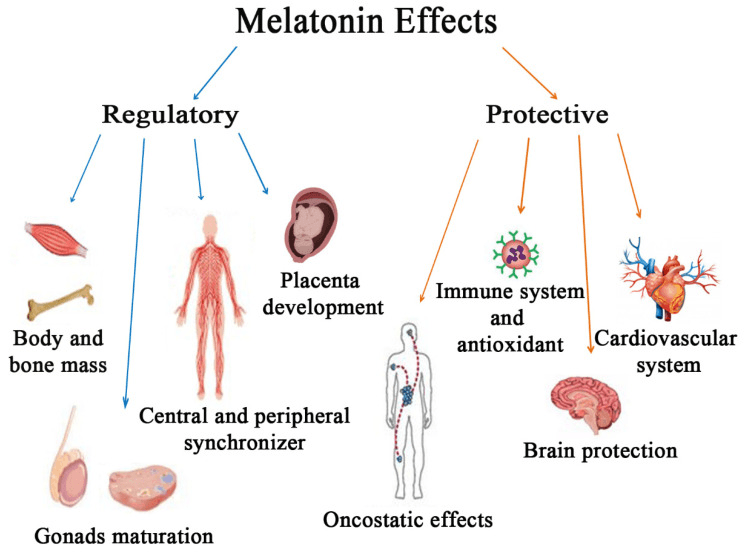
Because of these features, melatonin has been explored as an adjunct in a range of oxidative stress–related conditions, including neurodegenerative disease, cancer therapy, and inflammatory disorders. Its ability to act at the mitochondrial level while regulating broader redox dynamics is a central reason for this interest.
➔ Next, we examine how these antioxidant properties influence aging in two systems particularly vulnerable to oxidative stress: the brain and the cardiovascular system.
How Does Melatonin Interact With Cardiovascular and Neurodegenerative Aging?
Mechanistically, melatonin functions as a multi-targeted signaling molecule that modulates several hallmarks of aging—most notably mitochondrial dysfunction, chronic inflammation, and loss of cellular resilience. These roles become particularly relevant in the context of cardiovascular and neurodegenerative aging.
By the way, If you’ve been following my work on hormones and sleep, you’ll know how much depth there is beneath the surface.
If you’re ready to go deeper and take a systems-based approach to improving your sleep, Sleep OS Hormones is now available as a 60-day self-guided program with dedicated systems for estrogen, progesterone, and testosterone, or bundled together for a more complete approach.
or
➤ Cardiovascular Aging: Endothelial Function, Mitochondrial Dynamics, Nrf2, and Telomere Support
Aging cardiomyocytes and vascular cells display mitochondrial decline: impaired oxidative phosphorylation, elevated ROS production, altered calcium handling, and increased apoptotic signaling. Melatonin counters these effects through several converging pathways:
➔ It preserves mitochondrial membrane integrity by stabilizing cardiolipin and preventing the opening of the mitochondrial permeability transition pore.
➔ It activates sirtuins, particularly SIRT1 and SIRT3, which restore redox balance, support mitophagy, and inhibit pro-apoptotic signaling (↑ Bcl-2, ↓ Bax).
➔ In myocardial aging and ischemia-reperfusion models, melatonin reduced infarct size, restored NAD⁺/NADH balance, and improved mitochondrial dynamics.
➔ It suppressed inflammatory cytokines like IL-1β and IL-6, while upregulating Nrf2 and improving endothelial nitric oxide (NO) signaling.
➔ In vascular smooth muscle cells, melatonin prevented senescence and calcification via exosomal microRNAs (miR-204/211).
➔ It enhanced telomerase activity in vascular tissue and reduced oxidative DNA damage—delaying senescence at the genomic level.
The cumulative effect is restoration of mitochondrial efficiency, suppression of inflammaging, protection of endothelial function, and reduced risk of age-related arrhythmia, fibrosis, and vascular stiffness.
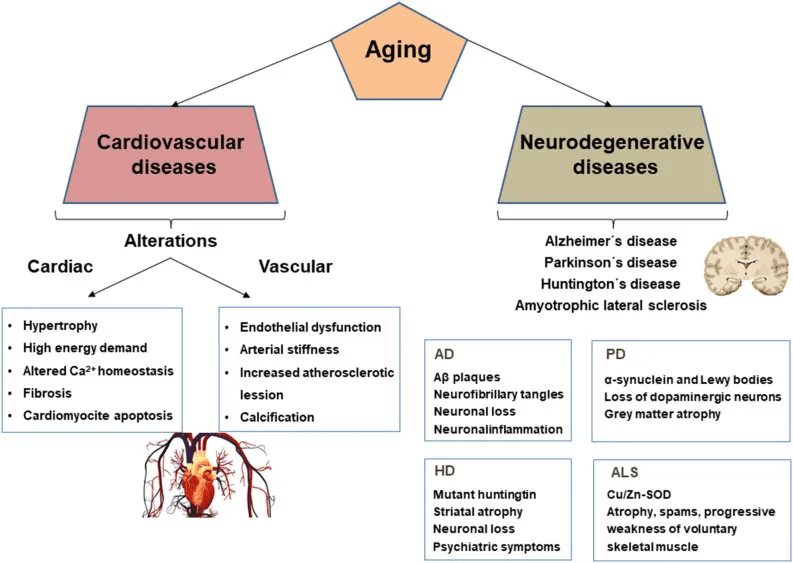
➤ Neurodegenerative Aging: Melatonin, Synaptic Resilience, Autophagy, and Neuroinflammation
Neurons, due to their high metabolic demands, are particularly vulnerable to redox imbalance, mitochondrial decline, and excitotoxicity. With aging, these stressors are compounded by protein misfolding, impaired autophagy, and loss of neurotrophic signaling.
Melatonin acts at several points in these pathological cascades:
➔ It boosts sirtuin activity (SIRT1, FOXO1/3a), enhancing autophagy, reducing apoptosis, and preserving mitochondrial function.
➔ It suppresses NF-κB, NLRP3 inflammasome, and glial activation—curbing neuroinflammation.
➔ It protects blood-brain barrier integrity via NADPH oxidase inhibition, AMPK activation, and reduced MMP-9 expression.
➔ In Alzheimer’s disease models, melatonin reduces amyloid and tau pathology by restoring mitophagy and regulating phosphorylation/dephosphorylation cycles.
➔ It supports synaptic plasticity via upregulation of BDNF, NMDA receptor subunits, and CAMKII, while reducing astrocytic and microglial activation.
➔ In Parkinson’s models, it attenuates α-synuclein aggregation, preserves dopaminergic neurons, and modulates microglia-mediated neurotoxicity.
➔ Post-stroke, it limits damage by activating SIRT1, reducing pro-inflammatory cytokines, and inhibiting apoptotic pathways.
Each of these actions converges on a central mechanism: the defense and restoration of mitochondrial and genomic integrity in aging neural circuits.
Summary chart of melatonin’s anti-aging effects, Description: Mechanism-specific table highlighting melatonin’s action across mitochondrial function, inflammation, sirtuins, autophagy, and neurovascular protection:
| Target | Effect | Mechanism / Pathway |
|---|---|---|
| Mitochondria | Preserves membrane integrity, prevents permeability transition | Stabilizes cardiolipin, inhibits pore opening, ↑ SIRT3, ↓ cardiomyocyte apoptosis |
| Oxidative Stress | Reduces ROS and lipid peroxidation | ↑ Antioxidant enzymes: SOD, catalase, GPx, glutathione reductase |
| Inflammation | Suppresses chronic low-grade inflammation (“inflammaging”) | ↓ NF-κB, ↓ NLRP3 inflammasome, ↓ TLR4, ↑ IL-10, macrophage M1→M2 shift |
| Sirtuins (1, 3, 6) | Enhances mitochondrial function and gene repair | ↑ SIRT1/3/6 expression, ↑ FOXO1/3a activity, ↓ p53 and pro-apoptotic signals |
| Autophagy | Stimulates autophagic clearance of damaged mitochondria and proteins | SIRT1-mediated activation of autophagy pathways |
| Vascular Function | Improves endothelial NO production, reduces arterial stiffness | ↑ eNOS, ↓ iNOS, ↓ arginase II, ↓ oxidative degradation of NO |
| Telomere Biology | Delays senescence, preserves vascular cell function | ↑ Telomerase activity, ↓ ROS-mediated telomere shortening |
| Neuroprotection | Preserves blood-brain barrier integrity and neuronal viability | ↓ MMP-9, ↓ gp91phox, ↑ SIRT1/AMPK, ↓ IL-6, ↓ IL-1β, ↑ BDNF |
| Alzheimer’s/AD Pathology | Reduces Aβ and tau pathology, restores circadian regulation | Enhances mitophagy, inhibits tau phosphorylation, ↑ phosphatase activity |
| Parkinson’s/PD Pathology | Protects dopaminergic neurons, reduces α-synuclein aggregation | ↓ microglial activation, ↓ NLRP3, ↑ SIRT1, ↑ mitochondrial resilience |
| Cardioprotection (Aging Heart) | Restores mitochondrial dynamics, reduces hypertrophy, fibrosis, arrhythmia risk | ↓ β-MHC, ↓ IL-6/IL-1α, ↑ Nrf2, ↑ mitochondrial ultrastructure |
| Ischemia-Reperfusion Injury | Reduces infarct size, inhibits apoptosis | ↑ SIRT3, ↑ Bcl-2, ↓ Bax, ↑ FOXO1 signaling |
| Circadian Regulation | Stabilizes melatonin rhythms disrupted by aging and light exposure | Prevents pineal dysfunction, counters light-at-night-induced oxidative stress |
| DNA Damage / Repair | Enhances genomic maintenance | ↑ APE1 and OGG1 (base excision repair enzymes) |
| Gut-Microbiome Axis | Enhances mitochondrial function via butyrate + gut melatonin synthesis | Butyrate → ↑ melatonin via upregulated melatonergic pathway |
| Stroke Protection | Reduces infarct volume, preserves cognitive function post-ischemia | ↓ IL-1β, ↓ GFAP, ↑ SIRT1, ↑ BDNF, ↑ NMDA receptor subunits |
| Calcium Homeostasis | Improves cardiomyocyte relaxation and brain Ca2+ balance | Restores SERCA2b function, ↓ Cav2.3 channel activity (in PD model) |
| Klotho Upregulation | Anti-aging, antioxidant, neuroprotective | ↑ Klotho gene expression |
| Apoptosis Suppression | Inhibits age-related programmed cell death | ↑ Bcl-2, ↓ Caspase-3/9, ↓ p53, ↓ nucleosomes in brain and heart |
Melatonin’s regulatory effects span systems but converge on a consistent anti-aging architecture: mitochondrial protection, redox balance, and suppression of chronic inflammation.
Melatonin’s protective footprint spans neural, vascular, and metabolic systems—but the outcomes in humans depend on several variables: dose, timing, baseline melatonin levels, and underlying physiological state
Which raises an important question:
➔ If melatonin exhibits such wide-reaching antioxidant benefits, should healthy individuals supplement with it routinely—or even in high doses, regardless of whether they use it for sleep?
➔ If someone were using it for sleep, should they stop if it doesn’t work, or continue due to its potential non-sleep related benefits?
That’s where the data gets more complicated.
➔ Up next: What the evidence reveals about metabolic, neurological, and hormonal risks associated with melatonin use.
Get Weekly Sleep Insights In Your Inbox. Beyond Melatonin, Blue Light, and Caffeine.
What Are the Risks of High-Dose Melatonin on Metabolism, Brain Function, and Hormones?
While melatonin’s antioxidant and anti-aging properties have attracted significant attention, emerging research shows that its benefits—and risks—depend heavily on variables like dose, timing of dose, baseline melatonin levels, and the user’s metabolic or immune status
And contrary to what cursory internet look-up suggests, melatonin’s effects are not limited to daytime sleepiness or headache.
Rather, exogenous melatonin—especially with high or repeated doses—may
- impair glucose regulation,
- reduce synaptic plasticity,
- disrupt reproductive hormones,
- alter immune signaling,
- promote oxidative stress
To understand where these concerns arise, we need to examine the systems most sensitive to exogenous melatonin—starting with glucose metabolism.
How Does Melatonin Affect Glucose Metabolism and Insulin Sensitivity?
Exogenous melatonin has been shown to interfere with glucose regulation in both healthy individuals and people with metabolic disease.
➤ Healthy adults — melatonin and glucose metabolism: OGTT impairment, timing, and insulin dynamics
In a controlled study of 21 healthy adults (mean age 24, BMI ~23), a 5 mg dose of melatonin taken before a glucose tolerance test significantly impaired glycemic control compared to placebo. When administered in the morning, melatonin raised glucose AUC (area under the curve) by approximately 186% and peak glucose by 21%. Even when taken in the evening, it still caused a 54% increase in AUC.
Mechanistically, the impairment was time-dependent: morning melatonin reduced insulin secretion, while evening melatonin reduced insulin sensitivity.
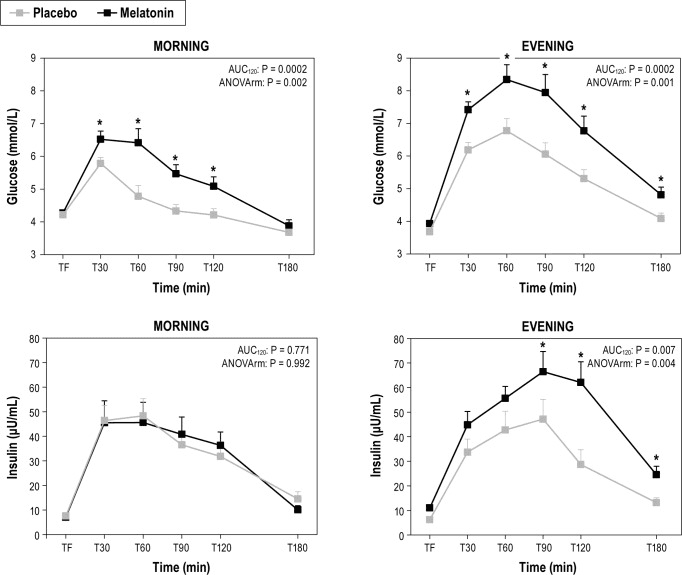
These results highlight that proximity to food intake plays a critical role in how melatonin influences glucose metabolism. Even in young and healthy individuals, melatonin may acutely worsen glucose tolerance depending on dosage and timing.
➤ Type 2 diabetes — high-dose melatonin risks: reduced insulin sensitivity and secretion
In metabolically vulnerable populations, melatonin’s effects appear even more pronounced.
A double‐blinded, randomized, placebo‐controlled, crossover trial involving 17 men with type 2 diabetes found that taking 10 mg of melatonin nightly, 1-hour before bedtime for 3-months reduced insulin sensitivity by ~12%.
The reduction in insulin sensitivity was assessed by an intravenous glucose tolerance test (0.3 g/kg) and measured by hyperinsulinemic-euglycemic clamp—a gold-standard method where insulin is infused to maintain steady glucose levels, allowing precise assessment of how much glucose the body absorbs in response.
This was also accompanied by reductions in glucose-stimulated insulin secretion and suggests that melatonin negatively affected both insulin release and peripheral sensitivity.
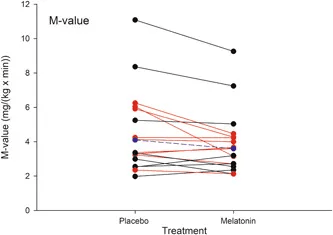
The authors concluded that high-dose melatonin should be used cautiously in people with existing glucose dysregulation, given the potential for exacerbating insulin resistance.
However, beyond metabolic effects, melatonin also interacts with neural pathways in the brain—especially those involved in plasticity and memory.
Want the step-by-step plan I use with readers 50+ to restore sleep?
How Can High-Dose Melatonin Affect Brain Plasticity and Cognitive Function?
That concern becomes more relevant when we consider melatonin’s widespread presence in the brain—particularly outside its role in sleep regulation.
Melatonin’s distribution in the central nervous system (CNS) extends well beyond the suprachiasmatic nucleus—the brain’s master clock that synchronizes circadian rhythms in response to light. It acts through two primary receptor types, MT₁ and MT₂—proteins on the surface of neurons that respond to melatonin and trigger downstream signaling.
MT₂ receptors, in particular, are found throughout the brain, including the hippocampus, amygdala, and olfactory structures—suggesting melatonin may influence neural processes outside circadian regulation.
One potentially concerning effect is melatonin’s suppression of long-term potentiation (LTP), a synaptic mechanism critical for learning and memory.
In mouse hippocampal slice experiments, exposure to 100 nM melatonin reduced LTP magnitude by approximately 50%. The suppression was dose-dependent, and involved inhibition of the cAMP/PKA signaling pathway via MT₂ receptors.
Given that LTP is a key process in memory consolidation, these findings raise questions about melatonin’s impact on cognitive function—especially under chronic, high-dose conditions.
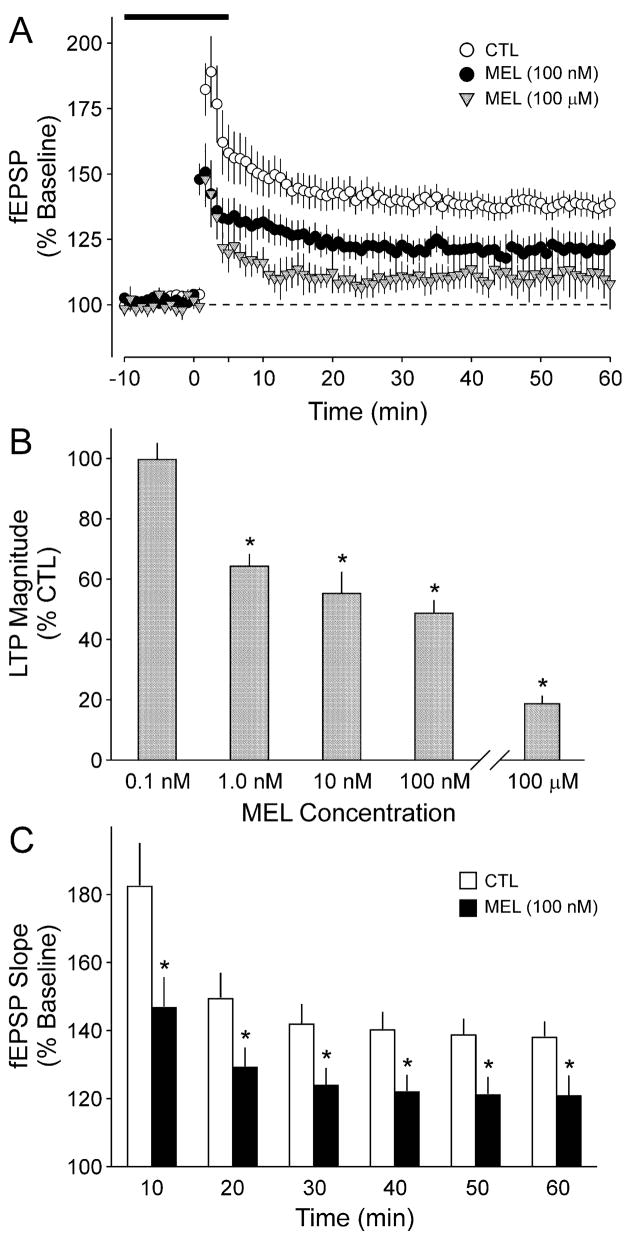
The idea that melatonin could dampen neuroplasticity seems to remain under-explored at this time but mechanistically plausible.
Further studies are needed to evaluate whether exogenous melatonin—especially taken during waking hours or in pharmacological doses—may unintentionally impair memory formation or learning capacity.
Although data on learning circuits remain limited, the presence of melatonin receptors in multiple brain regions invites a broader question: if melatonin can suppress signaling in the hippocampus, what are its effects in other tissues that also express its receptors?
One system where this question is also relevant is the reproductive axis.
How Does Melatonin Affect Reproductive Hormones and Fertility?
Melatonin’s influence on reproductive signaling is more directly supported by existing research. In humans, MT₁ and MT₂ receptors are present throughout the hypothalamic-pituitary-gonadal (HPG) axis: in the hypothalamus (regulating gonadotropin-releasing hormone, or GnRH), the anterior pituitary (modulating the release of luteinizing hormone [LH] and follicle-stimulating hormone [FSH], which govern ovulation and testosterone production), and the gonads—including both testes and ovaries—where they may influence steroid hormone production and local reproductive signaling.
Human studies have explored whether this receptor distribution translates into functional changes in reproductive hormones and fertility outcomes.
➤ Human studies — melatonin and reproductive hormones: semen quality, aromatase, and HPG axis signaling
In a double-blind crossover study, 8 healthy men were given either 3 mg of melatonin or placebo daily for 6-months. Semen quality, serum and seminal hormone levels, and gonadotropins were assessed throughout.
In 6 of the participants, no significant changes were noted. However, 2 men showed a marked decline in sperm concentration (to 3 × 10⁶/mL and 12 × 10⁶/mL, respectively) and motility (to 32% and 30%).
After discontinuing melatonin for 6 months, sperm parameters returned to baseline in one of the affected men, but remained abnormal in the other. The authors proposed that melatonin may inhibit testicular aromatase activity, thereby reducing estradiol synthesis without altering gonadotropin levels.
In rare clinical cases, elevated melatonin levels have been linked to delayed puberty or hypogonadism—a condition characterized by reduced sex hormone production and impaired sexual development. While not conclusive, these observations raise the possibility that exogenous melatonin at standard over-the-counter doses could impact male reproductive health—even in the absence of changes to upstream pituitary hormones.
➤ Animal models — photoperiod signaling: melatonin, LH/FSH suppression, and fertility timing
In seasonally breeding mammals, melatonin plays a role in regulating fertility according to photoperiod. In long-day breeders such as hamsters or sheep, elevated melatonin during extended dark periods suppresses the reproductive axis. This involves inhibition of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) secretion from the pituitary. Conversely, in short-day breeders, melatonin acts as a cue to initiate reproduction during winter months.
When administered out of phase with normal circadian or seasonal rhythms, melatonin can suppress gonadotropin levels and induce hypogonadotropic states in animal models. This suggests a plausible pathway by which chronic or high-dose melatonin could blunt reproductive signaling in humans as well.
While human data remain limited, these animal models highlight how sensitive reproductive signaling can be to melatonin’s timing and dose. So far, we’ve examined its impact on glucose metabolism, neural signaling, and reproductive hormones—all systems where dose and timing matter.
But that’s not the full picture.
Melatonin also interacts with the immune system—where its signaling appears both therapeutically promising and, under some conditions, potentially disruptive.
Get Weekly Sleep Strategies In Your Inbox.
When Does Melatonin Produce Pro-Inflammatory Rather Than Anti-Inflammatory Effects?
Melatonin is widely recognized for its anti-inflammatory properties.
However, there are evidence that suggest its effects on the immune system are bidirectional—dependent on context, timing, and immune status.
In isolated immune cells, including human leukocytes and Th1/Th17 lymphocytes, melatonin has been shown to increase production of several pro-inflammatory cytokines. These include IL‑1β, IL‑2, IL‑6, TNF‑α, IFN‑γ, and IL‑17. This stimulatory effect may reflect a physiological role in priming the immune system—particularly in the early morning hours, and is not necessarily a pathological process.
In autoimmune diseases such as rheumatoid arthritis (RA), however, this same mechanism may become maladaptive. RA patients have been found to exhibit elevated melatonin levels during the night and early morning—timing that coincides with symptom flares like joint stiffness and increased inflammatory activity. In this setting, melatonin may amplify Th1 and Th17 responses, intensifying inflammation rather than resolving it, and as a result, exacerbate joint damage and discomfort.
This dual behavior—anti-inflammatory under some conditions, pro-inflammatory under others—emerges across multiple models:
| Context | Inflammatory Role | Mechanism / Notes |
|---|---|---|
| In vitro – human leukocytes, lymphocytes (Th1/Th17) | Pro-inflammatory | Upregulates IL‑1β, IL‑2, IL‑6, TNF‑α, IFN‑γ, IL‑17; enhances immune readiness. |
| Autoimmune diseases (esp. RA) | Pro-inflammatory | Increases immune activation; elevated nighttime melatonin aligns with worsened morning symptoms. |
| Circadian variation in RA patients | Pro-inflammatory | Melatonin peaks correlate with increased symptom severity. |
| Acute inflammation (e.g., sepsis, I/R injury) | Anti-inflammatory | Suppresses iNOS, COX‑2, NF‑κB, TLR4, and NLRP3 inflammasome. |
| Neuroinflammation (e.g., Alzheimer’s models) | Anti-inflammatory | Reduces oxidative stress and cytokine load; limits microglial overactivation. |
| Aging-related low-grade inflammation | Anti-inflammatory | Lowers TNF‑α and IL‑6 through SIRT1 and Nrf2 activation. |
| Clinical trials in osteoarthritis (OA) | Anti-inflammatory | Reduces TNF‑α, IL‑8, VEGF via MT1 receptor pathways. |
| COVID-19, metabolic disease, obesity | Anti-inflammatory | Modulates immune response, suppresses systemic inflammation (e.g., CRP, cytokine storm). |
These findings suggest that melatonin’s immune effects are context-sensitive. In individuals with autoimmune predisposition or heightened immune tone, melatonin may unintentionally exacerbate inflammatory signaling.
A similar pattern of dual effects has been observed in another core system: redox balance. Although melatonin is typically described as an antioxidant, under certain cellular or metabolic conditions, it may instead promote oxidative activity.
➔ In the next section, we examine how melatonin’s redox behavior can shift from protective to potentially pro-oxidant.
How Can High-Dose Melatonin Switch from Antioxidant to Pro-Oxidant?
Melatonin is most often described as an antioxidant, with well-documented effects on mitochondrial protection and oxidative stress reduction.
However, under certain conditions—especially at high concentrations—it has demonstrated the opposite effect: promoting the generation of reactive oxygen species (ROS).
In cell culture studies using pharmacological doses of melatonin (in the micromolar to millimolar range), melatonin has been shown to increase intracellular ROS levels. These pro-oxidant effects were observed in both tumor and non-tumor cell lines.
Mechanistically, melatonin may promote ROS formation by:
- Activating calmodulin, which stimulates phospholipase A2 (PLA2) and downstream 5-lipoxygenase (5‑LOX), leading to pro-oxidant lipid mediators.
- Interacting directly with mitochondrial complex III or the mitochondrial permeability transition pore (MPTP), potentially enhancing electron leak and ROS production.
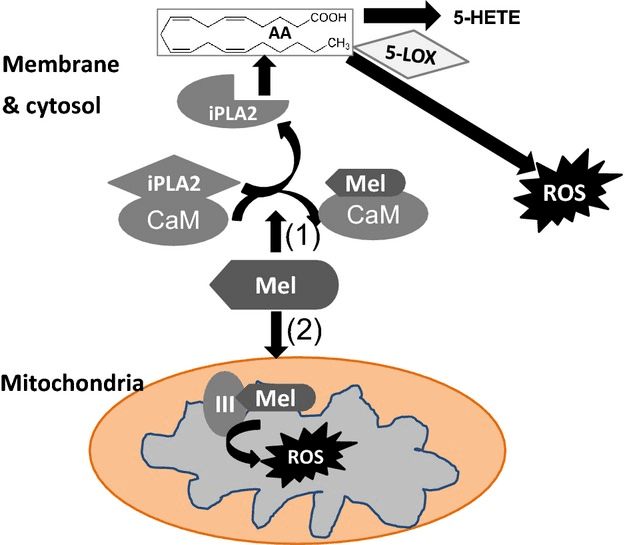
These findings raise the possibility that at high enough concentrations—melatonin could exert cytotoxic or pro-oxidant effects. However, such effects have mainly been reported in vitro, and researchers caution that “whether the reported in vitro pro-oxidant actions come into play in live organisms remains to be established.”
| Condition | Outcome |
|---|---|
| Physiological doses, in vivo | Strongly antioxidant and protective |
| Pharmacological doses, in vivo | Still antioxidant; pro‑oxidant not observed |
| High concentrations in vitro (cells) | Can be pro-oxidant → ROS increase, apoptosis |
| Mechanism | Calmodulin activation, complex III/MPTP involvement for ROS; in vivo, enzyme modulation and mitochondrial support for protection |
In summary, while melatonin is typically antioxidant under physiological conditions, the possibility of dose-dependent pro-oxidant behavior in cells invites careful consideration. Although pro-oxidant effects have not been demonstrated in humans, their biological plausibility warrants attention, especially in the context of high-dose supplementation.
Absence of evidence is not evidence of absence.
This brings us to a more grounded question: given the range of mechanisms covered so far—metabolic, neural, endocrine, immune, and oxidative—what do we actually know about the safety of melatonin supplementation in humans, especially at doses that exceed endogenous levels?
➔ With these concerns in view, the next section examines clinical safety data—what’s been observed, what’s still unclear, and where the research falls short.
What Do Trials and Meta-Analyses Show About High-Dose Melatonin Safety?
Although melatonin is often perceived as harmless, formal safety data—especially for high doses, used over a long period of time—remain surprisingly sparse. Adverse event (AE) reporting is inconsistent across trials, and only a minority of studies meet rigorous criteria for bias control.
Melatonin users are the unwitting subjects in a large-scale uncontrolled experiment. NIH, 1996
In a 2022 systematic review and meta-analysis by Menczel Schrire et al., 79 studies encompassing 3,861 participants were assessed for safety outcomes. These included individuals with a broad range of health conditions, and melatonin dosages varied substantially. Notably:
- 29 of the 79 studies (37%) provided no adverse event (AE) data at all.
- Only 4 out of the 79 studies met low-risk-of-bias criteria sufficient for quantitative synthesis.
Within this rigorously selected subset, melatonin did not increase serious adverse events (SAEs) or withdrawals due to AEs:
- SAEs: Rate Ratio = 0.88 [0.52, 1.50], p = .64
- Withdrawals due to AEs: RR = 0.93 [0.24, 3.56], p = .92
However, melatonin did increase the rate of general AEs, such as drowsiness, dizziness, and headache:
- Overall AE risk: RR = 1.40 [1.15, 1.69], p < .001
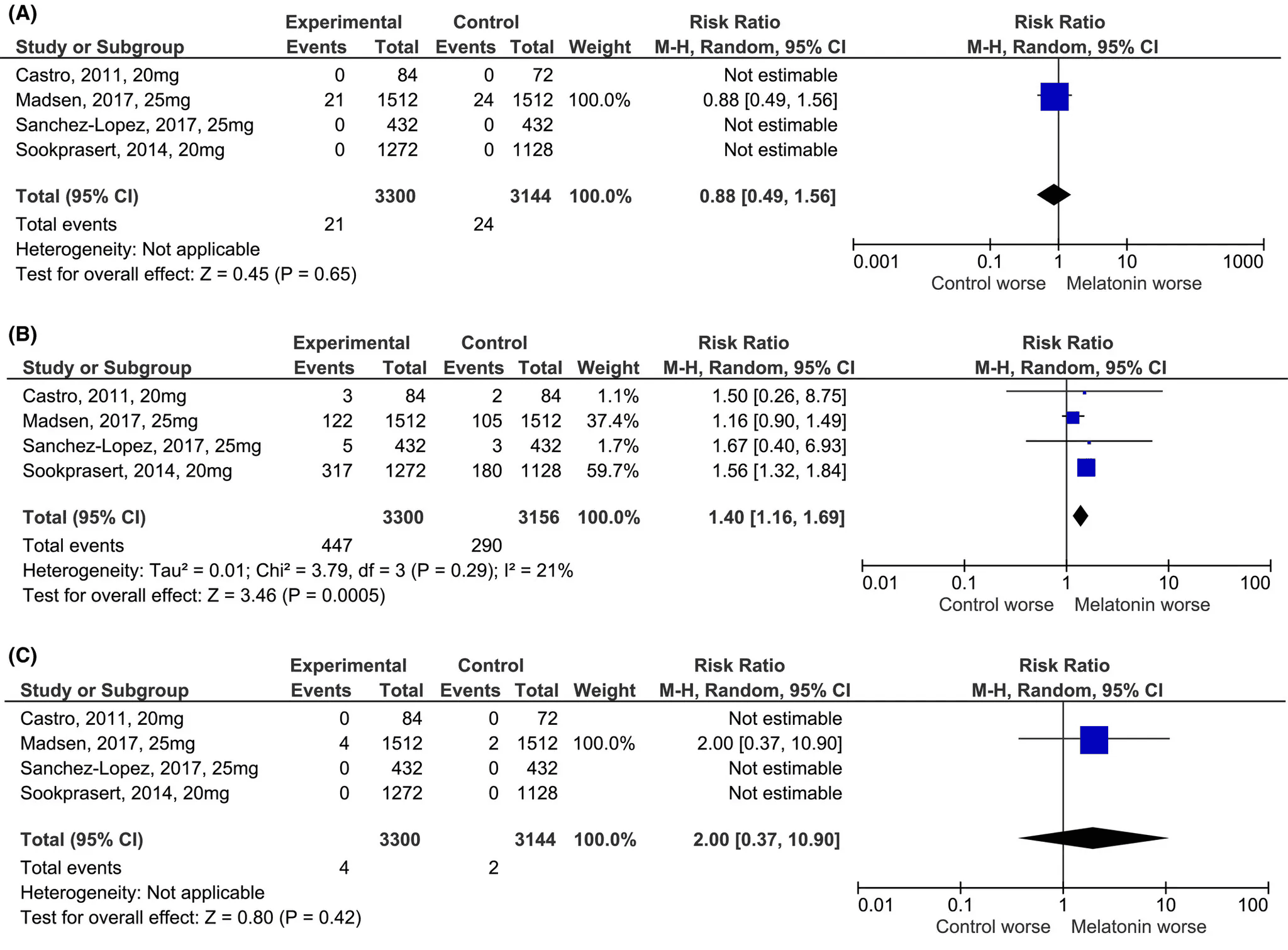
The researchers note that while melatonin appears safe in the short term, especially for common side effects, its long-term safety remains inadequately evaluated—particularly at high doses. They cite the NIH’s 1996 warning that melatonin users may be participating in a “large-scale uncontrolled experiment,” and conclude that additional, long-duration trials with comprehensive AE tracking are still needed.
Twenty-five years later, the current study still finds that additional studies in adults, using higher doses for longer durations are needed, with comprehensive reporting on AEs to confirm the current widespread belief that melatonin is indeed safe. Menczel Schrire 2022
Subsequent reviews have expanded on this question.
In a 2023 review by Givler et al., low-to-moderate daily doses (typically ≤5–6 mg) were described as “generally safe,” with long-term benefits observed in some patient groups, including individuals with autism. However, here, the researchers also emphasized that data on chronic high-dose use are still limited, especially in the context of aging and neurodegenerative prevention.
How Should You Weigh Melatonin’s Longevity Benefits Against Its Sleep and Metabolic Risks?
Melatonin has been studied as both a circadian hormone and a longevity candidate. The mechanistic promise is clear, but so are the gaps: long-term, high-dose use hasn’t been well tested, and risks appear when dose, timing, or baseline physiology are mismatched.
That means whether you’re thinking about melatonin for sleep or for longevity, the decision has to be intentional and measured. The real question isn’t just “does it work?” but “does it work for me — in this context, at this dose, at this timing?” And if it doesn’t, what else explains poor sleep or rising inflammation?
Next step. That’s the role of Vault Sleep OS: a system that helps you interpret your melatonin rhythms, place supplementation in context, and troubleshoot other drivers of sleep issues — when melatonin isn’t the lever.
Sleep OS Hormones is now available as a 60-day self-guided program with dedicated systems for estrogen, progesterone, and testosterone, or bundled together for a more complete approach.
Frequently Asked Questions
Does melatonin affect blood sugar?
In a controlled study of 21 healthy adults, a 5 mg dose before a glucose tolerance test raised glucose AUC by approximately 186% when taken in the morning and 54% when taken in the evening. Morning melatonin reduced insulin release; evening melatonin reduced insulin sensitivity. In a double-blinded, placebo-controlled crossover trial of 17 men with type 2 diabetes, 10 mg nightly for three months reduced insulin sensitivity by approximately 12%, assessed by hyperinsulinemic-euglycemic clamp.
Does melatonin have anti-aging benefits?
Melatonin accumulates in mitochondria and directly neutralizes reactive oxygen species and reactive nitrogen species, including hydroxyl, peroxyl, and nitric oxide radicals. It upregulates antioxidant enzymes like glutathione peroxidase, superoxide dismutase, and catalase, and activates sirtuins SIRT1 and SIRT3, which are linked to mitochondrial function and cellular resilience. Whether these mechanistic effects translate to meaningful longevity outcomes in healthy humans depends on dose, timing, and baseline health status.
Can melatonin affect memory or learning?
MT₂ receptors, through which melatonin acts, are present in the hippocampus—a brain region central to memory consolidation. In mouse hippocampal slice experiments, 100 nM melatonin reduced long-term potentiation magnitude by approximately 50%, through inhibition of the cAMP/PKA pathway via MT₂ receptors. Whether chronic or high-dose exogenous melatonin produces comparable effects in humans has not been established, but the mechanism is present and dose-dependent in these models.
Is high-dose melatonin safe to take regularly?
High-dose melatonin—5 mg or more—has been shown to impair glucose tolerance even in healthy adults, and 10 mg nightly for three months reduced insulin sensitivity in men with type 2 diabetes. Some data also suggest possible interference with reproductive hormones and immune homeostasis, particularly in vulnerable populations. These effects depend heavily on dose, timing, and the individual’s metabolic or immune status.
References
- Rubio-Sastre P, Scheer FA, Gómez-Abellán P, Madrid JA, Garaulet M. Acute melatonin administration in humans impairs glucose tolerance in both the morning and evening. Sleep. 2014 Oct 1;37(10):1715-9. doi: 10.5665/sleep.4088. PMID: 25197811; PMCID: PMC4173928.
- Candyce Hamel, Jennifer Horton, CADTH Health Technology Review, Melatonin for the Treatment of Insomnia: A 2022 Update 3
- Givler D, Givler A, Luther PM, Wenger DM, Ahmadzadeh S, Shekoohi S, Edinoff AN, Dorius BK, Jean Baptiste C, Cornett EM, Kaye AM, Kaye AD. Chronic Administration of Melatonin: Physiological and Clinical Considerations. Neurol Int. 2023 Mar 15;15(1):518-533. doi: 10.3390/neurolint15010031. PMID: 36976674; PMCID: PMC10053496.
- Tuft C, Matar E, Menczel Schrire Z, Grunstein RR, Yee BJ, Hoyos CM. Current Insights into the Risks of Using Melatonin as a Treatment for Sleep Disorders in Older Adults. Clin Interv Aging. 2023 Jan 12;18:49-59. doi: 10.2147/CIA.S361519. PMID: 36660543; PMCID: PMC9842516.
- Gobbi G, Comai S. Differential Function of Melatonin MT1 and MT2 Receptors in REM and NREM Sleep. Front Endocrinol (Lausanne). 2019 Mar 1;10:87. doi: 10.3389/fendo.2019.00087. PMID: 30881340; PMCID: PMC6407453.
- Baskett JJ, Broad JB, Wood PC, Duncan JR, Pledger MJ, English J, Arendt J. Does melatonin improve sleep in older people? A randomised crossover trial. Age Ageing. 2003 Mar;32(2):164-70. doi: 10.1093/ageing/32.2.164. PMID: 12615559.
- SMYTHE, G., LAZARUS, L. Growth Hormone Regulation by Melatonin and Serotonin. Nature 244, 230–231 (1973). https://doi.org/10.1038/244230a0
- Duffy JF, Wang W, Ronda JM, Czeisler CA. High dose melatonin increases sleep duration during nighttime and daytime sleep episodes in older adults. J Pineal Res. 2022 Aug;73(1):e12801. doi: 10.1111/jpi.12801. Epub 2022 May 13. PMID: 35436355; PMCID: PMC9288519.
- Luboshitzky R, Shen-Orr Z, Nave R, Lavi S, Lavie P. Melatonin administration alters semen quality in healthy men. J Androl. 2002 Jul-Aug;23(4):572-8. PMID: 12065466.
- Buscemi N, Vandermeer B, Pandya R, Hooton N, Tjosvold L, Hartling L, Baker G, Vohra S, Klassen T. Melatonin for Treatment of Sleep Disorders. Evidence Report/Technology Assessment No. 108. (Prepared by the University of Alberta Evidence-based Practice Center, under Contract No. 290-02-0023.) AHRQ Publication No. 05-E002-2. Rockville, MD: Agency for Healthcare Research and Quality. November 2004.
- Wang LM, Suthana NA, Chaudhury D, Weaver DR, Colwell CS. Melatonin inhibits hippocampal long-term potentiation. Eur J Neurosci. 2005 Nov;22(9):2231-7. doi: 10.1111/j.1460-9568.2005.04408.x. PMID: 16262661; PMCID: PMC2581482.
- Valcavi R, Zini M, Maestroni GJ, Conti A, Portioli I. Melatonin stimulates growth hormone secretion through pathways other than the growth hormone-releasing hormone. Clin Endocrinol (Oxf). 1993 Aug;39(2):193-9. doi: 10.1111/j.1365-2265.1993.tb01773.x. PMID: 8370132.
- Comai S, Gobbi G. Melatonin, Melatonin Receptors and Sleep: Moving Beyond Traditional Views. J Pineal Res. 2024 Oct;76(7):e13011. doi: 10.1111/jpi.13011. PMID: 39400423.
- Ferracioli-Oda E, Qawasmi A, Bloch MH. Meta-analysis: melatonin for the treatment of primary sleep disorders. PLoS One. 2013 May 17;8(5):e63773. doi: 10.1371/journal.pone.0063773. PMID: 23691095; PMCID: PMC3656905.
- Lelak K, Vohra V, Neuman MI, Toce MS, Sethuraman U. Pediatric Melatonin Ingestions – United States, 2012-2021. MMWR Morb Mortal Wkly Rep. 2022 Jun 3;71(22):725-729. doi: 10.15585/mmwr.mm7122a1. Erratum in: MMWR Morb Mortal Wkly Rep. 2022 Jul 08;71(27):885. doi: 10.15585/mmwr.mm7127a4. PMID: 35653284; PMCID: PMC9169525.
- Menczel Schrire Z, Phillips CL, Chapman JL, Duffy SL, Wong G, D’Rozario AL, Comas M, Raisin I, Saini B, Gordon CJ, McKinnon AC, Naismith SL, Marshall NS, Grunstein RR, Hoyos CM. Safety of higher doses of melatonin in adults: A systematic review and meta-analysis. J Pineal Res. 2022 Mar;72(2):e12782. doi: 10.1111/jpi.12782. Epub 2021 Dec 30. PMID: 34923676.
- Wyatt JK, Dijk DJ, Ritz-de Cecco A, Ronda JM, Czeisler CA. Sleep-facilitating effect of exogenous melatonin in healthy young men and women is circadian-phase dependent. Sleep. 2006 May;29(5):609-18. doi: 10.1093/sleep/29.5.609. PMID: 16774150.
- Lauritzen ES, Kampmann U, Pedersen MGB, Christensen LL, Jessen N, Møller N, Støy J. Three months of melatonin treatment reduces insulin sensitivity in patients with type 2 diabetes-A randomized placebo-controlled crossover trial. J Pineal Res. 2022 Aug;73(1):e12809. doi: 10.1111/jpi.12809. Epub 2022 Jun 9. PMID: 35619221; PMCID: PMC9540532.
- Marupuru S, Arku D, Campbell AM, Slack MK, Lee JK. Use of Melatonin and/on Ramelteon for the Treatment of Insomnia in Older Adults: A Systematic Review and Meta-Analysis. J Clin Med. 2022 Aug 31;11(17):5138. doi: 10.3390/jcm11175138. PMID: 36079069; PMCID: PMC9456584.
- Reiter RJ, Mayo JC, Tan DX, Sainz RM, Alatorre-Jimenez M, Qin L. Melatonin as an antioxidant: under promises but over delivers. J Pineal Res. 2016;61(3):253-278. doi:10.1111/jpi.12360. PMID: 27500468.
- Ferlazzo N, Andolina G, Cannata A, et al. Is Melatonin the Cornucopia of the 21st Century? Antioxidants (Basel). 2020;9(11):1088. doi:10.3390/antiox9111088. PMID: 33167396; PMCID: PMC7694322.
- Martín Giménez VM, de Las Heras N, Lahera V, Tresguerres JAF, Reiter RJ, Manucha W. Melatonin as an Anti-Aging Therapy for Age-Related Cardiovascular and Neurodegenerative Diseases. Front Aging Neurosci. 2022;14:888292. doi:10.3389/fnagi.2022.888292. PMID: 35721030; PMCID: PMC9204094.
- Wang LM, Suthana NA, Chaudhury D, Weaver DR, Colwell CS. Melatonin inhibits hippocampal long-term potentiation. Eur J Neurosci. 2005;22(9):2231-2237. doi:10.1111/j.1460-9568.2005.04408.x. PMID: 16262661; PMCID: PMC2581482.
- (Receptor distribution review used in text.) Melatonin receptor MT1/MT2 distribution in the mouse brain (review). PubMed PMID: 30937953. https://pubmed.ncbi.nlm.nih.gov/30937953
- Hardeland R. Melatonin and inflammation—Story of a double-edged blade. J Pineal Res. 2018;65(4):e12525. doi:10.1111/jpi.12525. PMID: 30242884 https://pubmed.ncbi.nlm.nih.gov/30242884
- Zhang HM, Zhang Y. Melatonin: a well-documented antioxidant with conditional pro-oxidant actions. J Pineal Res. 2014;57(2):131-146. doi:10.1111/jpi.12162. PMID: 25060102. https://pubmed.ncbi.nlm.nih.gov/25060102
- Menczel Schrire Z, Phillips CL, Chapman JL, et al. Safety of higher doses of melatonin in adults: A systematic review and meta-analysis. J Pineal Res. 2022;72(2):e12782. doi:10.1111/jpi.12782. PMID: 34923676. https://pubmed.ncbi.nlm.nih.gov/34923676